Mastering GROMACS mdrun for Energy Minimization: A Practical Guide for Biomedical Researchers
This comprehensive guide details the use of the GROMACS mdrun command for energy minimization, a critical step in preparing stable molecular dynamics simulations of biomolecules and drug candidates.
Mastering GROMACS mdrun for Energy Minimization: A Practical Guide for Biomedical Researchers
Abstract
This comprehensive guide details the use of the GROMACS mdrun command for energy minimization, a critical step in preparing stable molecular dynamics simulations of biomolecules and drug candidates. It covers foundational principles of minimization algorithms, step-by-step methodology for setup and execution, advanced troubleshooting for common failures, and validation techniques to ensure simulation readiness. Tailored for researchers and drug development professionals, the article provides actionable strategies to overcome practical challenges, interpret results correctly, and establish robust simulation workflows for reliable biomedical research outcomes.
Understanding Energy Minimization: Why Your Simulation's Success Depends on It
The Critical Role of Energy Minimization in MD Simulations
Energy minimization (EM) is a foundational step in molecular dynamics (MD) simulations, crucial for ensuring system stability and reliability before proceeding to dynamics. This process removes unrealistic atomic clashes and inappropriate geometry inherited from the initial system construction, leading the configuration to the nearest local minimum on the potential energy surface. Without proper minimization, the high initial forces can cause simulation instability or even catastrophic failure. This application note details the protocols for performing energy minimization within the GROMACS mdrun framework, providing researchers and drug development professionals with the practical knowledge to robustly prepare their systems.
Theoretical Foundation of Energy Minimization
Energy minimization algorithms navigate the potential energy landscape to find a stable configuration. GROMACS provides three primary minimizers, each with distinct advantages [1].
The Steepest Descent algorithm follows the negative gradient of the potential energy. It is robust and efficient in the initial stages of minimization, making it ideal for relieving severe steric clashes in poorly equilibrated systems. The algorithm calculates new positions based on the force and a maximum displacement parameter, adaptively adjusting the step size: it increases upon successful steps that lower the energy and decreases upon unsuccessful ones [1]. Although not the most efficient near the minimum, its simplicity and stability make it a popular choice for initial minimization.
Conjugate Gradient methods are more sophisticated. They use information from previous steps to construct conjugate search directions, which generally leads to more efficient convergence than steepest descent, particularly closer to the energy minimum [1]. A key operational restriction is that it cannot be used with constraints in GROMACS; flexible water models are required if water is present. This makes it particularly suitable for minimizations preceding normal-mode analysis [1].
The L-BFGS (Limited-memory Broyden–Fletcher–Goldfarb–Shanno) algorithm is a quasi-Newton method. It builds an approximation of the inverse Hessian matrix, using a fixed number of corrections from previous steps to guide the minimization. This "sliding-window" technique makes it memory-efficient for large biomolecular systems. L-BFGS typically converges faster than conjugate gradients, though its parallelization is more complex. The presence of switched or shifted interactions can improve its convergence by reducing discontinuities in the potential [1].
Table 1: Comparison of Energy Minimization Algorithms in GROMACS
| Algorithm | Principle | Strengths | Weaknesses | Ideal Use Case |
|---|---|---|---|---|
| Steepest Descent [1] | Follows the negative energy gradient | Robust, easy to implement, efficient for initial stages | Slow convergence near minimum | Relieving severe steric clashes |
| Conjugate Gradient [1] | Uses conjugate directions from previous steps | More efficient than steepest descent near minimum | Cannot be used with constraints (e.g., SETTLE water) | Pre-normal-mode analysis minimization |
| L-BFGS [1] | Approximates the inverse Hessian matrix | Fast convergence, memory efficient for large systems | Not yet fully parallelized in GROMACS | General-purpose minimization for large biomolecules |
Practical Implementation withgmx mdrun
The GROMACS mdrun program is the computational engine that performs energy minimization. Its function is to read the run input file, distribute the topology, and execute the minimization algorithm, producing output files including a log file, a full-precision trajectory, an energy file, and a structure file containing the final minimized coordinates [2].
Input File Preparation
Execution begins with the grompp module, which assembles the structure, topology, and simulation parameters into a portable binary input file (.tpr) [3]. The parameters for the minimization are defined in a molecular dynamics parameters (.mdp) file. A typical minim.mdp file for steepest descent minimization is configured as follows [3] [4]:
Execution and Output Analysis
The minimization is executed by passing the .tpr file to mdrun [3]:
Here, the -v flag enables verbose output to monitor progress, and -deffnm em defines a common prefix (em) for all output files.
Successful minimization produces several output files [3]:
em.log: ASCII-text log file of the EM process.em.edr: Binary energy file.em.trr: Binary full-precision trajectory.em.gro: Energy-minimized structure.
Two critical factors must be evaluated to confirm successful minimization [3] [5]:
- Potential Energy (
Epot): This should be a large, negative value, typically on the order of 10⁵–10⁶ for a solvated protein system, indicating favorable non-bonded interactions. - Maximum Force (
Fmax): This must be below the specified tolerance (emtol, e.g., 1000 kJ mol⁻¹ nm⁻¹). Convergence is achieved when the maximum force is smaller than this value.
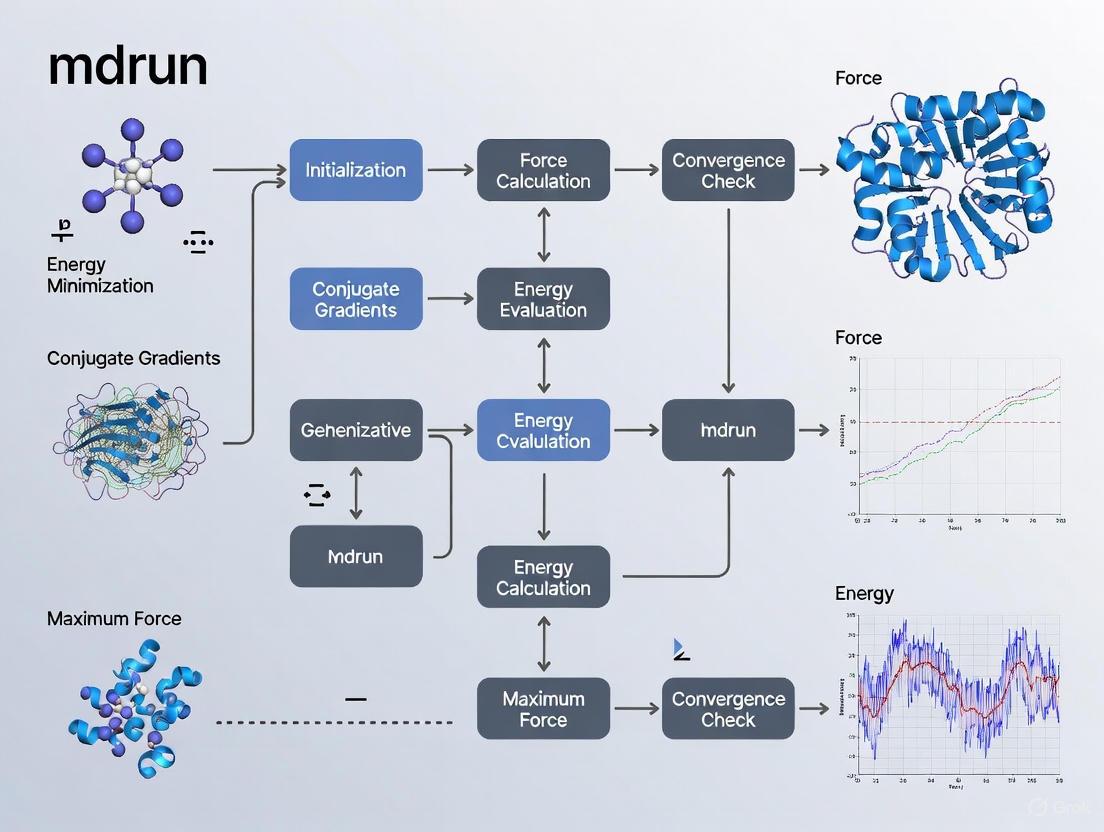
The following workflow diagram illustrates the complete energy minimization process in GROMACS, from system preparation to analysis.
Validation, Troubleshooting, and Advanced Protocols
Validation of Results
A steady decrease in potential energy throughout the minimization steps, as visualized from the em.edr file using the gmx energy module, is a strong indicator of proper convergence [3] [5]. The resulting potential.xvg file can be plotted to demonstrate this smooth energy decline.
Table 2: Success Criteria and Troubleshooting Guide for Energy Minimization
| Aspect to Check | Indicator of Success | Common Problem | Potential Solution |
|---|---|---|---|
Potential Energy (Epot) [3] [6] |
Large, negative value (e.g., -10⁵ to -10⁶) | Positive or insufficiently negative value | Check for charge imbalance; add counter-ions [6] |
Maximum Force (Fmax) [3] [4] |
Fmax < specified emtol |
Fmax remains above emtol |
Increase nsteps; try a different integrator [4] |
| Energy Plot [3] [5] | Smooth, monotonic decrease that plateaus | Energy oscillates or fails to decrease | Reduce emstep; check for severe steric clashes |
| Algorithm Convergence [4] | Completes with "converged to Fmax" message | Stops prematurely with "steps too small" | Verify force field compatibility; check topology |
Troubleshooting Common Issues
A frequent issue is when minimization stops because the algorithm can no longer take a step large enough to reduce the energy, yet the maximum force remains above the target emtol [4]. The output may state that it "converged to machine precision" but not to the requested Fmax. This often indicates residual, localized strains in the structure, such as improper bonding or steric parameters.
If one minimizer fails, a robust strategy is to use the output structure from a previous run as input for a new minimization, potentially with a different algorithm. For instance, one might start with L-BFGS for rapid initial progress, then switch to steepest descent, and finish with another round of L-BFGS for fine convergence [7]. This protocol, em_schedule, can be more effective than a single long minimization.
Segmentation faults during minimization or subsequent equilibration often point to deeper issues, such as problems with the system's topology or force field parameters, rather than the minimization parameters themselves [4]. Mismatched force fields for different molecules in a complex (e.g., protein, DNA, ions) can create such instability. Careful validation of the topology and ensuring force field compatibility for all components is essential.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Software and File Components for GROMACS Energy Minimization
| Tool/Reagent | Type | Primary Function |
|---|---|---|
gmx mdrun [2] |
GROMACS Module | Main computational engine that performs the energy minimization. |
.tpr File [3] [2] |
Run Input File | Portable binary file containing system coordinates, topology, and parameters for the run. |
.mdp File [3] [4] |
Parameter File | Plain-text file specifying the minimization algorithm, stopping criteria, and force calculation methods. |
| Force Field Files [8] [9] | Parameter Set | Defines the equations and parameters (masses, charges, bond constants, etc.) for calculating potential energy. |
gmx energy [3] |
Analysis Tool | Extracts and analyzes energy terms from the .edr file for plotting and validation. |
Energy minimization is a non-negotiable step in the MD workflow, ensuring that the simulation starts from a stable, low-energy configuration. A rigorous approach—selecting the appropriate algorithm, validating convergence through both potential energy and maximum force, and systematically troubleshooting failures—is fundamental for obtaining physically meaningful results. By mastering these protocols for the GROMACS mdrun command, researchers can reliably prepare robust systems for subsequent equilibration and production MD, laying a solid foundation for scientific discovery in computational biophysics and drug development.
Energy minimization (EM), also known as geometry optimization, is a fundamental computational process of finding an atomic arrangement where the net inter-atomic force on each atom is acceptably close to zero and the position on the potential energy surface (PES) is a stationary point [10]. In the context of molecular dynamics simulations with GROMACS, this process is crucial for removing steric clashes and inappropriate geometry that may have been introduced during system construction, thus ensuring system stability before proceeding to molecular dynamics simulations [3] [11]. The GROMACS mdrun program implements three principal algorithms for this purpose: Steepest Descent, Conjugate Gradient, and L-BFGS, each with distinct characteristics, performance profiles, and ideal application scenarios [12] [13].
The mathematical goal of energy minimization is to find the vector of atomic positions, r, that minimizes the potential energy function, E(r). This is achieved when the gradient (or force, F = -∂E/∂r) is sufficiently close to zero, indicating a local minimum on the potential energy surface [10]. The efficiency and effectiveness with which different algorithms achieve this minimization directly impact the preparatory phase of computational research in fields like drug development, where reliable initial structures are prerequisite to meaningful simulation.
Theoretical Foundations of the Algorithms
Steepest Descent
The Steepest Descent algorithm is a fundamental gradient-based optimization method known for its robustness and straightforward implementation [12] [13]. It operates by iteratively moving atomic coordinates in the direction of the negative energy gradient, which corresponds to the direction of the instantaneous, local maximum force. The core update formula in GROMACS is given by:
r{n+1} = rn + (hn / max(|Fn|)) * F_n
Here, rn represents the atomic coordinates at step *n*, Fn is the force vector, and hn is a maximum displacement parameter [12] [13]. The algorithm employs a heuristic approach to adjust this maximum step size: if a step decreases the potential energy (V{n+1} < Vn), the displacement is increased by 20% (h{n+1} = 1.2 hn) for the next iteration. Conversely, if the energy increases, the step is rejected and the displacement is reduced by 80% (hn = 0.2 h_n) [12] [13]. This simple mechanism ensures stability, making it particularly effective in the initial stages of minimization where the system may be far from a minimum and contain high-energy clashes.
Conjugate Gradient
The Conjugate Gradient (CG) method represents a more sophisticated approach that builds upon the principles of steepest descent. While it may be slower in the initial stages, it typically becomes more efficient as the system approaches the energy minimum [12] [13]. Unlike Steepest Descent, which can oscillate in narrow valleys of the potential energy surface, CG constructs a series of search directions that are conjugate to each other with respect to the Hessian (the matrix of second derivatives). This means that each step preserves the minimization achieved in previous directions, leading to more efficient convergence near the minimum.
In practice, this property allows CG to theoretically converge to a minimum in at most N steps for a quadratic problem in N dimensions, a significant improvement over Steepest Descent. It's important to note that in GROMACS, the Conjugate Gradient algorithm cannot be used with constraints, including the SETTLE algorithm for water. Therefore, any water present must be of a flexible model, which is specified in the MDP file using define = -DFLEXIBLE [13].
L-BFGS
The Limited-memory Broyden–Fletcher–Goldfarb–Shanno (L-BFGS) algorithm is a quasi-Newton method that offers a balance between computational efficiency and convergence performance [12] [13]. Newton's method uses the Hessian matrix to achieve faster convergence but calculating the exact Hessian is computationally prohibitive for large systems. The original BFGS algorithm circumvents this by successively building an approximation of the inverse Hessian, but its memory requirements scale with the square of the number of particles, making it impractical for biomolecular systems [12] [13].
L-BFGS, the limited-memory variant implemented in GROMACS, approximates the inverse Hessian using a fixed number of vector corrections from previous steps rather than storing a full dense matrix [12] [13]. This sliding-window technique maintains much of the efficiency of the original BFGS method while reducing memory requirements to a level proportional to the system size multiplied by the number of correction steps. In practice, L-BFGS has been found to converge faster than Conjugate Gradients, though its implementation in GROMACS is not yet parallelized due to the nature of its correction steps [12] [13].
Algorithm Workflows and Implementation
The following diagram illustrates the fundamental decision logic and workflow implemented by the mdrun command in GROMACS when performing energy minimization, highlighting the roles of the three core algorithms.
Steepest Descent Protocol
The Steepest Descent algorithm follows a specific iterative protocol within GROMACS. The process begins with the calculation of forces F and the potential energy V for the initial atomic configuration. New positions are then calculated using the update formula r{n+1} = rn + (hn / max(|Fn|)) * F_n. Following this position update, forces and energy are recomputed for the new configuration [12] [13].
The critical decision point in the algorithm involves evaluating the new potential energy, V{n+1}. If this energy is lower than the previous value (V{n+1} < Vn), the new positions are accepted, and the maximum displacement parameter is increased by 20% (h{n+1} = 1.2 hn) to accelerate convergence. If the energy instead increases or remains the same (V{n+1} ≥ Vn), the positional update is rejected, the system reverts to the previous coordinates, and the step size is substantially reduced to 20% of its previous value (hn = 0.2 h_n) to promote stability. This evaluate-adjust-recur process continues until the maximum force component falls below a specified tolerance (emtol) or a maximum number of steps is reached [12] [13].
GROMACS-Specific Implementation
The implementation of these algorithms in GROMACS is controlled through the mdrun command and a molecular dynamics parameter (MDP) file. The key parameter that selects the minimization algorithm is integrator, set to steep, cg, or l-bfgs for Steepest Descent, Conjugate Gradient, and L-BFGS, respectively [12] [11]. A standard command sequence to prepare and run a minimization is as follows:
The MDP file for energy minimization must specify the integrator, the force tolerance (emtol; typically 100-1000 kJ mol⁻¹ nm⁻¹), and the maximum number of iterations (nsteps; often 50-500 for Steepest Descent, potentially more for CG/L-BFGS) [3] [12]. Successful minimization is judged by two primary metrics: the potential energy (Epot) should be negative and on the order of 10⁵-10⁶ for a solvated protein system, and the maximum force (Fmax) must be below the specified emtol [3] [5].
Comparative Analysis and Performance
Quantitative Algorithm Comparison
The table below provides a structured comparison of the three energy minimization algorithms based on key performance and implementation characteristics.
| Algorithm Characteristic | Steepest Descent | Conjugate Gradient | L-BFGS |
|---|---|---|---|
| Initial Convergence Speed | Fast (early stages) [12] [13] | Slower (early stages) [12] [13] | Fast [14] |
| Final Convergence Efficiency | Less efficient (near minimum) [12] [13] | More efficient (near minimum) [12] [13] | Most efficient in practice [12] [13] |
| Memory Requirements | Low | Low | Moderate (proportional to system size × correction steps) [12] [13] |
| Robustness | High (good for poorly starting structures) [12] [13] | Moderate | High |
| Parallelization in GROMACS | Supported | Supported | Not yet parallelized [12] [13] |
| Typical Use Case | Initial stabilization, removing severe clashes [11] | Minimization prior to normal-mode analysis [13] | General-purpose, efficient minimization [12] [11] |
| Constraints Compatibility | Compatible | Requires flexible water (no constraints) [13] | Compatible |
Empirical Performance Data
A comparative study in intensity-modulated radiation therapy optimization provides insightful empirical data on the relative performance of these algorithm classes. The study found that L-BFGS was, on average, six times faster at reaching the same objective function value compared to a quasi-Newton algorithm, and also terminated at a final objective function value that was 37% lower on average [14]. The Conjugate Gradient algorithm ranked between the others, with an average speedup factor of two and an average improvement of the objective function value of 30% compared to the baseline quasi-Newton method [14]. While this study was conducted in a different domain, the relative performance trends for these standard numerical algorithms are consistent with those observed in molecular mechanics minimization.
Experimental Protocols and Application
The Scientist's Toolkit: Essential Research Reagents
The following table outlines the key computational "reagents" and components required to perform energy minimization experiments in GROMACS.
| Research Reagent / Component | Function / Purpose |
|---|---|
| Molecular Structure File (.gro, .pdb) | Provides the initial atomic coordinates of the system to be minimized. |
| Topology File (.top) | Defines the molecular composition, connectivity, and force field parameters. |
| Molecular Dynamics Parameters File (.mdp) | Contains all algorithmic settings (integrator, emtol, nsteps) controlling the minimization process. |
| GROMACS Binary Input File (.tpr) | The portable binary produced by grompp containing all simulation data for mdrun. |
| Force Field | A set of mathematical functions and parameters (e.g., CHARMM, AMBER, OPLS) defining the potential energy surface. |
| Solvent Model (e.g., SPC, TIP4P) | Defines the water molecules and ions in the system, critical for modeling biological environments. |
Standard Operating Procedure for Energy Minimization
The following workflow diagram outlines the complete experimental procedure for setting up and running an energy minimization in GROMACS, from system preparation to result validation.
A recommended hybrid protocol for complex biomolecular systems (e.g., a protein-ligand complex for drug development) involves a multi-stage approach to maximize efficiency and robustness. Step 1 involves running Steepest Descent for 50-500 steps with a relatively relaxed force tolerance (e.g., emtol = 1000) to quickly resolve the worst steric clashes and bring the system to a stable, low-energy basin [11]. Step 2 uses the output structure from Steepest Descent as the starting point for a more refined minimization with either Conjugate Gradient or L-BFGS. This second stage should use a tighter force tolerance (e.g., emtol = 10-100) to achieve a well-minimized structure suitable for subsequent simulation stages [11]. If the system contains flexible water or no constraints, Conjugate Gradient is a good choice; otherwise, L-BFGS is generally preferred for its faster convergence [12] [13] [14].
Validation and Analysis Methods
Validating the success of an energy minimization run is critical. Two primary factors must be evaluated post-execution. First, the potential energy (Epot) printed at the end of the process should be negative and, for a solvated protein system, typically on the order of 10⁵-10⁶, scaling with system size [3] [5]. Second, the maximum force (Fmax) on any atom must be below the target tolerance (emtol) specified in the MDP file [3] [5]. The evolution of the potential energy can be plotted using the GROMACS energy module to visualize convergence:
At the prompt, select "Potential" and then "0" to terminate input. The resulting XVG file can be visualized with tools like Xmgrace, showing a characteristic steady decrease in potential energy to a plateau, indicating convergence [3]. If minimization fails to meet these criteria (e.g., Fmax remains above emtol after the maximum number of steps), potential solutions include increasing nsteps, decreasing the step size (emstep for Steepest Descent), or re-evaluating the system for underlying structural issues [5].
In molecular dynamics (MD) simulations, the initial constructed molecular system often contains steric clashes and inappropriate geometry due to overlapping atoms or strained bonds introduced during the modeling process. Energy minimization (EM) is a critical preliminary step that removes these instabilities by iteratively adjusting atomic coordinates to find a local minimum on the potential energy surface. This process is essential for ensuring system stability before proceeding to dynamical simulations, as it reduces excessive forces that could otherwise cause simulation failure. Within the GROMACS package, the mdrun module serves as the primary computational engine responsible for executing this energy minimization, employing various algorithms to efficiently relax the molecular structure.
This application note details the operational workflow of gmx mdrun during energy minimization, providing researchers and drug development professionals with a comprehensive protocol for implementing this crucial step in their computational pipelines.
Algorithmic Foundations of Energy Minimization in GROMACS
The mdrun program in GROMACS implements three primary algorithms for energy minimization, each with distinct operational characteristics and suitability for different scenarios.
Steepest Descent
The Steepest Descent algorithm is a robust and straightforward method ideal for the initial stages of minimization when the system is far from equilibrium. It operates by moving atomic coordinates in the direction of the negative energy gradient (the force). The algorithm calculates new positions using:
[ \mathbf{r}{n+1} = \mathbf{r}n + \frac{hn}{\max(|\mathbf{F}n|)} \mathbf{F}_n ]
Where (hn) is the maximum displacement, (\mathbf{F}n) is the force vector, and (\max(|\mathbf{F}n|)) represents the largest scalar force on any atom [1]. The step size is dynamically adjusted based on energy changes: it increases by 20% ((h{n+1} = 1.2 hn)) if the energy decreases, and decreases by 80% ((hn = 0.2 h_n)) if the energy increases, ensuring stable convergence [1].
Conjugate Gradient
The Conjugate Gradient algorithm exhibits slower initial convergence but becomes significantly more efficient as the system approaches the energy minimum. Unlike Steepest Descent, it utilizes information from previous steps to construct conjugate search directions, enabling more direct paths to minima. A critical limitation is that it cannot be used with constraints, meaning flexible water models must be employed when using this method [1]. This makes it particularly suitable for minimization prior to normal-mode analysis, where high accuracy is required [1].
L-BFGS Algorithm
The L-BFGS (limited-memory Broyden-Fletcher-Goldfarb-Shanno) algorithm implements a quasi-Newton approach that approximates the inverse Hessian matrix using a fixed number of corrections from previous steps [1]. This sliding-window technique provides convergence rates similar to the full BFGS method while maintaining memory requirements proportional to the number of particles multiplied by the correction steps. In practice, L-BFGS has demonstrated faster convergence than conjugate gradients, though it is not yet parallelized and benefits from switched or shifted interactions to improve convergence stability [1].
Table 1: Comparative Analysis of Energy Minimization Algorithms in GROMACS
| Algorithm | Convergence Efficiency | Memory Requirements | Constraint Compatibility | Optimal Use Case |
|---|---|---|---|---|
| Steepest Descent | Robust initial convergence, efficient for initial stages | Low | Fully compatible with all constraints | Initial minimization of poorly structured systems |
| Conjugate Gradient | Slow initial, efficient near minimum | Moderate | Not compatible with constraints (e.g., SETTLE waters) [1] | Pre-normal-mode analysis minimization [1] |
| L-BFGS | Faster convergence than CG in practice [1] | Moderate (proportional to particles × steps) | Compatible with constraints | General purpose minimization, particularly with switched interactions [1] |
mdrun Energy Minimization Workflow
The energy minimization process follows a structured workflow encompassing preparation, execution, and validation phases to ensure system stability before production dynamics.
Input Preparation and Preprocessing
The energy minimization workflow begins with comprehensive input preparation. Three essential components are required:
- Molecular Structure File: Contains atomic coordinates of the solvated and ion-neutralized system in
.groor.pdbformat [3] [11] - Topology File (
topol.top): Defcribes molecular composition, connectivity, and force field parameters, must be properly maintained throughout system assembly [3] - Parameters File (
em.mdp): Contains minimization algorithm specifications and convergence criteria [11]
These inputs are processed by the grompp (GROMACS preprocessor) module to generate a single portable binary input file (em.tpr) containing all system information:
This command validates input consistency and integrates the three components into a unified simulation input file [3] [11].
Execution with mdrun
The minimization process is executed using the mdrun module with the generated TPR file:
The -v flag enables verbose output for progress monitoring, -deffnm specifies a common prefix for all output files, and -c defines the output structure file [3] [11]. During execution, the selected minimization algorithm iteratively adjusts coordinates while monitoring energy and force convergence.
Convergence Criteria and Validation
The minimization algorithm terminates when either the maximum force falls below a specified threshold (emtol) or the maximum number of steps is reached. Successful convergence is indicated by a message similar to:
Two critical metrics validate minimization success:
- Potential Energy (
Epot): Should be negative, typically on the order of 10⁵-10⁶ for a solvated protein system [3] [5] - Maximum Force (
Fmax): Must be below the specified tolerance (e.g., 1000 kJ mol⁻¹ nm⁻¹) [3] [11]
Table 2: Energy Minimization Output Files and Their Applications in Analysis
| Output File | Format | Content | Analysis Application |
|---|---|---|---|
| em.log | ASCII text | Step-by-step log of minimization process | Convergence monitoring; Diagnostic information |
| em.edr | Binary | Energy components across steps | Energy analysis with gmx energy |
| em.trr | Binary | Full-precision trajectory | Detailed coordinate evolution analysis |
| em.gro | ASCII | Final minimized coordinates | Input for subsequent simulation steps |
Experimental Protocol for Energy Minimization
This section provides a detailed, executable protocol for performing energy minimization with mdrun in a research or drug development context.
Parameter Configuration
Create a minimization parameters file (em.mdp) with the following essential directives:
For systems requiring conjugate gradient minimization (e.g., prior to normal-mode analysis), set integrator = cg and ensure constraints = none with define = -DFLEXIBLE to employ flexible water models [1].
Execution and Monitoring
Execute the minimization process and monitor convergence in real-time:
The -nt flag specifies the number of CPU threads for parallel execution. During execution, monitor the terminal output for decreasing energy and force values. The verbose flag (-v) provides real-time feedback on progress.
Post-Minimization Analysis
Validate minimization success through comprehensive analysis:
Energy Convergence Plotting:
Select "Potential" (11) and "0" to terminate input [3]. Plot the resulting
potential.xvgto visualize energy convergence, which should show a steady decrease to a plateau.Convergence Verification: Examine the final values in
em.log:- Confirm
Fmax < emtol(e.g., < 1000 kJ mol⁻¹ nm⁻¹) - Verify potential energy is negative and reasonable for system size [3]
- Confirm
Structure Validation: Visually inspect the minimized structure (
em.pdb) in molecular viewers to confirm elimination of steric clashes while maintaining proper molecular geometry.
Table 3: Essential Research Reagent Solutions for GROMACS Energy Minimization
| Reagent/Resource | Function | Application Notes |
|---|---|---|
| GROMACS Installation | Primary simulation engine | Requires compatible C++ compiler, MPI for parallelization |
| Molecular Structure File | Atomic coordinate representation | GRO/PDB format; Should represent solvated, charge-neutralized system |
| Topology File | Molecular connectivity and parameters | Defines force field, molecular bonds, angles, dihedrals |
| Parameters File (.mdp) | Minimization algorithm settings | Specifies integrator, convergence criteria, step parameters |
| Xmgrace/Gnuplot | Visualization of energy convergence | Plotting potential energy versus steps to verify convergence |
| Molecular Viewer | Structural visualization | VMD, PyMOL, or Chimera for clash detection and structural validation |
Troubleshooting Common mdrun Challenges
Even properly configured minimizations can encounter convergence issues. This section addresses common challenges and their solutions.
Error: "Stepsize too small, or no change in energy. Converged to machine precision, but not to the requested Fmax"
Error: "Energy minimization has stopped because the force on at least one atom is not finite"
Poor Convergence (
Fmax > emtolafter maximum steps)Incompatibility with Conjugate Gradient Algorithm
For persistent issues, consider structural reevaluation of the initial model, verification of topology parameters, or utilization of double-precision GROMACS builds for enhanced numerical stability.
Energy minimization with gmx mdrun represents a fundamental preparatory step in molecular dynamics simulations, essential for eliminating structural instabilities and ensuring subsequent simulation reliability. Understanding the algorithmic foundations, implementation protocols, and troubleshooting approaches enables researchers to effectively employ this critical technique across diverse molecular systems. The structured workflow encompassing input preparation, algorithmic execution, and rigorous validation provides a robust framework for obtaining stable molecular configurations suitable for further computational investigation. As molecular simulations continue to play an expanding role in drug development and biomolecular research, mastery of these energy minimization principles remains indispensable for producing physiologically relevant and computationally stable results.
When and Why to Use Energy Minimization in Your Workflow
Energy minimization (EM) is a foundational step in molecular dynamics (MD) simulations, serving as a critical procedure to prepare a stable molecular system for subsequent dynamical studies. Within the GROMACS mdrun ecosystem, energy minimization functions as a non-dynamical algorithm that relieves structural stresses and prepares the system for the introduction of kinetic energy. When a molecular system is initially constructed—for example, by solvating a protein in water or adding counterions—the process can create steric clashes or place atoms in high-energy configurations. If molecular dynamics simulations were initiated directly from such a state, the excessively large forces could cause numerical instability and crash the simulation [3] [17].
The core objective of energy minimization is to find the nearest local minimum on the potential energy surface by iteratively adjusting atomic coordinates to reduce the total potential energy. This process results in a structure with minimal potential energy and, crucially, where the maximum force (Fmax) on any atom is below a specified tolerance. This yields a stable starting configuration that satisfies the physical assumptions of the force field [18] [3]. For researchers and drug development professionals, a properly minimized structure is not merely a technical formality but a prerequisite for obtaining physically meaningful and reproducible results from production MD simulations, which can inform critical decisions in areas like ligand binding studies and free-energy calculations [19] [20].
Theoretical Foundation: The "Why" Behind Energy Minimization
The Problem of Initial Structures and the Need for Minimization
Initial molecular models, often derived from experimental sources like X-ray crystallography or nuclear magnetic resonance (NMR), frequently contain atomic overlaps and strained bond geometries. Furthermore, the computational processes of solvation and ion addition introduce steric clashes between the solute and solvent atoms [3] [19]. From a statistical mechanics perspective, macroscopic properties are ensemble averages over a representative statistical ensemble [17]. A configuration with severe steric clashes is a high-energy outlier that is not representative of the equilibrium ensemble. Energy minimization removes these unrealistic interactions, bringing the system to a representative low-energy starting point [17].
Another critical reason for energy minimization is the removal of all kinetic energy from the system. When comparing multiple "snapshots" from different dynamic simulations, the thermal noise from atomic velocities can obscure meaningful structural differences. Energy minimization quenches this thermal motion, allowing for a clearer comparison of potential energies and geometries, which is essential for techniques like normal mode analysis [17].
Algorithmic Approaches to Energy Minimization inmdrun
GROMACS provides several algorithms for energy minimization, accessible via the integrator keyword in the parameter (.mdp) file. The choice of algorithm involves a trade-off between robustness, computational efficiency, and the required precision of the final minimized structure [18] [21].
Table 1: Energy Minimization Algorithms in GROMACS
| Algorithm | Key Principle | Advantages | Disadvantages | Typical Use Case |
|---|---|---|---|---|
| Steepest Descent | Moves atoms in the direction of the negative energy gradient (the force) [18]. | Robust, easy to implement; suitable for initial steps on very distorted structures [18]. | Slower convergence near the minimum [18]. | Initial relaxation of systems with severe steric clashes [18]. |
| Conjugate Gradient | Uses a sequence of conjugate (non-interfering) search directions [18]. | More efficient than Steepest Descent closer to the energy minimum [18]. | Cannot be used with constraints (e.g., rigid water models) [18]. | Minimization prior to normal-mode analysis requiring high precision [18] [21]. |
| L-BFGS (Limited-memory Broyden–Fletcher–Goldfarb–Shanno) | A quasi-Newton method that builds an approximation of the inverse Hessian matrix [18] [21]. | Faster convergence than Conjugate Gradients in practice [18]. | Not yet parallelized due to its correction steps [18] [21]. | Efficient minimization of medium-sized systems where serial performance is acceptable. |
The fundamental stopping criterion for these algorithms is the maximum force tolerance (emtol). The minimization is considered successful when the absolute value of the largest force component on any atom falls below this specified value, typically set between 10 and 1000 kJ mol⁻¹ nm⁻¹, depending on the required precision [18] [3].
Practical Application: The "When" and "How"
When is Energy Minimization Necessary?
Energy minimization is a critical step in nearly every MD workflow. Specific scenarios include:
- After System Construction: It is mandatory after building a simulation system, which includes steps like solvation and adding ions, to relieve the steric clashes introduced during these processes [3] [19].
- Prior to Molecular Dynamics: A minimized structure is the essential starting point for all subsequent MD simulations (e.g., equilibration and production runs) to ensure numerical stability [3].
- Before Normal Mode Analysis: Normal mode analysis requires a structure to be precisely at a local energy minimum. This typically demands high-precision minimization, often using the Conjugate Gradient algorithm in double precision [18] [21].
- Structural Comparison: To compare structures from different trajectories without the obscuring effect of thermal noise, researchers first minimize each snapshot to remove kinetic energy [17].
A Standard Protocol for Energy Minimization
The following protocol details a standard workflow for energy minimization of a solvated and neutralized protein system using GROMACS [3] [19].
Figure 1: A standard GROMACS energy minimization workflow. The three red nodes (grompp_genion, grompp_em, mdrun_em) represent the core steps of the minimization run itself.
Step 1: Assemble the System and Generate Inputs
First, generate the initial structure and topology, place it in a simulation box, and add solvent and ions. The following commands assume your parameter file for the minimization step is named minim.mdp.
Note: The genion command will prompt you to select a group of molecules (e.g., "SOL") to be replaced by ions [3] [19].
Step 2: Configure and Run the Minimization
Assemble the binary input and run the minimization using mdrun.
The -deffnm em flag defines a common prefix (em) for all output files, which include:
em.log: ASCII-text log file of the process.em.edr: Binary energy file.em.trr: Binary full-precision trajectory.em.gro: Energy-minimized structure [3].
Step 3: Validate the Results
A successful minimization is determined by evaluating two key outputs printed to the terminal and the log file.
- Potential Energy (
Eₚₒₜ): The potential energy of the system should be negative. For a solvated protein system, its magnitude is typically on the order of 10⁵ to 10⁶, scaling with system size [3]. - Maximum Force (
Fmax): This is the most critical criterion. TheFmaxvalue must be below the force tolerance (emtol) specified in yourminim.mdpfile. A common target for initial minimization isemtol = 1000.0 kJ mol⁻¹ nm⁻¹[3].
Example of a successful termination message:
To analyze the convergence, you can plot the potential energy over the minimization steps:
The resulting plot should show a monotonic decrease in potential energy, demonstrating steady convergence [3].
The Scientist's Toolkit: Essential Materials and Reagents
Table 2: Key Research Reagents and Computational Tools for Energy Minimization
| Item Name | Function/Description | Example/Format |
|---|---|---|
| Protein Structure File | The initial atomic coordinates of the molecule(s) to be simulated. | protein.pdb (from RCSB PDB) [19] |
| Molecular Topology File | Describes the molecular system: atoms, bonds, angles, dihedrals, and force field parameters. | topol.top [19] |
| Force Field | A set of empirical functions and parameters that define the potential energy of the system. | ffG53A7 (recommended in GROMACS v5.1) [19] |
| Simulation Box | A periodic boundary condition cell that contains the system to eliminate edge effects. | Cubic, Dodecahedron [19] |
| Water Model | A set of parameters defining the behavior of water molecules in the simulation. | SPC, TIP3P, TIP4P (Flexible or rigid) [18] [21] |
| Ions | Counterions (e.g., Na⁺, Cl⁻) added to neutralize the net charge of the system. | Ions are added via genion [3] [19] |
| Parameter File (.mdp) | The input file specifying all control parameters for the simulation, including the EM algorithm. | minim.mdp [3] [21] |
Advanced Considerations and Integration with Broader Workflows
Integration withmdrunFeatures and Free-Energy Calculations
Energy minimization is often the first step in a more complex pipeline. Understanding how it interacts with other mdrun features is crucial for advanced applications.
- Multi-Simulation Workflows:
mdruncan handle multi-simulations (e.g., replica-exchange or sets of lambda windows for free-energy perturbation (FEP)) using the-multidiroption. If energy minimization is required for each member of such an ensemble, it must be performed separately for each system before the multi-simulation begins [22]. - Free-Energy Perturbation (FEP): FEP calculations estimate binding affinities by alchemically transforming one ligand into another. These workflows require structurally stable starting configurations for each lambda window to ensure accurate sampling. Energy minimization is therefore critical for preparing these initial structures [20].
- Reproducible Mode: For debugging purposes,
mdrun -reprodcan be used to run a simulation (including minimization) in a mode that guarantees binary-identical results upon repeated invocations on the same hardware and software. This is useful for verifying that a minimization protocol is stable and unaffected by non-deterministic floating-point operations [22].
Parameter Selection: A Practical.mdpTemplate
The following table outlines key parameters for a typical energy minimization run, suitable for inclusion in your minim.mdp file.
Table 3: Key Parameters for Energy Minimization in the .mdp File
| Parameter | Value & Unit | Description and Rationale |
|---|---|---|
integrator |
steep / cg / l-bfgs |
Chooses the minimization algorithm. steep is robust for initial minimization of distorted structures [18] [21]. |
emtol |
1000.0 [kJ mol⁻¹ nm⁻¹] |
The stopping criterion: maximum force tolerance. A value of 1000 is a common, relatively loose target for initial minimization [3]. |
emstep |
0.01 [nm] |
(Steepest Descent) The initial maximum displacement. A smaller value is more conservative for unstable systems [18]. |
nstcgsteep |
1000 |
(Conjugate Gradient) The frequency of performing a steepest descent step during CG minimization, which can improve efficiency [21]. |
nsteps |
-1 / 50000 |
The maximum number of steps. -1 means no limit, allowing the minimizer to run until emtol is met [21]. |
define |
-DFLEXIBLE |
(If using Conjugate Gradient) This preprocessor define is required to use flexible water models, as CG cannot handle constraints like rigid water [18] [21]. |
Energy minimization is an indispensable step in the molecular dynamics workflow, serving to resolve steric clashes and produce a stable, low-energy configuration from which meaningful dynamics can be initiated. The choice of algorithm—Steepest Descent, Conjugate Gradient, or L-BFGS—involves a strategic trade-off between robustness and efficiency. Successful minimization is validated by a negative potential energy and, most importantly, a maximum force below the specified tolerance.
For researchers leveraging GROMACS mdrun, a rigorous minimization protocol ensures the integrity of subsequent simulation stages, from equilibration and production runs to advanced free-energy calculations. By providing a stable foundation, energy minimization enables the generation of reliable, reproducible data that can confidently inform scientific conclusions and drug development efforts.
Potential Energy, Forces, and Convergence Criteria
Within the framework of molecular dynamics (MD) simulations using GROMACS, energy minimization (EM) represents a fundamental preparatory step that is critical for the success of subsequent dynamical studies. This procedure is indispensable for relaxing molecular structures, removing unfavorable atomic contacts, and achieving a stable starting configuration with minimal potential energy. The mdrun command in GROMACS executes the core minimization algorithms, which operate by iteratively adjusting atomic coordinates to reduce the potential energy of the system. The efficiency and outcome of this process are governed by the intricate relationship between the potential energy landscape, the interatomic forces derived as its negative gradient, and the precise convergence criteria that terminate the minimization algorithm [1] [23]. For researchers in drug development, a robust understanding of these concepts is crucial for preparing realistic protein-ligand complexes, solvated systems, and other macromolecular assemblies, thereby ensuring the reliability of following production MD simulations used for binding affinity prediction or conformational analysis. This application note details the core concepts, algorithms, and practical protocols for effective energy minimization within the broader thesis research on the GROMACS mdrun command.
Theoretical Foundation
The Energy Landscape and the Role of Forces
In molecular modeling, the potential energy ( V ) of a system is a function of the coordinates of all ( N ) atoms, denoted as the vector ( \mathbf{r} ) [1]. The primary objective of energy minimization is to locate a local minimum on this multidimensional potential energy surface. At such a minimum, the net force on every atom vanishes. The force ( \mathbf{F}i ) on an atom ( i ) is defined as the negative gradient of the potential energy with respect to the atom's position [23]: [ \mathbf{F}i = - \frac{\partial V}{\partial \mathbf{r}_i} ] During minimization, the algorithm uses these forces to determine the direction in which to adjust the atomic coordinates to achieve a lower energy. An infinite or very large force typically indicates severe atomic overlap, which must be resolved for minimization to succeed [24].
Convergence Criteria
The minimization process is halted when the system satisfies a user-defined convergence criterion. In GROMACS, the primary criterion is that the maximum absolute value of any force component on any atom (( F_{max} )) must fall below a specified tolerance, ( \epsilon ) [1]. A reasonable value for ( \epsilon ) can be estimated from the root mean square force of a harmonic oscillator at a given temperature. For a weak oscillator with a wave number of 100 cm(^{-1}), a reasonable ( \epsilon ) lies between 1 and 10 kJ mol(^{-1}) nm(^{-1}) [1]. Proper setting of this tolerance is vital; an overly tight criterion may lead to endless iterations without meaningful energy reduction, while a too-loose criterion may result in an inadequately minimized structure.
Energy Minimization Algorithms in GROMACS
The mdrun program in GROMACS implements several minimization algorithms, specified via the integrator parameter in the molecular dynamics parameter (.mdp) file [1] [25]. The choice of algorithm involves a trade-off between robustness, computational efficiency, and system constraints.
Table 1: Key Energy Minimization Algorithms in GROMACS
| Algorithm | integrator Keyword |
Principle of Operation | Key Parameters | Typical Use Case |
|---|---|---|---|---|
| Steepest Descent [1] | steep |
Moves atoms in the direction of the instantaneous force (negative gradient). | emtol, emstep |
Robust initial minimization, especially for relieving severe steric clashes. |
| Conjugate Gradient [1] | cg |
Uses a sequence of conjugate search directions for more efficient convergence. | emtol, nstcgsteep |
Minimization prior to normal-mode analysis (requires flexible water). |
| L-BFGS [1] | l-bfgs |
A quasi-Newton method that builds an approximation of the inverse Hessian. | emtol |
Faster convergence for large systems when parallelization is not critical. |
Algorithm Workflows
The Steepest Descent algorithm exemplifies the iterative process. Starting with an initial maximum displacement ( h0 ) (e.g., 0.01 nm), new positions are calculated using [1]: [ \mathbf{r}{n+1} = \mathbf{r}n + \frac{hn}{\max(|\mathbf{F}n|)} \mathbf{F}n ] The algorithm employs a heuristic to adjust the step size: if the energy decreases (( V{n+1} < Vn )), the step size is increased by 20% for the next step; if the energy increases, the step is rejected and the step size is reduced by 80% [1]. This makes the method robust for poorly starting structures.
In contrast, the Conjugate Gradient and L-BFGS algorithms are more complex but offer superior convergence properties near the energy minimum. A critical limitation is that the Conjugate Gradient algorithm cannot be used with constraints, meaning rigid water models like SETTLE are incompatible, and flexible water must be specified instead [1]. The L-BFGS algorithm generally converges faster than Conjugate Gradients but is not yet parallelized, which may influence its selection for very large systems [1] [25].
The following diagram illustrates the logical workflow and decision process within the Steepest Descent algorithm, as implemented in GROMACS mdrun:
Practical Protocols and Setup
Configuration via the .mdp File
Configuring an energy minimization run in GROMACS primarily involves setting the correct parameters in a .mdp file. The following table outlines the essential parameters and their functions:
Table 2: Essential .mdp Parameters for Energy Minimization
| Parameter | Function | Recommended Setting |
|---|---|---|
integrator |
Selects the minimization algorithm. | = steep, cg, or l-bfgs |
emtol |
Sets the force tolerance (( \epsilon )) for convergence in kJ mol⁻¹ nm⁻¹. | = 1000.0 (initial, for rough minimization), = 10.0 (standard) |
emstep |
The initial step size (nm) for Steepest Descent. | = 0.01 |
nsteps |
The maximum number of steps allowed. | = -1 (no limit, rely on emtol) |
nstcgsteep |
Frequency of steepest descent steps in Conjugate Gradient. | = 1000 |
define |
Preprocessor defines for topology control. | = -DFLEXIBLE (if using CG with water) |
A typical .mdp file for an initial steepest descent minimization would contain:
For a more refined minimization using conjugate gradients, the following settings could be used, noting the requirement for flexible water [1]:
Execution and Analysis Workflow
The standard workflow for energy minimization involves preparing the input structure and topology, generating the binary input (tpr) file, running mdrun, and analyzing the output.
To analyze the results, the gmx energy command is used to extract energy components. The user is prompted to select the desired energy terms, such as Potential, from the energy file (em.edr). The tool calculates the average potential energy and its root mean square deviation, providing a quantitative measure of the minimization's success [26].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Computational Reagents for GROMACS Energy Minimization
| Reagent / Tool | Function | Protocol-Specific Notes |
|---|---|---|
GROMACS mdrun [1] |
The core engine that executes the energy minimization algorithm. | Configure via .mdp file parameters; use -deffnm flag for output filenames. |
| Molecular Topology | Defines the chemical structure, bonded terms, and non-bonded parameters of the system. | Must be consistent with the coordinate file; mismatches cause infinite forces [24]. |
| Steepest Descent Algorithm [1] | A robust minimizer for initial relaxation of structures with steric clashes. | Use with emtol=1000 and a small emstep (0.01) for problematic structures. |
| Conjugate Gradient Algorithm [1] | An efficient minimizer for final minimization stages near the energy minimum. | Requires -DFLEXIBLE water in the topology; incompatible with constraints. |
gmx energy Tool [26] |
Extracts and analyzes energy components from the output .edr file. |
Used to plot potential energy over steps and verify steady convergence. |
| Visualization Software (VMD) | Provides a graphical representation of the molecular structure before and after minimization. | Critical for diagnosing issues like large molecular displacements or remaining clashes [27]. |
Troubleshooting Common Issues
Despite a seemingly correct setup, minimization can fail. Two common issues and their remedies are:
Infinite Forces and Atomic Overlap: This is the most frequent error, manifesting as
Fmax= infin the log file and an extremely high potential energy [24].- Cause: The most common cause is a mismatch between the atomic coordinates in the structure file (
.gro/.pdb) and the definitions in the topology file. This can create unrealistic bonded interactions or van der Waals contacts [24]. - Solution: Carefully check the topology against the structure file, ensuring all atoms are present and in the expected order. Visualization can help identify the specific atom (reported in the log file, e.g.,
atom= 1251) that is causing the problem [24].
- Cause: The most common cause is a mismatch between the atomic coordinates in the structure file (
Unexpected Molecular Displacement: The entire molecule may shift significantly within the box after minimization.
- Cause: This can be a visual artifact due to periodic boundary conditions (PBC) when using visualization tools. The energy minimization process primarily alters internal degrees of freedom, and the overall translation of a molecule in vacuum does not change its potential energy.
- Solution: Use
gmx trjconvto rebuild the box and center the molecule before and after minimization for consistent visualization. This helps distinguish between actual movement and PBC representation issues [27].
Advanced Applications and Integration
Energy minimization is not an isolated task but is deeply integrated into broader simulation workflows. It is a prerequisite for normal-mode analysis (integrator=nm), which requires a highly minimized structure, typically achieved using the conjugate gradient algorithm [1] [25]. Furthermore, minimization is a critical first step in free energy calculations [28]. Before performing alchemical transformations using thermodynamic integration or slow-growth methods, the system must be stably minimized at both the initial (( \lambda=0 )) and final (( \lambda=1 )) states to ensure valid results. The forces with respect to the coupling parameter ( \lambda ) (( \partial H/\partial \lambda )) are sensitive to atomic overlaps, making proper minimization essential [28].
Hands-On Guide: Configuring and Running Energy Minimization with mdrun
Energy minimization (EM) is a fundamental preparatory step in molecular dynamics (MD) simulations, serving to relieve atomic clashes, reduce excessive potential energy, and create a stable starting configuration for subsequent dynamics. Within the GROMACS mdrun workflow, EM is controlled by a specific set of parameters in the molecular dynamics parameters (.mdp) file. The proper configuration of four key parameters—integrator, emtol, emstep, and nsteps—is critical for achieving a stable, low-energy system efficiently. This protocol details the function, optimal settings, and practical application of these parameters, providing a structured guide for researchers embarking on energy minimization procedures within a broader thesis on GROMACS mdrun command.
Theoretical Foundation of Minimization Algorithms
Energy minimization algorithms operate by iteratively adjusting atomic coordinates to locate a local minimum on the potential energy surface. The integrator MDP option selects the specific algorithm for this task. GROMACS offers three primary minimizers, each with distinct convergence properties and computational characteristics [21] [18] [29].
The steepest descent algorithm follows the negative gradient of the potential energy function. It is characterized by robust convergence in the early stages of minimization, even from highly distorted initial structures. The step size is adaptively controlled; it is increased by 20% following a successful step that lowers the energy and decreased by 80% following a rejected step that raises the energy [18]. This makes it particularly suitable for the initial steps of minimization, where it can rapidly relieve severe steric clashes.
The conjugate gradient method builds upon gradient information from previous steps to construct a more efficient search direction. While potentially slower than steepest descent initially, it exhibits superior quadratic convergence properties closer to the energy minimum. A notable implementation detail is that nstcgsteep determines the frequency of performing a steepest descent step during conjugate gradient minimization, which can improve efficiency [21] [29]. It is important to note that this algorithm cannot be used with constraints (e.g., rigid water models like SETTLE) and requires the use of flexible water, typically specified with define = -DFLEXIBLE in the mdp file [18] [30].
The L-BFGS (limited-memory Broyden-Fletcher-Goldfarb-Shanno) algorithm is a quasi-Newton method that approximates the inverse Hessian matrix, providing faster convergence than conjugate gradients in many practical scenarios. The accuracy of this approximation is controlled by the nbfgscorr parameter (number of correction steps), where a higher value increases accuracy at the cost of greater computational requirements and memory usage [29]. A current limitation is that the L-BFGS algorithm is not yet parallelized in GROMACS [21] [18].
Table 1: Core Energy Minimization Parameters in GROMACS
| Parameter | Function | Default Value | Common Range | Units |
|---|---|---|---|---|
integrator |
Selects minimization algorithm | N/A | steep, cg, l-bfgs |
N/A |
emtol |
Convergence tolerance (max force) | 10.0 | 10.0 - 1000.0 | kJ mol⁻¹ nm⁻¹ |
emstep |
Initial/Maximum step size | 0.01 | 0.001 - 0.02 | nm |
nsteps |
Maximum number of iterations | 0 | 500 - 50,000 | steps |
Parameter Specifications and Quantitative Guidelines
Convergence Tolerance (emtol) and Step Limit (nsteps)
The emtol parameter defines the convergence criterion for minimization, specifying the maximum force (the largest absolute value of the force gradient component on any atom) below which minimization is considered successful. The choice of emtol depends on the intended use of the minimized structure. For preparatory minimization before MD, a value of 1000 kJ mol⁻¹ nm⁻¹ is often sufficient [3] [11], while minimization preceding normal mode analysis requires a much tighter tolerance, potentially 1-10 kJ mol⁻¹ nm⁻¹ [18]. The nsteps parameter provides a crucial safeguard by setting the maximum number of minimization steps, preventing infinite loops in case of non-convergence. For typical protein-water systems, values between 1000 and 5000 steps are common starting points [3] [31].
Step Size Control (emstep)
The emstep parameter has algorithm-dependent behavior. For steepest descent, it defines the maximum displacement allowed in any iteration [18] [29]. For conjugate gradient and L-BFGS, it typically serves as the initial step size [21]. An appropriate value balances convergence speed against stability; too large a value may cause instability or energy increases, while too small a value unnecessarily slows convergence. A common initial value is 0.01 nm [3] [31], which can be adjusted based on system response.
Table 2: Algorithm Selection Guide for Energy Minimization
| Algorithm | Typical emtol |
Typical emstep (nm) |
Strengths | Limitations |
|---|---|---|---|---|
| Steepest Descent | 100 - 1000 | 0.01 - 0.02 | Robust, efficient initial convergence | Slow near minimum |
| Conjugate Gradient | 10 - 100 | 0.01 | Better convergence near minimum | Requires flexible water |
| L-BFGS | 1 - 100 | 0.01 | Fastest convergence | Not parallelized |
Experimental Protocols and Methodologies
Standard Energy Minimization Workflow
The following protocol outlines the standard procedure for configuring and executing energy minimization in GROMACS, from parameter selection through result validation.
Sample MDP Configuration File
A typical energy minimization parameter file (em.mdp) incorporates the critical parameters discussed, along with essential non-bonded interaction settings.
Execution and Monitoring Commands
After configuring the em.mdp file, the simulation input (em.tpr) is generated and executed using the following commands [3]:
The -v flag enables verbose output, providing real-time progress updates, while -deffnm em specifies a default filename for all output files. During execution, monitor the terminal output or em.log file for the current maximum force (Fmax) and potential energy values. Successful convergence is indicated by a message similar to: "Steepest Descents converged to Fmax < 1000 in 566 steps" [3].
Validation and Analysis of Results
Assessing Minimization Success
Following minimization, two critical metrics must be evaluated to determine success [3]:
- Potential Energy (Epot): Should be negative and, for a solvated protein system, typically on the order of 10⁵-10⁶ kJ/mol. A positive or insufficiently negative value suggests significant unresolved steric clashes.
- Maximum Force (Fmax): Must be less than the specified
emtolvalue. The minimization output explicitly states whether this condition was met. It is possible to reach a reasonable Epot with Fmax > emtol, indicating the system may not be stable enough for subsequent simulation.
Energy Time Series Analysis
The GROMACS energy module extracts energy terms from the binary energy file (em.edr) for analysis and visualization:
When prompted, select "Potential" by entering the corresponding number (typically 11), followed by "0" to terminate input. The resulting potential.xvg file charts the potential energy throughout minimization, which should show a monotonic decrease to a stable plateau, indicating successful convergence [3].
Troubleshooting and Optimization Strategies
Algorithm Selection Logic
Common Issues and Solutions
- Failure to Converge Within
nsteps: Increasenstepsor use a two-stage approach: initial minimization withintegrator=steepand looseemtol(e.g., 1000), followed byintegrator=cgorl-bfgswith tighteremtol(e.g., 100) [11]. - Energy Minimization Stops with Large Forces: The algorithm may stop when step sizes become too small, indicating convergence to the available machine precision. This may be acceptable for subsequent MD if the potential energy is reasonable [31].
- Unstable Minimization (Energy Increasing): Reduce
emstepby a factor of 2-5 to improve stability, particularly for steepest descent. - High Final Potential Energy: Check for unresolved steric clashes, inappropriate box size, or incorrect topology. Visualization tools can help identify atomic overlaps.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Computational Tools for Energy Minimization
| Tool/Component | Function | Example/Format |
|---|---|---|
GROMACS grompp |
Preprocessor: combines structure, topology, and parameters into a binary input file | gmx grompp -f em.mdp -c system.gro -p topol.top -o em.tpr |
GROMACS mdrun |
MD engine: executes energy minimization algorithm | gmx mdrun -v -deffnm em |
GROMACS energy |
Analysis: extracts energy terms for plotting and validation | gmx energy -f em.edr -o potential.xvg |
| MDP File | Parameter input: defines minimization algorithm and convergence criteria | Text file with integrator, emtol, emstep, nsteps |
| TPR File | Portable binary input: contains complete system and run parameters | em.tpr (binary) |
| Topology File | Molecular definition: specifies atoms, bonds, and force field parameters | topol.top (text) |
| Structure File | Atomic coordinates: initial configuration for minimization | system.gro or .pdb (text) |
| Energy File | Output: contains time series of energies and forces | em.edr (binary) |
| Log File | Output: records minimization progress and convergence message | em.log (text) |
In molecular dynamics (MD) simulations using GROMACS, energy minimization (EM) is a critical preparatory step that removes steric clashes and inappropriate geometry that may have been introduced during system construction, such as solvation or ion addition [11] [3]. The gmx mdrun module is the primary computational engine that performs this minimization, utilizing the system topology, coordinates, and parameters defined in an input file to find a low-energy configuration before beginning production dynamics [32]. This process is essential for achieving stable simulation conditions and preventing simulation crashes due to excessively high forces [3]. Successfully minimizing a system results in a structure with a negative potential energy and sufficiently low maximum force, creating a suitable starting point for subsequent equilibration and production phases [3].
This application note provides researchers and drug development professionals with a comprehensive guide to constructing and executing effective energy minimization commands using gmx mdrun, including algorithm selection, syntax, flag optimization, and results validation.
Theoretical Framework of Minimization Algorithms
The gmx mdrun program implements three primary algorithms for energy minimization, each with distinct mathematical approaches and performance characteristics [1].
Steepest Descent
The Steepest Descent algorithm follows the negative gradient of the potential energy to locate the nearest local minimum. It updates positions according to: [ \mathbf{r}{n+1} = \mathbf{r}n + \frac{hn}{\max(|\mathbf{F}n|)} \mathbf{F}n ] where (\mathbf{r}) represents atomic coordinates, (hn) is the maximum displacement, and (\mathbf{F}n) is the force vector [1]. The algorithm employs a heuristic adjustment of the step size: if the potential energy decreases ((V{n+1} < Vn)), the step size increases by 20% ((h{n+1} = 1.2 hn)); if energy increases, positions are rejected and the step size is reduced by 80% ((hn = 0.2 h_n)) [1]. Although not the most efficient search method, Steepest Descent is robust and easy to implement, making it particularly effective in the initial stages of minimization where large forces may be present [1] [11].
Conjugate Gradient
The Conjugate Gradient method generally converges more efficiently than Steepest Descent, particularly closer to the energy minimum [1]. However, it cannot be used with constraints, including the SETTLE algorithm for water [1]. This limitation necessitates using flexible water models when employing Conjugate Gradient minimization, specified in the mdp file with define = -DFLEXIBLE [1]. The primary application for Conjugate Gradient is minimization prior to normal-mode analysis, where the increased accuracy is required [1].
L-BFGS Algorithm
The L-BFGS (limited-memory Broyden-Fletcher-Goldfarb-Shanno) algorithm is a quasi-Newtonian method that approximates the inverse Hessian matrix using a fixed number of corrections from previous steps [1]. This sliding-window technique provides efficiency nearly equivalent to the full BFGS method while requiring significantly less memory - proportional to the number of particles multiplied by the correction steps rather than the square of the number of particles [1]. In practice, L-BFGS has been found to converge faster than Conjugate Gradients, though it is not yet parallelized [1]. For optimal performance with L-BFGS, using switched or shifted interactions is recommended rather than sharp cut-offs, as the latter can impair convergence due to slight differences in the potential function at current versus previous coordinates [1].
Table 1: Comparison of Energy Minimization Algorithms in GROMACS
| Algorithm | Mathematical Approach | Strengths | Limitations | Typical Applications |
|---|---|---|---|---|
| Steepest Descent | First-order gradient following | Robust, handles large forces, easy implementation | Slow convergence near minimum | Initial minimization, systems with steric clashes |
| Conjugate Gradient | Sequential conjugate direction optimization | Faster convergence near minimum | Cannot be used with constraints (e.g., SETTLE) | Pre-normal-mode analysis, unconstrained systems |
| L-BFGS | Quasi-Newtonian with approximate inverse Hessian | Fast convergence, memory efficient | Not yet parallelized, sensitive to interaction treatment | Final minimization stages, large systems |
Experimental Protocol for Energy Minimization
Input Preparation Workflow
Before invoking gmx mdrun for energy minimization, researchers must properly prepare all required input files. The following workflow outlines the complete preparation and execution process:
The gmx grompp (GROMACS preprocessor) command assembles the structure, topology, and simulation parameters into a single portable binary input file with the .tpr extension [11] [3]. The syntax is:
Where -f specifies the parameter file, -c provides the coordinate file (typically the solvated and ion-neutralized system), -p defines the topology file, and -o names the output TPR file [3]. It is crucial to maintain consistent topology and coordinate files throughout this process; the topology must accurately reflect all components present in the coordinate file, including solvent molecules and ions added during system preparation [3].
mdrun Command Execution
The core minimization is executed using gmx mdrun with the following fundamental syntax:
This command performs energy minimization with verbose output (-v), reads input from em.tpr (-s), uses a default filename root of em for all outputs (-deffnm), and writes the final minimized structure to em.pdb (-c) [11] [3]. The -deffnm flag is particularly useful as it automatically applies a consistent naming scheme to all output files, including the log file (.log), energy file (.edr), and full-precision trajectory (.trr) [3].
Table 2: Essential mdrun Flags for Energy Minimization
| Flag | Argument Type | Function | Example Usage |
|---|---|---|---|
-s |
File name | Input TPR run file [32] | -s em.tpr |
-deffnm |
String | Root name for all output files [3] | -deffnm em |
-v |
Boolean | Verbose output to terminal [3] | -v |
-c |
File name | Output structure after minimization [32] [3] | -c em.pdb |
-cpi |
File name | Input checkpoint file to continue run [32] | -cpi state.cpt |
-nsteps |
Integer | Override maximum steps in MDP file [22] | -nsteps 500 |
-maxh |
Real | Terminate before specified hours elapse [32] [22] | -maxh 2.5 |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Computational Reagents for Energy Minimization
| Reagent/Resource | Function | Implementation Example |
|---|---|---|
| Force Field Parameters | Defines atomic interactions, bonded and non-bonded terms | AMBER99SB-ILDN, CHARMM36, OPLS-AA [33] |
| Solvent Models | Provides aqueous environment for solvated systems | SPC, TIP3P, TIP4P water models [1] |
| Ion Parameters | Neutralizes system charge and mimics physiological conditions | Sodium/chloride ions with matching force field parameters [3] |
| Topology Database (RTP) | Defines building blocks for molecular components | Residue topology database for standard amino acids [33] |
| Hydrogen Database (HDB) | Provides templates for adding hydrogen atoms | Standard protonation states for histidine residues [33] |
| Position Restraints | Constrains specific atoms during minimization | posre.itp files for protein backbone atoms [33] |
Results Validation and Troubleshooting
Evaluating Minimization Success
After completing energy minimization, researchers must validate the results before proceeding to subsequent simulation stages. Two critical metrics determine minimization success:
Potential Energy ((E{pot})): Should be negative and, for a solvated protein system, typically on the order of (10^5)-(10^6) depending on system size [3]. A positive or insufficiently negative (E{pot}) suggests significant structural issues requiring investigation.
Maximum Force ((F_{max})): Should fall below the specified tolerance threshold (e.g., 1000 kJ mol(^{-1}) nm(^{-1})) [3]. The minimization algorithm converges when the maximum absolute value of the force components drops below this specified value (\epsilon) [1].
The gmx energy tool extracts energy information from the binary energy file (.edr) for quantitative analysis:
When prompted, selecting term "11" (Potential) generates an XVG file plotting the energy convergence, which should demonstrate a steady decrease to a stable plateau [3].
Advanced mdrun Features for Specialized Scenarios
For challenging minimization scenarios, gmx mdrun provides specialized features:
Rerun Functionality: The
-rerunflag calculates energies and forces for an existing trajectory using the physics model in a TPR file, useful for single-point energy evaluation or computing quantities based on trajectory subsets [22]. Example:mdrun -s topol.tpr -rerun traj.xtc[22].Reproducible Mode: The
-reprodoption eliminates all sources of variation within a run, ensuring binary identical results across repeated invocations on the same hardware and software configuration [22]. This is valuable when investigating potential problems but typically sacrifices some performance optimizations.Checkpointing and Restart:
gmx mdrunwrites checkpoint files (.cpt) at regular intervals, containing the complete simulation state [32]. To continue an interrupted minimization:mdrun -cpi state.cpt -noappend[32]. The-noappendflag creates new output files with part numbers rather than appending to existing files [32].Force Monitoring: The
-pforceflag is valuable for debugging systems that crash due to excessively high forces [32]. When specified,mdrunprints coordinates and forces of atoms exceeding the threshold to stderr and terminates if non-finite forces are detected [32].
Troubleshooting Common Minimization Issues
Even with proper setup, minimization may encounter problems requiring investigator intervention:
Failure to Converge: If the maximum force remains above the tolerance threshold despite extended minimization, consider switching algorithms (e.g., from Steepest Descent to L-BFGS for final convergence) or examining the structure for steric clashes or incorrect bonding [11] [3].
Excessively High Potential Energy: A positive or insufficiently negative (E_{pot}) often indicates missing topology parameters, incorrectly assigned atom types, or severe atomic overlaps [3]. Verify topology consistency with the coordinate file and ensure all molecular components have proper force field parameters [33].
LINCS Warnings: Excessive LINCS warnings during minimization suggest bond length issues that may require careful investigation of the starting structure and topology [33]. The
GMX_MAXCONSTRWARNenvironment variable can be set to-1to prevent exit due to too many LINCS warnings, though this does not address the underlying cause [34].
Proper execution of energy minimization using gmx mdrun is a foundational step in molecular dynamics simulations that directly impacts subsequent simulation stability and reliability. By understanding the theoretical basis of minimization algorithms, following systematic preparation protocols, utilizing appropriate command-line flags, and rigorously validating results, researchers can ensure their molecular systems are properly prepared for production dynamics. This application note provides the comprehensive framework needed for researchers and drug development professionals to effectively implement energy minimization protocols within their broader computational research objectives, establishing a critical foundation for successful molecular simulations in drug discovery and structural biology.
Within the broader context of GROMACS mdrun command research for energy minimization, this protocol provides a standardized workflow for preparing molecular systems for production molecular dynamics simulations. Energy minimization (EM) is a critical preliminary step designed to resolve steric clashes and inappropriate geometry introduced during system assembly, ensuring numerical stability during subsequent dynamics [3]. Without proper minimization, systems may exhibit unrealistic forces leading to simulation instability or complete failure [35]. This application note details a comprehensive methodology from initial structure files to a minimized system ready for equilibration, specifically tailored for researchers, scientists, and drug development professionals working with biomolecular systems.
The theoretical foundation of energy minimization relies on algorithms that iteratively adjust atomic coordinates to locate local minima on the potential energy surface [1]. GROMACS provides multiple minimization algorithms, including steepest descent, conjugate gradient, and L-BFGS, each with distinct convergence properties and computational characteristics [1]. This protocol emphasizes practical implementation while maintaining scientific rigor, enabling researchers to generate structurally sound systems for investigating protein-ligand interactions, membrane protein dynamics, and other pharmaceutically relevant phenomena.
Table 1: Essential research reagents and computational tools for GROMACS energy minimization
| Tool Name | Type/Format | Primary Function in Workflow |
|---|---|---|
| Molecular structure file (.pdb, .gro) | Coordinate file | Provides initial atomic coordinates for the system [3] |
| Molecular topology file (.top) | Topology file | Defines molecular connectivity, atom types, and force field parameters [35] |
| Simulation parameters file (.mdp) | Input parameter file | Configures minimization algorithm, convergence criteria, and output options [11] |
GROMACS .tpr file |
Portable binary input | Integrated system representation for mdrun [32] |
| VMD, PyMOL, Chimera | Visualization software | Enables structural validation and visual inspection of minimized systems [36] |
| DuIvyTools, Grace, matplotlib | Analysis & plotting tools | Processes output files and visualizes energy convergence [36] [37] |
Algorithm Selection and Quantitative Comparison
Table 2: Quantitative comparison of energy minimization algorithms in GROMACS
| Algorithm | Convergence Speed | Memory Requirements | Implementation Robustness | Optimal Use Cases |
|---|---|---|---|---|
| Steepest Descent | Fast initial convergence | Low | High [1] | Initial minimization of poorly structured systems [1] |
| Conjugate Gradient | Slower initially, efficient near minimum | Moderate | Medium (cannot be used with constraints) [1] | Pre-normal mode analysis; systems with flexible water [1] |
| L-BFGS | Fastest near minimum | Low (compared to full BFGS) | High [1] | Well-structured systems; production minimization [1] |
The steepest descent algorithm updates atomic positions according to $\mathbf{r}{n+1} = \mathbf{r}n + \frac{hn}{\max(|\mathbf{F}n|)} \mathbf{F}n$, where $hn$ is the maximum displacement and $\mathbf{F}n$ is the force vector [1]. This method employs an adaptive step size control where $h{n+1} = 1.2 hn$ when energy decreases and $hn = 0.2 h_n$ when energy increases [1]. For conjugate gradients and L-BFGS, the same convergence criteria apply, though the coordinate update strategies differ significantly in their utilization of historical gradient information [1].
The appropriate stopping criterion can be estimated from the root mean square force of a harmonic oscillator at a given temperature: $f = 2 \pi \nu \sqrt{2mkT}$ [1]. For a weak oscillator with a wave number of 100 cm⁻¹, mass of 10 atomic units, at 1 K, this yields approximately 7.7 kJ mol⁻¹ nm⁻¹, suggesting an acceptable ε value between 1 and 1000 kJ mol⁻¹ nm⁻¹ depending on the system and desired accuracy [1] [3].
Step-by-Step Experimental Protocol
System Preparation and File Generation
Initial Structure Preparation: Obtain or generate initial coordinate files for each molecule in the system. For proteins, this often begins with a PDB file from experimental structure determination or homology modeling [35].
Topology Generation: Create topology files describing the molecular connectivity and force field parameters using tools such as
gmx pdb2gmx, CHARMM-GUI, or manual editing [35]. The topology must remain synchronized with the coordinate file throughout system assembly.System Assembly: Define a simulation box using
gmx editconf, solvate the system withgmx solvate, and add counter-ions usinggmx genionto achieve electroneutrality [35]. At each step, update the topology file to reflect changes in the coordinate file [35].
Energy Minimization Parameter Configuration
Algorithm Selection: Choose an appropriate minimizer based on system characteristics and research goals (refer to Table 2). For initial minimization of systems with potential steric clashes, steepest descent is recommended [1] [11].
Parameter Specification: Create an
.mdpfile configuring the minimization parameters. Key settings include:
Binary Input File Generation and Execution
Input Assembly: Use
gmx gromppto integrate structure, topology, and parameters into a portable binary (.tpr) file:Ensure topology and coordinate files remain synchronized to avoid "number of coordinates in coordinate file does not match topology" errors [3].
Minimization Execution: Execute energy minimization using
gmx mdrun:The
-vflag enables verbose output, while-deffnmspecifies a common prefix for all output files [3].
Output Validation and Analysis
Success Verification: Examine the final output for two critical factors:
Energy Convergence Analysis: Use
gmx energyto extract potential energy data and visualize convergence:Select "Potential" when prompted [3].
Structural Validation: Visually inspect the minimized structure using visualization software such as VMD or PyMOL to ensure reasonable geometry [36].
Figure 1: Complete workflow for GROMACS energy minimization from initial structure to minimized system.
Advanced Methodologies and Specialized Applications
Micelle and Membrane Protein Systems
For complex systems such as micelles or membrane proteins, additional preprocessing steps are necessary to prevent artifacts from periodic boundary conditions. Implement the following clustering protocol prior to property calculation:
gmx trjconv -f initial.xtc -o clustered.gro -e 0.001 -pbc clustergmx grompp -f em.mdp -c clustered.gro -p topol.top -o clustered.tprgmx trjconv -f initial.xtc -o clustered.xtc -s clustered.tpr -pbc nojump
This ensures the micelle remains intact across periodic boundaries, preventing erroneous calculations of properties like radius of gyration [36].
Multi-Stage Minimization Protocols
For challenging systems with significant steric clashes, implement a multi-stage minimization approach:
- Initial steepest descent minimization with conservative step size (e.g.,
emstep = 0.001) and relaxed convergence criteria - Secondary conjugate gradient or L-BFGS minimization with tighter convergence criteria (
emtol = 10-100) to refine the structure [11]
The minimized structure from the first stage serves as input for the second stage, potentially leveraging different constraint schemes or algorithm-specific parameters [11].
Troubleshooting and Quality Control
Common Failure Modes and Solutions
Excessively high potential energy: Often indicates persistent steric clashes. Consider increasing the number of minimization steps or implementing multi-stage minimization with different algorithms [11].
Maximum force above target despite reasonable Epot: Evaluate whether the
emtolcriterion is appropriately set for your system. For large or complex systems, a slightly higher Fmax may be acceptable for initial equilibration [3].Simulation crashes during minimization: Verify that constraints are disabled when using conjugate gradient methods [1]. Consider using flexible water models (
define = -DFLEXIBLE) during minimization [1].
Validation Metrics and Acceptance Criteria
A successfully minimized system should exhibit:
- Negative potential energy with magnitude appropriate for system size [3]
- Stable energy convergence throughout the minimization procedure [3]
- Maximum force below the target threshold (e.g., < 1000 kJ mol⁻¹ nm⁻¹) [3]
- Reasonable molecular geometry without distorted bond lengths or angles upon visual inspection [36]
This application note has detailed a comprehensive workflow for energy minimization in GROMACS, from initial structure preparation to a minimized system ready for equilibration. Proper implementation of this protocol ensures numerically stable simulations and reliable scientific outcomes. The integration of robust algorithm selection, appropriate convergence criteria, and systematic validation establishes a foundation for successful molecular dynamics investigations in basic research and drug development contexts.
Within the computational biochemistry workflow, energy minimization (EM) serves as a critical preliminary step to remove steric clashes and inappropriate geometry before molecular dynamics (MD) production runs. The mdrun engine in GROMACS provides several algorithms for this task, including steepest descent, conjugate gradients, and the L-BFGS quasi-Newtonian minimizer [1]. The efficient execution of EM requires careful consideration of hardware utilization, particularly regarding the division of labor between central processing units (CPUs) and graphics processing units (GPUs). This application note examines the performance characteristics and limitations of CPU versus GPU resources during energy minimization within the broader context of GROMACS mdrun command research, providing structured data and protocols for researchers and drug development professionals.
Energy minimization algorithms in GROMACS exhibit different computational characteristics that respond differently to hardware acceleration. The steepest descent algorithm provides robust convergence characteristics, calculating new positions based on the force and a maximum displacement parameter [1]. While not the most efficient algorithm for final convergence, its simplicity and stability make it suitable for initial minimization steps. The conjugate gradient method offers improved efficiency closer to the energy minimum but cannot be used with constraints, requiring flexible water models when applied to solvated systems [1]. The L-BFGS algorithm implements a limited-memory quasi-Newtonian approach that typically converges faster than conjugate gradients but currently lacks parallelization in GROMACS, making it primarily suitable for CPU-bound computations [1].
The hardware implications of these algorithmic differences are significant. GPU acceleration provides substantial performance benefits for parallelizable computational workloads, particularly for non-bonded interactions which dominate MD simulations. Recent GROMACS versions have made considerable strides in GPU offloading, with performance improvements of 5-20% for CUDA kernels and even greater gains (>50%) for OpenCL on AMD devices with specific force field combinations [38]. However, certain algorithmic constraints limit these benefits for specific minimization scenarios, particularly when bonded interactions or specialized kernels are computationally dominant.
Table 1: Energy Minimization Algorithms in GROMACS and Hardware Compatibility
| Algorithm | Computational Characteristics | GPU Compatibility | Optimal Use Case |
|---|---|---|---|
| Steepest Descent | Robust, stable convergence; Uses force and displacement parameters [1] | Full | Initial minimization steps; Systems with steric clashes |
| Conjugate Gradient | More efficient near minimum; Cannot use constraints [1] | Full (requires flexible water) | Intermediate to final minimization; Systems without constraints |
| L-BFGS | Fast convergence; Quasi-Newtonian method [1] | Limited (not parallelized) [1] | Final minimization stages; CPU-bound systems |
CPU vs GPU Performance Characteristics
Quantitative Performance Metrics
The performance differential between CPU and GPU implementations varies significantly based on the specific computational kernels involved. GROMACS performance optimizations have particularly benefited GPU implementations, with CUDA performance improvements of 5-20% depending on parameters and compilers [38]. OpenCL performance on AMD devices has demonstrated even more substantial gains, exceeding 50% improvement for specific force field and PME combinations [38]. These enhancements make GPU acceleration particularly advantageous for large systems where parallelization benefits can be fully realized.
CPU performance optimizations have primarily focused on Single Instruction Multiple Data (SIMD) vectorization, which provides dramatic improvements for specific computational tasks. The SIMD implementation of SETTLE, the constraint algorithm for water molecules, demonstrates a 5x speedup on Haswell CPUs [38]. Similarly, SIMD implementations of Lennard-Jones 1-4 interactions and periodic boundary condition transformations provide significant performance gains [38]. These optimizations are particularly valuable for smaller systems or when GPU resources are occupied with other computational tasks.
Table 2: Performance Comparison of Computational Kernels on CPU vs GPU
| Computational Kernel | CPU Performance Features | GPU Performance Features | Performance Implications |
|---|---|---|---|
| Non-bonded Interactions | Moderate performance; Benefits from SIMD [38] | High performance; 5-20% recent CUDA improvements [38] | GPU significantly faster for large systems |
| SETTLE (Water Constraints) | 5x faster with SIMD on Haswell [38] | Limited benefit from offloading | CPU often superior for constrained water |
| Bonded Interactions | Multi-threading improvements; Factor of 3 speedup with domain decomposition [38] | Can be offloaded (-bonded gpu) but CPU handles some [39] |
Mixed workload; CPU reduction optimized |
| PME Long-range | Traditional CPU implementation | Single GPU acceleration now available [40] | GPU significantly reduces CPU core requirements |
| LJ Combination Rules | Existing optimization | 10-15% improvement on GPU with certain force fields [38] | GPU superior for OPLS, GROMOS, AMBER |
Hardware Selection Guidelines
The choice between CPU-centric and GPU-centric minimization strategies depends on multiple factors including system size, available hardware, and minimization algorithm. Recent GROMACS versions have demonstrated that systems with powerful GPUs achieve optimal performance when offloading the majority of computational work to the GPU, even when this results in CPU underutilization [39]. This is particularly true for simulations achieving approximately 0.2 ms per step, where CPU struggle with efficiently launching GPU work units [39].
For CPU-bound minimization, particularly with the L-BFGS algorithm, thread allocation strategies become critical. When using GPUs, setting -ntmpi 1 (one MPI rank per GPU) typically provides optimal performance, with -ntomp set to the number of CPU cores [39]. In GROMACS 2024, reducing -ntomp may incur minimal performance penalty since much CPU work utilizes only one thread, though this has improved in GROMACS 2025 where OpenMP plays a larger role [39].
Experimental Protocols and Methodologies
GPU-Accelerated Energy Minimization Protocol
For researchers with access to GPU hardware, the following protocol optimizes energy minimization performance:
System Preparation: Ensure solvated and electroneutral system assembly is complete with proper topology file configuration [3].
Parameter File Configuration: Create an EM-specific parameter file (minim.mdp) with appropriate settings:
Binary File Assembly: Execute grompp to assemble structure, topology, and parameters:
GPU-Accelerated Execution: Run minimization with optimized GPU flags:
Note: The
-nstlist 300flag increases neighbor search interval, reducing CPU-side interruptions and improving GPU utilization [39].Result Validation: Verify successful minimization by checking:
CPU-Centric Energy Minimization Protocol
For systems where GPU acceleration is unavailable or suboptimal (e.g., using L-BFGS algorithm):
System Preparation: Confirm proper system assembly with correct topology references.
Algorithm Selection: Configure parameter file for L-BFGS minimization:
Binary File Preparation: Assemble input files using grompp:
CPU-Optimized Execution: Run minimization with thread configuration matched to available cores:
Adjust MPI and OpenMP thread counts based on available CPU resources.
Convergence Analysis: Monitor both potential energy and maximum force, ensuring stable convergence. For L-BFGS, note that switched or shifted interactions typically improve convergence due to smoother potential transitions [1].
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for GROMACS Energy Minimization
| Tool/Component | Function | Implementation Notes |
|---|---|---|
| GROMACS 2024+ | MD simulation engine | Provides GPU-accelerated minimization; Requires CUDA 7.5+ or OpenCL for optimal performance [38] |
| Steepest Descent Algorithm | Initial minimization workhorse | Robust for strained systems; Compatible with all constraints [1] |
| L-BFGS Minimizer | Quasi-Newtonian minimization | Faster convergence for final stages; Currently CPU-bound [1] |
| GPU Compute Capability 6.0+ | Hardware acceleration | Pascal architecture or newer; Enables wider 128 threads/block kernels [38] |
| SIMD Optimizations | CPU vectorization | Critical for CPU performance; 5x improvement for SETTLE on Haswell [38] |
| nstlist Tuning | Neighbor search frequency | Increasing to 300 reduces CPU interruptions, improving GPU utilization [39] |
Performance Optimization Strategies
Hardware-Specific Tuning
Optimal performance for energy minimization requires different strategies based on hardware configuration. For GPU-accelerated runs, ensure that the -bonded gpu flag is active to offload bonded interactions, though note that the CPU still handles some bonded forces even with this setting [39]. When using CUDA, setting GMX_CUDA_GRAPH=1 as an environment variable can reduce GPU kernel launch overhead, particularly beneficial for simulations approaching 0.2 ms per step where CPU struggle to maintain launch rates [39].
For CPU-centric minimization, leverage recent SIMD optimizations including the vectorized implementations of PME spread/gather (11% speedup on KNL processors), simple update kernels, and Urey-Bradley angle kernels [40]. On AMD Zen architectures, note that tabulated Ewald kernels consistently outperform analytical approaches, and AVX2128 SIMD often provides better performance than wider AVX2256 instructions due to microarchitectural implementation [40].
Memory and Parallelization Configuration
GPU memory configuration significantly impacts minimization performance. Recent GROMACS versions implement improved memory settings that leverage increased L1 cache availability on specific hardware (K40, K80, Tegra K1, & CC 5.2) through commands like "-dlcm=ca" for global load caching [38]. Additionally, GPU pair-list splitting has been optimized to generate lists closer to the target size, reducing heuristic overestimation from 10% to 0-1%, resulting in fewer pair lists and performance gains [38].
Threading configuration requires careful balancing. For Verlet-scheme pair-lists, GROMACS implements near-perfect workload balancing for CPU computations, increasing search cost by 3% but improving non-bonded kernel balance, particularly for small systems [38]. Virtual-site code threading has been enhanced to reduce serial bottlenecks, and OpenMP parallelization has been extended to pull-code loops over atoms, addressing previous scaling limitations that could consume up to one-third of compute time for simulations with large pull groups [38].
Energy minimization in GROMACS presents distinct hardware utilization patterns that researchers must consider for optimal performance. GPU acceleration provides substantial benefits for parallelizable workloads, particularly non-bonded interactions, with recent performance improvements making GPUs increasingly dominant for large-system minimization. However, CPU resources remain essential for specific algorithms like L-BFGS and certain computational kernels that benefit from SIMD vectorization. The optimal hardware configuration depends on system size, minimization algorithm, and available resources, with the protocols and guidelines presented herein providing researchers with structured approaches to maximize efficiency. As GROMACS development continues to enhance both CPU and GPU performance, particularly in the forthcoming 2025 version with expanded OpenMP utilization, these hardware considerations will remain dynamic, requiring continued evaluation of the tradeoffs between computational resources.
Energy minimization (EM) is a foundational step in molecular dynamics (MD) simulations, serving to relieve unfavorable atomic clashes and strained geometries in initial structures prior to dynamics. This process is essential for obtaining stable and physically meaningful simulation trajectories. Within the GROMACS mdrun engine, several minimization algorithms are available, each with distinct characteristics and optimal use cases. The choice of algorithm is governed by the integrator parameter in the molecular dynamics parameter (.mdp) file [25]. The primary goal of energy minimization is to locate a local minimum on the potential energy surface, a state characterized by forces acting on all atoms falling below a specified tolerance [1]. For researchers in drug development, a properly minimized starting structure is critical for the reliability of subsequent simulations, such as ligand binding studies or free-energy calculations.
Energy Minimization Algorithms and Parameters
Available Minimization Integrators
GROMACS provides three principal algorithms for energy minimization, each with specific performance and robustness trade-offs [1] [25].
Steepest Descent (
integrator = steep): This robust algorithm is ideal for initial minimization steps, particularly for relieving severe steric conflicts in poorly conditioned starting structures. It uses a simple approach where new positions are calculated along the negative gradient of the potential energy. The step size is dynamically adjusted: increased by 20% after successful steps and reduced by 80% after rejected steps [1]. Although not the most efficient algorithm for final convergence, its stability makes it well-suited for the initial stages of minimization.Conjugate Gradient (
integrator = cg): More efficient than steepest descent in the later stages of minimization, the conjugate gradient algorithm is recommended for achieving tight convergence when high accuracy is required. A notable limitation is its incompatibility with constraints, including the SETTLE algorithm for rigid water models. When using conjugate gradient, flexible water models must be specified in the mdp file usingdefine = -DFLEXIBLE[1] [25]. This makes it particularly valuable for minimization prior to normal-mode analysis, which demands very high precision [1].L-BFGS (
integrator = l-bfgs): The limited-memory Broyden–Fletcher–Goldfarb–Shanno quasi-Newtonian minimizer generally converges faster than conjugate gradients. However, due to its internal correction steps, it has not yet been parallelized [1] [25]. In practice, L-BFGS often represents the most efficient choice for systems where ultimate precision is not required, provided that parallelization is not essential for the minimization task.
Critical Energy Minimization Parameters
The control parameters for energy minimization are specified in the .mdp file. Key parameters include [25]:
emtol: The tolerance for convergence, defined as the maximum absolute value of any force component (in kJ·mol⁻¹·nm⁻¹). Minimization terminates when the forces fall below this threshold. A reasonable value typically ranges between 10 and 100, with 10.0 being a common default.emstep: The initial step size (in nm) for steepest descent minimization.nsteps: The maximum number of minimization steps allowed. Setting this to-1allows minimization to continue until convergence based onemtol.
Table 1: Key .mdp Parameters for Energy Minimization
| Parameter | Default Value | Description | Recommended Setting |
|---|---|---|---|
integrator |
Minimization algorithm | steep, cg, or l-bfgs |
|
emtol |
10.0 | Force tolerance (kJ·mol⁻¹·nm⁻¹) | 10.0 for preliminary; 1.0 for precise |
emstep |
0.01 | Initial step size (nm) | 0.01 |
nsteps |
0 | Maximum minimization steps | -1 (no limit) or a large number (e.g., 50000) |
Interpreting Minimization Output and Log Files
Analyzing the Log File Output
During execution, gmx mdrun generates detailed output to the log file (specified with -g). For energy minimization, this output provides critical insights into the progression and convergence of the algorithm. Key sections to monitor include [2]:
- Initial System Information: The log begins with a summary of the system composition, including the number of atoms, particles, and the simulation box dimensions.
- Algorithm Configuration: Details of the selected minimizer and its parameters are echoed.
- Minimization Steps Table: The core of the log file is an iterative table reporting the step number, the current potential energy (Epot), the maximum force (Fmax), and the step size (step).
A typical minimization step entry appears as:
The most critical quantity to monitor is Fmax, the maximum force acting on any atom in the system. Successful convergence is achieved when Fmax drops below the specified emtol value [1].
Convergence Criteria and Troubleshooting
The minimization process terminates successfully when the maximum force, Fmax, is less than the emtol threshold [1]. Understanding the behavior of the Epot and Fmax values throughout the process is key to diagnosing issues.
Normal Convergence: Both Epot and Fmax should decrease steadily, albeit with occasional oscillations. The run concludes with a message such as "Optimization converged: Fmax < [value]".
Slow Convergence: If Fmax plateaus at a value above
emtolafter many steps, the system may be trapped in a shallow region of the potential energy surface. Mitigation strategies include:Lack of Convergence: If the maximum number of steps (
nsteps) is reached without achieving the force tolerance, the minimization is incomplete. The solution is to continue the minimization from the last obtained structure. This can be done by creating a new .tpr file withgmx grompp, using the last frame (e.g.,.grofile) as the-cinput and the checkpoint (.cpt) file with the-tflag, then rerunningmdrun[41].
Table 2: Troubleshooting Common Energy Minimization Issues
| Problem | Possible Causes | Solutions |
|---|---|---|
| Slow convergence | System trapped in shallow minimum; poor initial structure. | Increase emstep; switch minimizer; ensure proper system preparation [42]. |
| Fmax oscillates | Step size too large. | Use a smaller emstep; employ steepest descent for initial stabilization. |
| nsteps reached | Inadequate step limit for system size/complexity. | Extend minimization with gmx convert-tpr -extend or restart with gmx grompp [41]. |
| Very high initial Epot | Severe atomic overlaps; incorrect protonation states. | Check initial structure; verify topology building with gmx pdb2gmx [42]. |
Successful energy minimization and analysis require a suite of computational "reagents" and tools. The following table details the essential components.
Table 3: Key Research Reagent Solutions for GROMACS Energy Minimization
| Item | Function | Usage Notes |
|---|---|---|
| Input Structure (.gro, .pdb) | Provides initial atomic coordinates. | Must be properly solvated and ionized; check for missing atoms [42]. |
| Topology File (.top) | Defines molecular structure and force field parameters. | Generated via gmx pdb2gmx or manually; critical for accurate energy evaluation [43] [42]. |
| Run Input File (.tpr) | Portable binary containing system topology, parameters, and state. | Produced by gmx grompp; the input for gmx mdrun [2] [43]. |
| Parameter File (.mdp) | Specifies minimization algorithm and control parameters. | Contains integrator, emtol, emstep, nsteps [25]. |
| Checkpoint File (.cpt) | Saves complete state of the simulation for restart. | Allows exact continuation after interruption [2] [41]. |
| Force Field Databanks | Libraries of residue templates and parameters. | Located in share/top; modified by placing edited copies in working directory [42]. |
| Energy File (.edr) | Contains energy values, temperature, pressure. | Analyzed with gmx energy to plot energy trends during minimization [2] [43]. |
Advanced Protocols and Performance Considerations
Protocol for Multi-Stage Energy Minimization
For complex systems, such as protein-ligand complexes in drug development, a multi-stage minimization protocol significantly enhances stability.
Stage 1: Solvent and Ion Relaxation
- Objective: Relax the solvent and ions around a fixed solute to relieve bad contacts from the solvation process.
- Method: Use steepest descent with position restraints applied to the protein and ligand heavy atoms. The
.mdpsettings include: - Analysis: Verify that the potential energy and Fmax decrease significantly.
Stage 2: Full System Minimization
- Objective: Achieve a stable, low-energy configuration for the entire system.
- Method: Employ a combination of algorithms. Start with steepest descent for robustness, then switch to conjugate gradient or L-BFGS for efficient convergence.
- Analysis: Confirm Fmax is below the target tolerance. The final structure is suitable for equilibration in a production MD run.
Performance and Hardware Utilization
A critical performance consideration is that energy minimization is not currently supported on GPUs in GROMACS [44]. Attempts to use GPU flags (-nb gpu -pme gpu) will result in an error. Consequently, minimization performance relies entirely on CPU parallelization [45].
- MPI Parallelization: Use multiple MPI ranks (
mpirun -np <N> gmx_mpi mdrun ...) to distribute the computational load across CPU cores. The optimal number of ranks depends on the system size and processor architecture. - OpenMP Multithreading: Combining MPI with OpenMP threads (controlled via
-ntomp) can improve performance by leveraging shared memory within a compute node. - SIMD Acceleration: Compiling GROMACS with the appropriate SIMD instructions (e.g., AVX2, AVX512) for the target CPU is essential for achieving the highest possible performance in the non-bonded and bonded kernels [45].
In summary, proficient interpretation of GROMACS energy minimization output is a cornerstone of robust molecular simulation. By systematically analyzing log files, understanding convergence metrics, and applying structured protocols, researchers can ensure their systems are properly prepared for subsequent dynamics simulations, thereby laying a solid foundation for scientifically sound results in drug development and structural biology.
Solving Common Energy Minimization Failures and Performance Issues
Energy minimization (EM) is a foundational step in molecular dynamics (MD) simulations, serving to relieve steric clashes, eliminate unrealistic geometries, and prepare the system for subsequent dynamics simulations [46]. Within the GROMACS ecosystem, the mdrun command executes this crucial process, seeking to find the nearest local minimum in the potential energy landscape. Successful minimization is characterized by the maximum force (Fmax) falling below a specified threshold (emtol), indicating that the system has reached a stable configuration [1]. However, researchers frequently encounter a persistent and problematic scenario: energy minimization halts without achieving the target Fmax. This convergence failure presents a significant obstacle, potentially compromising the stability and physical validity of all subsequent simulation stages, including equilibration and production MD.
This application note, framed within broader thesis research on the GROMACS mdrun command, provides a comprehensive diagnostic and procedural framework for addressing force convergence failures. We synthesize methodological insights with practical protocols to assist researchers, scientists, and drug development professionals in effectively troubleshooting and resolving these issues, thereby ensuring the robustness of their computational workflows.
Theoretical Foundation: Energy Minimization Algorithms in GROMACS
GROMACS provides several algorithms for energy minimization, each with distinct mechanical foundations and performance characteristics. Understanding these algorithms is prerequisite for selecting the appropriate method and diagnosing its failures.
The steepest descent algorithm follows the negative energy gradient, moving atom positions in the direction of the greatest force reduction. New positions are calculated using (\mathbf{r}{n+1} = \mathbf{r}n + \frac{hn}{\max(|\mathbf{F}n|)} \mathbf{F}n), where (hn) is the maximum displacement and (\mathbf{F}_n) is the force vector [1]. The step size is dynamically adjusted; it increases by 20% after a successful step that lowers the potential energy and decreases by 80% after a rejected step [1]. While not the most efficient search method, steepest descent is highly robust for initially relieving severe steric clashes and is therefore often recommended for the initial stages of minimization [1].
The conjugate gradient algorithm builds upon steepest descent by incorporating information from previous search directions. This allows it to form a series of conjugate search directions, which generally leads to more efficient convergence near the energy minimum. However, a significant limitation is that conjugate gradient cannot be used with constraints, meaning flexible water models must be employed if this algorithm is selected [1].
The L-BFGS (limited-memory Broyden-Fletcher-Goldfarb-Shanno) algorithm is a quasi-Newtonian method that approximates the inverse Hessian matrix to achieve faster convergence. It maintains a fixed number of corrections from previous steps to build this approximation, resulting in memory requirements proportional to the number of particles rather than its square [1]. In practice, L-BFGS often converges faster than conjugate gradients but has the current limitation of lacking parallelization [1].
Table 1: Key Energy Minimization Algorithms in GROMACS
| Algorithm | Mechanism | Strengths | Limitations | Typical Use Case |
|---|---|---|---|---|
| Steepest Descent [1] | Follows negative energy gradient | High robustness; Handles severe clashes | Slow convergence near minimum | Initial minimization of strained systems |
| Conjugate Gradient [1] | Builds conjugate search directions | More efficient near minimum | Incompatible with constraints [1] | Pre-normal-mode analysis minimization |
| L-BFGS [1] | Approximates inverse Hessian | Fastest convergence | Not yet parallelized [1] | Minimization of medium-sized systems |
The stopping criterion for these algorithms is typically defined by the emtol parameter, which sets the threshold for the maximum force (Fmax) on any atom. A reasonable value for (\epsilon) (emtol) can be estimated from the root mean square force of a harmonic oscillator, with values between 1 and 100 kJ mol⁻¹ nm⁻¹ generally being acceptable, depending on the system and desired accuracy [1].
Diagnosing Non-Convergence: A Systematic Workflow
When energy minimization fails to converge, a systematic diagnostic approach is essential to identify the root cause. The process involves checking key output metrics, analyzing energy trends, and inspecting the molecular structure.
Key Diagnostic Metrics and Their Interpretation
Upon completion of an EM run, two primary metrics must be immediately examined: the potential energy (Epot) and the maximum force (Fmax) [46].
- Potential Energy (Epot): For a solvated protein system, a reasonable Epot should be negative and typically on the order of 10⁵ to 10⁶, scaled by system size [46] [3]. A positive Epot, or an excessively high negative value, often indicates serious structural problems such as atomic overlaps or incorrect topology.
- Maximum Force (Fmax): This is the largest force component on any single atom, and it is compared directly to the
emtoltarget. Convergence is achieved only when Fmax <emtol[46]. The value of Fmax provides a direct indicator of local strain within the structure.
The following diagnostic workflow provides a logical pathway for investigating convergence failures:
Diagram Title: Diagnostic Workflow for Force Convergence Failure
Quantitative Diagnostics Table
The table below summarizes key diagnostic indicators, their acceptable ranges, and associated interpretations to guide the troubleshooting process.
Table 2: Diagnostic Indicators for Energy Minimization Convergence
| Diagnostic Indicator | Acceptable Range/Pattern | Problematic Signature | Potential Interpretation |
|---|---|---|---|
| Potential Energy (Epot) [46] [3] | Negative (-10⁵ to -10⁶ for protein-water) | Positive or excessively high negative value | Severe steric clashes, topology errors |
| Maximum Force (Fmax) [46] | Fmax < emtol (e.g., < 1000 kJ mol⁻¹ nm⁻¹) |
Fmax >> emtol (e.g., 10⁴-10⁵) |
Localized strain, atomic overlaps |
| Energy Trajectory [46] | Steady decrease to plateau | Rapid initial drop then early plateau | Algorithm cannot find lower minimum |
| Force Norm | Decreasing trend | Stagnant or increasing | System strain too high for parameters |
Case examples from user forums illustrate these diagnostics in practice. One researcher reported an EM run stopping with Fmax = 7.07×10⁴ kJ mol⁻¹ nm⁻¹ (far above the target of 1000) despite a seemingly reasonable Epot of -4.70×10⁵ [4]. Another case showed Fmax actually increasing during minimization (from 1.92×10⁵ to 2.31×10⁵) while Epot became less negative, indicating potential structural issues preventing proper relaxation [47].
Resolution Protocols and Experimental Methodologies
When diagnostics identify the nature of the convergence failure, specific resolution protocols can be implemented. The following workflow outlines a sequential approach to resolving persistent convergence problems:
Diagram Title: Sequential Resolution Protocol for Convergence Problems
Parameter Adjustment Methodology
The most straightforward interventions involve modifying the minimization parameters in the molecular dynamics parameters (.mdp) file.
Increasing Iteration Count: The maximum number of minimization steps is controlled by the
nstepsparameter. If the energy trajectory shows a steady downward trend when minimization stops, simply increasingnstepsfrom 50,000 to 100,000 or higher may provide sufficient iterations to reach convergence [46] [4]. The GROMACS documentation suggests that a reasonable value for the force toleranceemtolis between 1 and 100, with tighter tolerances requiring more steps [1].Step Size Optimization: The
emstepparameter sets the initial step size for steepest descent minimization. If the system is highly strained, a large step size (e.g., 0.01 nm) might cause instability, while an overly small step size (e.g., 0.0001 nm) can needlessly prolong minimization. A recommended protocol is to start with a moderate step size (0.001 nm) and adjust based on system response [46].Algorithm Selection and Sequencing: For systems that fail to converge with steepest descent, switching to a more efficient algorithm may be beneficial. A robust protocol employs steepest descent initially to relieve severe clashes, then switches to conjugate gradients or L-BFGS for finer convergence [1]. Note that conjugate gradients require flexible water models as they cannot handle constraints [1].
Table 3: Resolution Protocols for Common Convergence Problems
| Problem Scenario | Primary Intervention | Alternative Approach | mdp Parameter Modifications |
|---|---|---|---|
| Early Step Termination [46] | Increase maximum steps | - | nsteps = 100000 (from 50000) |
| System Instability [46] | Reduce step size | Switch to steepest descent | emstep = 0.001 integrator = steep |
| Slow Convergence near Minimum [1] | Switch to more efficient algorithm | Combine algorithms | integrator = cg or l-bfgs |
| Constraint Conflicts [1] [4] | Use flexible water models | Remove constraints | define = -DFLEXIBLE constraints = none |
Structural and Topology Verification Protocols
When parameter adjustments prove insufficient, the problem likely lies with the molecular structure or its topological representation.
Steric Clash Identification and Relief: Visual inspection of the initial structure using molecular visualization software (e.g., SAMSON [46], VMD, or PyMOL) is essential for identifying atomic overlaps, particularly in side-chain packing, ligand binding sites, or loop regions. These clashes can generate extremely high local forces that prevent convergence. In severe cases, a multi-stage minimization protocol with progressively tighter position restraints may be necessary to gradually relax the system.
Topology and Charge Validation: Incorrect topologies—such as missing atoms [48], incorrect bond parameters, or non-neutral system charge—can create unnatural forces that resist minimization. Verification protocols include:
- Checking for missing atoms or bonds in the topology [48] [49]
- Ensuring the total system charge is properly neutralized with ions [49]
- Validating that all residue and ligand names match force field expectations [48]
- Confirming position restraint files are correctly ordered and included in the topology [48]
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful energy minimization requires careful preparation and validation of system components. The following table catalogues essential "research reagents" in computational chemistry and their functions within the minimization workflow.
Table 4: Essential Research Reagent Solutions for Energy Minimization
| Reagent/Material | Function/Purpose | Implementation Example | Validation Method |
|---|---|---|---|
| Force Field [49] | Defines potential energy function; atomic parameters | ffamberSB for proteins, OPLS-AA for small molecules |
Match residue/ligand names to database [48] |
| Water Model [1] | Solvation environment; handling of solvent-solute interactions | SPC, TIP3P, TIP4P (flexible for CG) | Use -DFLEXIBLE with conjugate gradients [1] |
| Ion Parameters [49] | System neutralization; physiological ionic concentration | genion for replacing solvent molecules with ions |
Check final system charge is neutral [49] |
| Position Restraints [48] | Constrain specific atoms during minimization | posre.itp files for protein backbone heavy atoms |
Verify restraint file order in topology [48] |
| Topology File [48] [49] | Describes molecular composition, connectivity, parameters | Generated by pdb2gmx or manually for ligands |
Check for missing atoms/bonds [48] [49] |
Diagnosing and resolving force convergence problems requires a systematic approach that combines algorithmic understanding with practical intervention strategies. Based on our analysis of GROMACS documentation and real-world case studies, the following best practices emerge:
First, always verify both the potential energy and maximum force after minimization. A negative and reasonable Epot with a slightly elevated Fmax may be acceptable for proceeding to equilibration, particularly when the energy has plateaued [46]. Second, implement a sequential optimization strategy, beginning with simple parameter adjustments (increasing nsteps, reducing emstep) before progressing to more complex structural and topological fixes. Third, recognize that different minimization algorithms have distinct strengths—steepest descent for initial strain relief, conjugate gradients or L-BFGS for finer convergence [1].
When these approaches fail, the solution often lies in revisiting the fundamental building blocks of the system: verifying topology integrity [48] [49], ensuring proper charge neutralization [49], and meticulously inspecting for structural anomalies [46]. By adopting this structured framework for diagnosing and addressing force convergence problems, researchers can significantly enhance the reliability and efficiency of their molecular dynamics pipelines, laying a solid foundation for robust and scientifically valid simulation outcomes.
Molecular dynamics (MD) simulations are a cornerstone of modern computational biology and drug development, enabling researchers to study the structure, dynamics, and function of biomolecular systems. The reliability of these simulations, however, is critically dependent on the initial structural model. Artifacts within this starting structure, primarily steric clashes and missing atoms, can lead to simulation instability, unphysical forces, and inaccurate results. Steric clashes, characterized by the unphysical overlap of non-bonding atoms, introduce severe repulsive forces that can destabilize a simulation. Missing atoms, often hydrogens omitted from crystal structures or incomplete side chains in homology models, result in an incomplete molecular description and incorrect interactions. This application note details a robust methodology for identifying and rectifying these structural issues using the GROMACS mdrun command for energy minimization, framed within a broader research context to ensure the production of stable, physically accurate simulation systems.
Quantitative Analysis of Structural Defects
A quantitative understanding of structural defects is essential for setting appropriate refinement targets. Steric clashes are not merely binary occurrences but vary in severity based on the degree of atomic overlap.
Defining and Quantifying Steric Clashes
A robust quantitative definition for a steric clash is any atomic overlap that results in a Van der Waals repulsion energy greater than 0.3 kcal/mol (approximately 0.5 kBT) [50]. This metric is more informative than a simple distance cutoff, as it accounts for the different repulsive potentials of various atom types. To normalize for protein size, the clash-score is defined as the total clash-energy divided by the number of inter-atomic contacts evaluated [50].
Statistical analysis of high-resolution crystal structures (resolution < 2.5 Å) provides a benchmark for acceptable clash levels in refined models. The distribution of clash-scores from a dataset of 4,311 unique protein chains establishes a quality threshold [50].
Table 1: Clash-score Statistics from High-Resolution Protein Structures [50]
| Dataset | Number of Structures | Mean Clash-score (kcal.mol⁻¹.contact⁻¹) | Acceptable Clash-score Threshold (Mean + 1 SD) |
|---|---|---|---|
| High-Resolution X-ray | 4,311 | ~0.01 | 0.02 |
A structure is considered acceptable for simulation if its clash-score is below the threshold of 0.02 kcal.mol⁻¹.contact⁻¹ [50]. Low-resolution structures and homology models typically exhibit significantly higher clash-scores, necessitating targeted minimization before MD simulation.
Consequences of Unresolved Clashes
Unresolved steric clashes present a major obstacle for MD simulations. The high repulsive forces can cause numerical instability, leading to simulation crashes. Furthermore, standard energy minimization algorithms like conjugate gradient may fail to resolve severe clashes, as the initial forces can be too large for the minimizer to handle effectively [50]. This underscores the need for specialized protocols, particularly for processing low-quality structural models.
Energy Minimization Algorithms in GROMACS
The GROMACS mdrun module provides several algorithms for energy minimization (EM), each with distinct advantages and limitations for addressing structural issues [1].
Table 2: Energy Minimization Algorithms in GROMACS [1]
| Algorithm | Principle | Strengths | Weaknesses | Recommended Use Case |
|---|---|---|---|---|
| Steepest Descent | Moves atoms along the direction of the negative energy gradient (force). Adaptively adjusts step size. | Robust, stable convergence from high-energy states. Easy to implement. | Slow convergence near the energy minimum. | Initial restructuring of models with severe steric clashes. |
| Conjugate Gradient | Uses the conjugate of previous gradients to find better search directions. | More efficient convergence than Steepest Descent near the minimum. | Cannot be used with constraints (e.g., SETTLE water). | Final polishing of systems with flexible water or pre-equilibration for normal-mode analysis. |
| L-BFGS (Limited-memory Broyden-Fletcher-Goldfarb-Shanno) | A quasi-Newton method that builds an approximation of the energy Hessian matrix. | Fastest convergence for large, complex systems. Low memory requirements. | Not yet parallelized in GROMACS. Sensitive to force field cut-offs. | Recommended for most minimization tasks, especially for large systems, when parallelization is not critical. |
The choice of algorithm is specified via the integrator parameter in the mdrun command. For structures with severe artifacts, a common strategy is to begin with the robust Steepest Descent algorithm to resolve major clashes, followed by a switch to L-BFGS or Conjugate Gradient for efficient convergence to the final minimum [1].
Experimental Protocols for Structural Refinement
The following protocols provide detailed methodologies for preparing and minimizing protein structures, incorporating steps to add missing atoms and resolve steric clashes.
Comprehensive Workflow for Structural Refinement
The diagram below outlines the logical sequence of steps involved in taking a raw input structure to a minimized system ready for simulation.
Protocol 1: Addressing Missing Atoms and Preparing the System
This protocol focuses on building a complete all-atom model, which is a prerequisite for meaningful energy minimization.
Add Missing Atoms: Use
gmx pdb2gmxto convert a protein structure file (e.g., .pdb) into a GROMACS-compatible format (.gro) and generate the corresponding topology.gmx pdb2gmx -f input.pdb -p topol.top -o protein.gro -ignh -ff [forcefield] -water [watermodel]- The
-ignhflag instructs the program to ignore hydrogen atoms present in the input file and instead add them according to the chosen force field's specifications. This ensures correct protonation states and geometry [51] [52]. - The tool uses a hydrogen database (.hdb) file to correctly place hydrogens on residues. Ensure atom names in the force field's .rtp and .hdb files are consistent to avoid errors [52].
Solvate the System: Use
gmx solvateto embed the protein in a box of water molecules.gmx solvate -cp protein.gro -cs [watermodel] -o protein_solv.gro -p topol.top
Add Ions: Use
gmx genionto replace water molecules with ions to neutralize the system's charge and achieve a desired ionic concentration.- First, prepare a run input file with
gmx grompp -f ions.mdp -c protein_solv.gro -p topol.top -o ions.tpr - Then, run
gmx genion -s ions.tpr -c protein_solv.gro -o protein_solv_ions.gro -p topol.top -pname NA -nname CL -neutral
- First, prepare a run input file with
Protocol 2: Energy Minimization with GROMACS mdrun
This protocol describes the actual energy minimization process to relieve steric clashes and strains introduced during system setup [3].
Prepare Parameters and Input: Create a parameter file (e.g.,
minim.mdp) that specifies the minimization settings. A basic example is:Assemble Binary Input: Use
gmx gromppto combine the structure, topology, and parameters into a single portable binary run input file (.tpr).gmx grompp -f minim.mdp -c protein_solv_ions.gro -p topol.top -o em.tpr
Run Energy Minimization: Execute the minimization using
mdrun.gmx mdrun -v -deffnm em- The
-vflag provides verbose output, and-deffnm emdefines a common filename prefix for all output files (em.log,em.edr,em.trr,em.gro) [3].
Analyze the Results:
- Success Criteria: The minimization is successful if the maximum force (
Fmax) reported in the log file is below the target tolerance (emtol, e.g., 1000 kJ mol⁻¹ nm⁻¹). The potential energy (Epot) should be negative and on the order of 10⁵-10⁶ for a solvated protein system [3]. - Energy Analysis: Use
gmx energyto extract and plot the potential energy over the minimization steps to visualize convergence.gmx energy -f em.edr -o potential.xvg
- Success Criteria: The minimization is successful if the maximum force (
The Scientist's Toolkit: Research Reagent Solutions
The following table details the essential software tools and file formats used in the protocols for structural refinement with GROMACS.
Table 3: Essential Research Reagents and Tools for GROMACS Minimization
| Item Name | Type/Format | Function in the Protocol |
|---|---|---|
| GROMACS | Software Suite | The primary simulation engine used for energy minimization and molecular dynamics. The mdrun command is the core executable for performing calculations [1] [3]. |
| Force Field | Topology/Parameter Files | A set of mathematical functions and parameters (e.g., CHARMM36, AMBER) that define the potential energy of the system. It is essential for accurately calculating atomic interactions during minimization [52]. |
| Hydrogen Database (.hdb) | Database File | Used by gmx pdb2gmx to determine how to correctly add hydrogen atoms to residues. Consistency with the residue topology (.rtp) file is critical [52]. |
| Portable Binary Input (.tpr) | Binary File | A self-contained file generated by gmx grompp that includes the system structure, topology, and all simulation parameters. It is the input for gmx mdrun [51]. |
| Structure File (.gro/.pdb) | Coordinate File | Contains the atomic coordinates of the system. The .gro format is GROMACS-native and is the typical output of gmx pdb2gmx and gmx solvate [51]. |
| Energy File (.edr) | Binary Data File | Records the energies of all interaction terms during the minimization. It is analyzed post-minimization with gmx energy to verify convergence [51] [3]. |
The integrity of the initial atomic model is a non-negotiable foundation for reliable molecular dynamics simulations. By systematically addressing steric clashes and missing atoms through the application notes and protocols outlined herein, researchers can ensure their systems are physically realistic and stable. The quantitative framework for assessing clashes, combined with the strategic use of GROMACS's energy minimization algorithms—leveraging the robustness of Steepest Descent for initial restructuring and the efficiency of L-BFGS for final convergence—provides a clear and effective path from a flawed input structure to a minimized system ready for subsequent equilibration and production MD. This process is a critical first step in any rigorous simulation study, directly impacting the validity of scientific conclusions in computational drug development and biomolecular research.
Solving Segmentation Faults and Memory Allocation Errors
Within the framework of research utilizing the GROMACS mdrun command for energy minimization, achieving a stable and efficiently running simulation is a prerequisite for obtaining scientifically valid results. Segmentation faults and memory allocation errors represent two of the most common and disruptive classes of problems encountered during this phase. A segmentation fault indicates that a program has attempted to access a memory location it is not permitted to, causing an immediate crash [53]. Conversely, an "Out of memory" error signifies that the program's request for additional RAM could not be fulfilled by the operating system [48]. For researchers and drug development professionals, these errors not only halt critical workflows but also lead to significant losses in computational time and resources. This application note provides a structured diagnostic and mitigation protocol to resolve these issues, ensuring robust and efficient energy minimization steps in the molecular dynamics pipeline.
Understanding the Errors
Segmentation Faults
A segmentation fault (core dumped) is a fatal error that terminates the mdrun process. In the context of GROMACS, this often occurs during the initialization of a simulation, before the first step is completed [53]. The error log may show no obvious issues, with the simulation crashing immediately after reporting on constraint solvers or communication setup [53]. The underlying causes can be diverse, including software bugs in specific GROMACS versions, incompatibilities in the molecular topology (such as certain virtual site types), or problems with the parallel execution environment [54]. Kernel messages often accompany these faults, pointing to memory access violations in specific processes or shared memory segments [53].
Memory Allocation Errors
Memory allocation failures occur when mdrun exhausts the available RAM on a node. The official GROMACS manual explicitly describes this error as an attempt "to assign memory... due to insufficient memory" [48]. A telltale sign of a configuration-based memory explosion is an exponential increase in RAM usage when scaling to more nodes. For instance, a simulation requiring 21 GB on 8 nodes might demand over 5 TB on 16 nodes, indicating a severe misconfiguration rather than a genuine memory shortage [55]. This often stems from an inefficient ratio of MPI tasks to nodes or OpenMP threads, leading to excessive memory duplication and communication overhead.
Diagnostic and Mitigation Protocol
The following workflow provides a systematic approach to diagnosing and resolving segmentation faults and memory errors in GROMACS mdrun. Adhere to this sequence for efficient troubleshooting.
Diagnostic Workflow
Step-by-Step Mitigation Procedures
Protocol 1: Resolving Segmentation Faults
- Inspect Log Files: Immediately after a crash, examine the
.logfile and terminal output. Look for the last informational message before the failure. In one documented case, the simulation crashed after the "Linking all bonded interactions" message and a warning about output file size, without reaching the first step [53]. - Identify Version-Specific Bugs: Compare your GROMACS version with known issues. A confirmed bug in version 2023.1 caused segmentation faults during energy minimization with certain Martini systems, which was resolved by reverting to version 2022.5 [54].
- Procedure: Install a known stable version of GROMACS (e.g., 2022.5). Re-run the
gromppcommand to regenerate the.tprfile and executemdrunagain.
- Procedure: Install a known stable version of GROMACS (e.g., 2022.5). Re-run the
- Check System Topology: Run a sanity check on your input files. Use
gmx checkon your.tprfile to identify potential inconsistencies. - Simplify the Test Case: If possible, create a minimal system (e.g., a single protein in a small water box) to test if the error persists. This helps isolate the problem to the system configuration or the software environment.
Protocol 2: Resolving Memory Allocation Errors
- Profile Memory Usage: Run your simulation on a small number of nodes (e.g., 1-2) and use tools like
sacctortopto monitor the maximum memory used (MaxRSS). This establishes a baseline [55]. - Optimize Parallelization Configuration: The primary lever for controlling memory usage is the balance between MPI processes and OpenMP threads. A configuration that uses too many MPI tasks is a common cause of memory bloat [55].
- Procedure: Drastically reduce the number of MPI tasks per node (
-ntmpi) and correspondingly increase the number of OpenMP threads per MPI task (-ntomp). For example, instead ofsrun -n 512 -c 2 gmx_mpi mdrun -ntomp 2, trysrun -n 64 -c 16 gmx_mpi mdrun -ntomp 16[55]. The goal is to have fewer, more powerful MPI ranks that each handle a larger domain.
- Procedure: Drastically reduce the number of MPI tasks per node (
- Adjust Domain Decomposition: Use
mdrunoptions like-ddto manually control the domain decomposition grid, ensuring the domains are as cubic as possible. Avoid creating domains that are too thin in one dimension.
Quantitative Data and Performance Optimization
Parallel Configuration Impact on Performance and Memory
The following table summarizes real-world data on how parallel configuration affects simulation stability and resource consumption.
Table 1: Impact of Parallel Configuration on Simulation Run Parameters
| Scenario | MPI Tasks (-ntmpi) |
OpenMP Threads (-ntomp) |
Memory Utilization | Simulation Outcome | Performance | Source |
|---|---|---|---|---|---|---|
| Problematic Setup | 512 | 2 | 5.31 TB (OOM) | OUTOFMEMORY | N/A | [55] |
| Optimized Setup | 64 | 16 | ~21 GB | Successful | 90 ns/day | [55] |
| Alternative Test 1 | 128 | 8 | Not Specified | Successful | To be benchmarked | [55] |
| Alternative Test 2 | 256 | 4 | Not Specified | Successful | To be benchmarked | [55] |
Key Configuration Concepts formdrun
Table 2: Essential GROMACS mdrun Options for Managing Memory and Stability
| Option | Function | Recommended Use for Mitigation |
|---|---|---|
-ntmpi |
Sets the number of MPI processes. | Reduce this number to lower memory overhead and communication costs. |
-ntomp |
Sets the number of OpenMP threads per MPI process. | Increase this number when reducing -ntmpi. Should not exceed the number of physical cores per socket. |
-dd |
Manually sets the domain decomposition grid. | Use to create balanced, cubic domains if auto-decomposition fails. |
-dlb |
Enables dynamic load balancing. | Can be set to "no" to improve stability if the system is heterogeneous. |
-nstlist |
Adjusts neighbor search frequency. | Increasing this (e.g., to 300) reduces CPU load and can improve GPU utilization [39]. |
The Scientist's Toolkit: Research Reagent Solutions
This table lists the essential software and computational "reagents" required for running and troubleshooting GROMACS energy minimization simulations.
Table 3: Essential Research Reagents for GROMACS Energy Minimization
| Item | Function in Protocol | Specification & Notes |
|---|---|---|
| GROMACS Installation | The primary simulation engine that executes the mdrun command. |
Use a stable version (e.g., 2022.5). Ensure it is compiled with the appropriate GMX_SIMD architecture (e.g., AVX2_256, AVX512) for the target CPU [45]. |
| Molecular Topology (.top) | Defines the molecular system's structure, forces, and parameters. | Must be consistent with the coordinate file. Generated via pdb2gmx or manually. Errors here can cause segmentation faults [48] [49]. |
| Run Input File (.mdp) | Defines all parameters for energy minimization and simulation. | For EM, key parameters include integrator = steep, emtol = 1000.0, and nsteps [3]. Incorrect settings can lead to instability. |
| Binary Input File (.tpr) | The portable binary input for mdrun. |
Assembled by grompp from the topology, coordinates, and .mdp file. The direct input for mdrun. |
| Thread-MPI/OpenMPI | Libraries enabling parallel computation. | Thread-MPI is for single-node; OpenMPI for multi-node. Misconfiguration is a primary source of memory errors [55]. |
| Job Scheduler (Slurm) | Manages computational resources on a cluster. | Used to request appropriate nodes, tasks, and memory. The srun command launches the parallel mdrun job [53]. |
Successfully navigating segmentation faults and memory allocation errors in GROMACS mdrun is a critical skill that ensures the reliability and throughput of energy minimization research. The protocols outlined herein emphasize a systematic approach: first, diagnose the error type via logs; second, rule out software version bugs; and third, meticulously optimize the parallelization configuration. The quantitative data demonstrates that a few key parameters, particularly the ratio of MPI tasks to OpenMP threads, have an outsized impact on both memory consumption and simulation stability. By adhering to these application notes, researchers can transform a process of troubleshooting from one of frustration into one of efficient problem-solving, thereby accelerating the journey from molecular setup to production simulation.
Within the framework of molecular dynamics (MD) simulations using GROMACS, energy minimization (EM) represents a critical preliminary step essential for ensuring numerical stability and achieving physically meaningful results in subsequent production runs. This protocol is situated within a broader thesis investigating the advanced use of the gmx mdrun command for biomolecular systems, with a particular focus on applications in rational drug development. The primary objective of energy minimization is to relieve unfavorable atomic contacts, resolve steric clashes, and relocate the system to the nearest local minimum on the potential energy surface by systematically reducing the total potential energy and the maximum force acting on the atoms [17] [5]. For researchers and scientists engaged in drug development, a meticulously minimized starting structure is indispensable for studying protein-ligand interactions, conducting free-energy calculations, and performing comparative structural analyses, as it removes the excessive strain that could otherwise lead to unrealistic dynamics or simulation failure. This document provides a comprehensive, practical guide to optimizing EM protocols by detailing the available algorithms, their associated parameters, and strategic considerations for maximizing computational efficiency and robustness, particularly for large biomolecular systems such as protein-ligand complexes.
Energy Minimization Algorithms in GROMACS
GROMACS provides three principal algorithms for energy minimization, each characterized by distinct operational mechanisms and performance profiles. The choice of algorithm is governed by the integrator keyword in the molecular dynamics parameter (.mdp) file [25].
Steepest Descent (integrator = steep)
The Steepest Descent algorithm is a robust and straightforward method that moves atomic coordinates in the direction of the negative energy gradient, which corresponds to the force. Its key operational parameters are the maximum displacement per step (emstep, typically in nm) and the force tolerance (emtol, in kJ mol⁻¹ nm⁻¹) that defines the convergence criterion [1] [25].
The algorithm proceeds by calculating new positions, (\mathbf{r}{n+1} = \mathbf{r}n + \frac{hn}{\max(|\mathbf{F}n|)} \mathbf{F}n), where (hn) is the current maximum step size and (\mathbf{F}_n) is the force vector. The step size is dynamically adjusted based on energy change: it is increased by 20% following a successful step that lowers the energy, and reduced by 80% after a rejected step that raises the energy [1]. This adaptive step size makes Steepest Descent particularly effective for the initial stages of minimization, where it can rapidly quench high-energy clashes in poorly equilibrated systems. However, its convergence rate slows down significantly as it approaches the energy minimum, becoming inefficient for achieving very low force tolerances [1].
Conjugate Gradient (integrator = cg)
The Conjugate Gradient method represents a more sophisticated approach that utilizes information from previous steps to construct a series of non-interfering, conjugate search directions. This strategy avoids the oscillatory behavior often observed with Steepest Descent, particularly in narrow energy valleys [1].
A notable implementation detail in GROMACS is that the Conjugate Gradient algorithm periodically incorporates a Steepest Descent step to enhance stability, with the frequency controlled by the nstcgsteep parameter [25]. Its primary convergence criterion is the force tolerance specified by emtol. A critical limitation for biomolecular simulations is that Conjugate Gradient cannot be used with constraints, meaning rigid water models (e.g., SETTLE) are incompatible. Simulations using this algorithm must employ flexible water models, which is specified in the .mdp file via define = -DFLEXIBLE [1] [25]. While Conjugate Gradient is generally slower than Steepest Descent during the initial relaxation phase, it demonstrates superior convergence efficiency closer to the energy minimum, making it the preferred algorithm for applications requiring high precision, such as minimization preceding normal-mode analysis [1].
L-BFGS (integrator = l-bfgs)
The L-BFGS (Limited-memory Broyden–Fletcher–Goldfarb–Shanno) algorithm is a quasi-Newton method that builds an approximation of the inverse Hessian matrix using a limited history of previous steps and gradient evaluations [1]. This approach enables more informed step decisions than first-order methods like Steepest Descent or Conjugate Gradient, often resulting in faster convergence.
In practice, L-BFGS has been observed to converge faster than Conjugate Gradients [1] [25]. However, a significant performance consideration is that, due to the correction steps required by the algorithm, it has not yet been parallelized in GROMACS [1] [25]. Consequently, for large systems where massive CPU parallelization is beneficial, the performance advantage of L-BFGS may be diminished. Furthermore, the use of switched or shifted interaction functions can improve its convergence by ensuring the potential energy function remains consistent across the steps used to build the Hessian approximation [1].
Table 1: Comparative Analysis of Energy Minimization Algorithms in GROMACS
| Algorithm | integrator Keyword |
Key Parameters | Strengths | Limitations | Ideal Use Case |
|---|---|---|---|---|---|
| Steepest Descent | steep |
emstep (max step), emtol (tolerance) |
Highly robust, efficient initial relaxation, easy to tune [1] | Slow convergence near minimum, inefficient for low tolerances [1] | Initial stabilization of poorly equilibrated structures [1] |
| Conjugate Gradient | cg |
emtol (tolerance), nstcgsteep (SD step freq) [25] |
Faster final convergence than SD, more efficient for low tolerances [1] | Incompatible with constraints (requires flexible water) [1] | Pre-normal mode analysis; general minimization where high precision is needed [1] |
| L-BFGS | l-bfgs |
(Built-in history length) | Fastest convergence for many systems, quasi-Newton efficiency [1] [25] | Not yet parallelized; performance may vary with force switches [1] [25] | Systems where serial performance is paramount and very low forces are required |
Parameter Selection and Optimization Strategy
The effective application of energy minimization requires careful selection of parameters and a strategic approach to algorithm usage.
Core Parameters and Convergence Criteria
The primary criterion for successful minimization is the reduction of the maximum force (Fmax) below a specified tolerance. The emtol parameter defines this target force tolerance. A reasonable value for emtol must be chosen with consideration of the system and the intended follow-up calculations. An overly tight tolerance may lead to endless iterations due to numerical noise from force truncation [1]. A value between 100 and 1000 kJ mol⁻¹ nm⁻¹ is generally acceptable for preparing a system for molecular dynamics. The ultimate goal is to achieve a potential energy (Epot) that is negative and, for a solvated protein system, on the order of 10⁵ to 10⁶, scaled according to the system size and number of water molecules [5].
For the Steepest Descent algorithm, the emstep parameter is critical. It sets the maximum allowed displacement in any single step. While a larger value can speed up initial relaxation, an excessively large emstep can lead to instability and step rejection. The default value is often sufficient, but for systems with severe clashes, starting with a smaller value (e.g., 0.001 nm) may be necessary.
A Hybrid Minimization Protocol
A highly effective strategy for balancing speed and robustness involves a hybrid, multi-stage approach:
- Initial Relaxation with Steepest Descent: Begin minimization using the Steepest Descent algorithm with a moderate
emtol(e.g., 1000 kJ mol⁻¹ nm⁻¹) and a reasonable number of steps (e.g., 500-1000). This stage efficiently relieves major steric clashes and high-energy distortions. - Fine-Minimization with L-BFGS or Conjugate Gradient: Once the system is partially relaxed, switch to a more efficient algorithm like L-BFGS (if parallelization is not critical) or Conjugate Gradient to drive the system to a lower force tolerance (e.g., 100-500 kJ mol⁻¹ nm⁻¹). This two-stage protocol leverages the initial robustness of Steepest Descent and the superior convergence near the minimum of the other algorithms.
Performance Considerations and Hardware Utilization
A critical performance note for researchers planning computations is that energy minimization is not supported on GPUs in GROMACS [44]. Attempts to use GPU offloading flags (-nb gpu -pme gpu) will result in an error, as PME on the GPU does not support non-dynamical integrators [44]. Therefore, performance optimization for EM must focus on CPU parallelization.
For large systems, such as a protein with 1987 amino acids in a solvated box, efficient parallelization across multiple CPU cores is essential for achieving acceptable performance [44]. Users should leverage mdrun options for domain decomposition (-dd) and OpenMP threads (-ntomp) to fully utilize available CPU resources. The built-in SIMD acceleration in GROMACS, which is determined at compile time, will also significantly enhance the performance of the force calculations during minimization [56].
The following workflow diagram synthesizes the strategic decision-making process for configuring and executing an efficient energy minimization protocol.
Diagram 1: A strategic workflow for configuring and executing energy minimization in GROMACS, highlighting key decision points and the recommended two-stage algorithm approach.
Experimental Protocol and Validation
Step-by-Step Application Note
This protocol outlines the procedure for performing energy minimization on a solvated protein-ligand complex, a common scenario in drug development.
- Input Preparation: Ensure you have the required input files: a post-solvation structure file (
protein_solvated.gro), the corresponding topology file (topol.top), and a parameter file (em.mdp) [19]. - Parameter File (
em.mdp) Configuration: A sample parameter file for a hybrid minimization protocol is provided below.- For the first stage (Steepest Descent):
- For the second stage (L-BFGS):
- Generate the Binary Input: Use the
gromppcommand to process the inputs into a single portable binary run input file (.tpr). For the first stage: - Execute the Minimization: Run the energy minimization using
mdrun. Leverage CPU parallelization, for example, using 8 MPI ranks and 2 OpenMP threads per rank: After the first stage completes, repeat steps 3 and 4 using the output of the first stage as input for the second (-c em_first.gro) and the second-stage parameter file. - Validation and Analysis: Upon completion, carefully check the output for convergence. The primary indicators of success are:
- The final maximum force (
Fmax) is less than the specifiedemtol. - The potential energy (
Epot) is negative and has reached a stable plateau, as visualized in a plot of energy versus step number [5].
- The final maximum force (
Table 2: The Scientist's Toolkit - Essential Files and Commands for GROMACS Energy Minimization
| Item | File Extension/Command | Function and Purpose |
|---|---|---|
| Structure File | .gro / .pdb |
Contains the atomic coordinates of the system to be minimized [19]. |
| Topology File | .top |
Defines the molecular system, including atom types, bonds, angles, and non-bonded parameters [19]. |
| Parameters File | .mdp |
Contains all the algorithmic settings and parameters controlling the minimization process [25] [19]. |
| Portable Binary | .tpr |
The complete run input file generated by grompp, combining structure, topology, and parameters [19]. |
| Pre-Processor | gmx grompp |
Validates and processes inputs into the .tpr file for mdrun [19]. |
| MD Engine | gmx mdrun |
The main simulation engine that performs the energy minimization calculation [44]. |
Troubleshooting and Optimization
If minimization fails to converge within the allotted steps, several actions can be taken:
- Increase Step Count: The simplest solution is to increase the
nstepsparameter in the.mdpfile. - Adjust Step Size: For Steepest Descent, reducing
emstepcan prevent step rejections and improve stability in problematic systems. - Re-examine System Setup: Persistent failure often indicates issues with the initial system, such as unresolved steric clashes from the solvation process or incorrect ligand parametrization. Visualizing the structure at the step where
Fmaxis highest can reveal the location of the problem.
The following diagram illustrates a detailed, step-by-step workflow from system preparation to validation, incorporating the specific GROMACS commands required at each stage.
Diagram 2: A comprehensive procedural workflow for system preparation and energy minimization execution, from an initial PDB file to a validated structure ready for molecular dynamics.
Within the framework of advanced molecular dynamics (MD) simulations using GROMACS, proper system preparation and energy minimization are not merely preliminary steps but foundational requirements for obtaining physically meaningful and reproducible results. This application note details sophisticated protocols for multi-stage energy minimization and comprehensive system preparation, contextualized within broader research employing the gmx mdrun command. The primary goal of energy minimization is to locate low-energy configurations by adjusting atomic coordinates to reduce the potential energy of the system, thereby removing unphysical contacts and steric clashes that inevitably arise during the initial system construction phase [1] [35]. For researchers and scientists in drug development, a meticulously minimized and equilibrated system is a critical prerequisite for reliable simulations of protein-ligand interactions, free-energy calculations, and the observation of biologically relevant phenomena [35] [17].
This guide provides a detailed exposition of the available minimization algorithms in GROMACS, outlines a structured, multi-stage minimization protocol, and presents a comprehensive system preparation workflow, complete with validated parameters and troubleshooting strategies.
Energy Minimization Algorithms in GROMACS
The gmx mdrun program in GROMACS implements three principal algorithms for energy minimization, each with distinct performance characteristics and application niches [1]. The choice of algorithm is specified via the integrator parameter in the mdrun configuration.
Table 1: Comparison of Energy Minimization Algorithms in GROMACS
| Algorithm | Key Principle | Advantages | Limitations | Typical Use Cases |
|---|---|---|---|---|
| Steepest Descent | Moves atoms in the direction of the negative energy gradient (i.e., the force) [1]. | Robust, simple, and effective for quickly relieving severe steric clashes [1]. | Converges slowly near the energy minimum [1]. | Initial minimization stages for poorly structured systems; removing bad contacts. |
| Conjugate Gradient | Uses a sequence of conjugate (non-interfering) directions for more efficient convergence [1]. | More efficient than Steepest Descent closer to the energy minimum [1]. | Cannot be used with constraints (e.g., SETTLE water) [1]. | Minimization prior to normal-mode analysis; requires flexible water models. |
| L-BFGS (Limited-memory Broyden–Fletcher–Goldfarb–Shanno) | A quasi-Newton method that builds an approximation of the inverse Hessian matrix [1]. | Faster convergence than Conjugate Gradients; lower memory requirements than full BFGS [1]. | Not yet parallelized for distributed computing [1]. | Final minimization stages for high-precision convergence. |
A critical aspect of any minimization is defining a termination criterion. The simulation halts when the maximum force component on any atom falls below a user-defined tolerance, emtol [1]. An overly tight tolerance can lead to excessive, unnecessary iterations due to numerical noise. A reasonable value can be estimated from the root-mean-square force of a harmonic oscillator at a given temperature; for many biological systems, a tolerance between 1 and 100 kJ mol⁻¹ nm⁻¹ is acceptable [1].
A Multi-Stage Minimization Protocol
A single minimization algorithm is often insufficient for complex biomolecular systems. A robust strategy employs a multi-stage approach that leverages the strengths of different algorithms sequentially. The following workflow diagram illustrates this protocol, from initial structure to a minimized system ready for equilibration.
Stage 1: Initial Relaxation via Steepest Descent
The first stage aims to rapidly quench the system from a potentially high-energy state, resolving major atomic overlaps and distortions.
- Objective: Relieve severe steric clashes and reduce the potential energy significantly.
- Algorithm:
integrator = steepest-descent[1]. - Parameters:
emtol: 100 - 500 kJ mol⁻¹ nm⁻¹nsteps: 50 - 500
- Rationale: The Steepest Descent algorithm is highly effective for large, crude adjustments to the structure. Its robustness prevents premature termination when far from a minimum. The liberal force tolerance allows it to complete quickly, having done the "heavy lifting" of resolving major issues.
Stage 2: Convergent Minimization via Conjugate Gradient or L-BFGS
The second stage refines the structure to a much tighter tolerance, bringing the system close to a local energy minimum.
- Objective: Achieve a well-minimized starting structure for subsequent MD equilibration.
- Algorithm:
integrator = cgorintegrator = l-bfgs[1]. - Parameters:
emtol: 10.0 - 100.0 kJ mol⁻¹ nm⁻¹ (A common target is 10.0 for all-atom simulations) [1].nsteps: 100 - 1000
- Rationale: After the initial relaxation, Conjugate Gradient or L-BFGS provides superior convergence efficiency. L-BFGS is generally preferred if available and if parallelization is not a constraint, as it often converges faster [1]. If the system contains flexible water (necessary for Conjugate Gradient) or requires parallelization, L-BFGS should be used.
Comprehensive System Preparation Workflow
Energy minimization is only one component of a larger system preparation pipeline. A properly prepared system is vital for the stability and physical accuracy of subsequent MD simulations [35]. The following diagram outlines the complete sequence from an initial structure to a production-ready system.
Key Considerations for Successful Preparation
- Topology Generation: Use tools like
gmx pdb2gmxor external resources like the Automated Topology Builder or CHARMM-GUI to create a topology file that accurately describes the system's chemical structure and interaction parameters within the chosen force field (e.g., AMBER, CHARMM) [35]. A critical rule is to never mix parameters from different force fields, as this leads to unphysical results [57]. - Solvation and Ions: The simulation box is filled with solvent (e.g., water) using
gmx solvate, and ions are added to neutralize the system's charge and achieve a desired physiological concentration usinggmx genion[35]. - Pre-Solvation Minimization: For complex solutes, it is often necessary to perform an in vacuo energy minimization before introducing solvent. This helps to resolve internal conflicts within the solute structure itself [35].
- Force Field and Water Model: The choice of force field and water model must be consistent. During minimization, consider using a flexible water model and avoiding bond constraints to allow the entire system to relax without artificial restrictions [1] [35].
Table 2: Key Research Reagent Solutions for GROMACS System Preparation
| Item | Function | Examples & Notes |
|---|---|---|
| Force Field | Describes the potential energy surface and interactions between atoms. | AMBER99sb-ildn, CHARMM36, OPLS-AA. Selection is critical and system-dependent [57]. |
| Solvent Model | Represents the water and/or other solvent molecules in the system. | SPC, TIP3P, TIP4P. Must be compatible with the chosen force field [35]. |
| Topology File (.top) | Defines the molecular structure, atom types, and bonded/non-bonded interactions. | Generated by gmx pdb2gmx or external tools like CHARMM-GUI or acpype [35]. |
| Run Input File (.tpr) | A portable binary containing all simulation parameters, topology, and coordinates. | Produced by gmx grompp; read by gmx mdrun to execute the simulation [32]. |
| Database Files (.dat) | Provide parameters for atom types, VDW radii, etc. | Located in share/top; can be customized by placing a modified copy in the working directory [35]. |
AdvancedmdrunFeatures for Minimization
The gmx mdrun command offers several features that are particularly useful for managing minimization and preparation workflows:
- Controlling Simulation Length: Use
mdrun -nsteps <value>to override the number of minimization steps defined in the input file, ormdrun -maxh <hours>to ensure the job terminates before a cluster queue time limit [22]. - Checkpointing and Restarts: The
-cpioption allows a minimization to be continued from a checkpoint file, providing fault tolerance and enabling a researcher to extend a minimization that did not converge within the initial step limit [32]. - Reproducible Mode: For debugging purposes,
mdrun -reprodcan be used to run a simulation in a perfectly reproducible manner, eliminating performance-related variations between runs on the same hardware [22].
A methodical approach to system preparation and energy minimization, as detailed in this application note, is indispensable for achieving stable and scientifically valid molecular dynamics simulations. The recommended multi-stage minimization protocol, which leverages the unique strengths of the Steepest Descent and Conjugate Gradient/L-BFGS algorithms, provides a robust framework for handling a wide array of biological systems. When integrated into the comprehensive system preparation workflow—encompassing topology generation, solvation, and ionization—this protocol ensures that researchers, especially those in drug development, can generate reliable starting points for probing complex biological phenomena and interactions at the atomic level.
Ensuring Success: Validating Results and Comparing Algorithm Performance
Energy minimization is a critical first step in molecular dynamics (MD) simulations using GROMACS, serving to remove steric clashes and inappropriate geometry from the initial molecular system configuration prior to production dynamics. [17] [3] This process ensures system stability and prevents excessively large forces that could cause simulation failure. Within the broader context of GROMACS mdrun command research, understanding the performance characteristics of different minimization algorithms is essential for computational efficiency and reliable results. This application note provides a systematic benchmarking analysis of three principal energy minimization algorithms available in GROMACS—steepest descent, conjugate gradient, and L-BFGS—evaluating their convergence behavior and computational efficiency across diverse molecular systems.
The mdrun program in GROMACS implements these minimization algorithms through the integrator parameter in the molecular dynamics parameter (MDP) file. [1] Proper energy minimization is particularly crucial when starting from configurations far from equilibrium, where forces may be large enough to cause MD simulation failure. [17] Additionally, minimization removes kinetic energy from the system, allowing better comparison of structures from different dynamic simulations by reducing thermal noise. [17]
Theoretical Background of Minimization Algorithms
Algorithmic Fundamentals
GROMACS provides three main algorithms for energy minimization, each with distinct mathematical foundations and performance characteristics. The choice of algorithm depends on the specific system requirements and the desired balance between robustness and efficiency.
Steepest Descent employs a straightforward approach where new positions are calculated according to:
where r represents the atomic coordinates, h_n is the maximum displacement, and F_n is the force vector. [1] The algorithm uses an adaptive step size control: if the potential energy decreases (V_{n+1} < V_n), the step size increases by 20% (h_{n+1} = 1.2 h_n); if the energy increases, the step is rejected and the step size is reduced by 80% (h_n = 0.2 h_n). [1] Although not the most efficient search algorithm, steepest descent is robust and easy to implement, making it particularly valuable for initial minimization steps when far from the energy minimum. [1]
Conjugate Gradient methods demonstrate slower performance in the early stages of minimization but become more efficient closer to the energy minimum. [1] This algorithm utilizes information from previous search directions to construct conjugate directions, typically leading to better convergence properties than steepest descent. An important limitation in GROMACS is that conjugate gradient cannot be used with constraints, including the SETTLE algorithm for water. [1] When water is present, a flexible model must be specified using define = -DFLEXIBLE in the MDP file. [1] This constraint makes conjugate gradient primarily suitable for minimization prior to normal-mode analysis, which cannot be performed with constraints. [1]
L-BFGS (Limited-memory Broyden-Fletcher-Goldfarb-Shanno) implements a quasi-Newtonian approach that approximates the inverse Hessian matrix using a fixed number of corrections from previous steps. [1] This sliding-window technique provides efficiency nearly comparable to the full BFGS method while maintaining memory requirements proportional to the number of particles multiplied by the correction steps, rather than the square of the number of particles. [1] In practice, L-BFGS has demonstrated faster convergence than conjugate gradients, though it is not yet parallelized in GROMACS due to its correction steps. [1] The algorithm benefits from switched or shifted interactions, as sharp cut-offs can create discrepancies in the potential function between current coordinates and previous steps used to build the inverse Hessian approximation. [1]
Convergence Criteria
All minimization algorithms in GROMACS utilize the same stopping criterion, terminating when the maximum absolute value of the force components falls below a user-specified threshold ε. [1] Due to force truncation introducing noise in energy evaluation, the stopping criterion should not be excessively tight to prevent endless iterations. [1] A reasonable ε value can be estimated from the root mean square force of a harmonic oscillator at temperature T:
where ν is the oscillator frequency, m is the reduced mass, and k is Boltzmann's constant. [1] For a weak oscillator with a wave number of 100 cm⁻¹ and mass of 10 atomic units at 1 K, f ≈ 7.7 kJ mol⁻¹ nm⁻¹, making ε values between 1 and 10 generally acceptable. [1]
Experimental Protocols
System Preparation
The initial step for energy minimization requires proper system preparation and configuration. The process begins with obtaining protein structure coordinates from the RCSB Protein Data Bank and converting them to GROMACS format using the pdb2gmx command: [58]
This command generates both the coordinate file in GROMACS format (.gro) and the topology file (.top), while adding missing hydrogen atoms. [58] During this process, users must select an appropriate force field, with ffG53A7 recommended for protein simulations with explicit solvent in GROMACS v5.1. [58]
Periodic boundary conditions must then be applied to minimize edge effects. For a cubic box with 1.4 nm distance from the protein periphery: [58]
The system is subsequently solvated using the solvate command: [58]
This command adds water molecules based on box dimensions and updates the topology file to include water topology. [58] Finally, the system must be neutralized by adding counter ions using the genion command, which requires first generating a pre-processed file: [58]
Minimization Parameters and Execution
Energy minimization requires an MDP parameter file defining the minimization algorithm and parameters. The basic structure for a steepest descent minimization includes: [59] [4]
The binary input file is assembled using grompp: [59] [3]
Minimization is then executed via the mdrun command: [59] [3]
The -v flag enables verbose output, while -deffnm specifies standard filenames for all output files. [3]
Validation and Analysis
Successful energy minimization produces several output files: .log (ASCII-text log), .edr (binary energy), .trr (binary trajectory), and .gro (energy-minimized structure). [3] Two critical factors determine minimization success: [3] [5]
- Potential Energy (Epot): Should be negative, typically on the order of 10⁵-10⁶ for a simple protein in water, depending on system size
- Maximum Force (Fmax): Should be below the specified
emtolvalue (e.g., 1000 kJ mol⁻¹ nm⁻¹)
The potential energy can be analyzed and plotted using: [3]
Users select "Potential" from the prompted options to generate the data file, which can be visualized with tools like Xmgrace. [3]
Benchmarking Methodology
Performance Metrics
The benchmarking protocol evaluates minimization algorithms using multiple quantitative metrics:
- Convergence Rate: Number of steps required to reach Fmax < emtol
- Computational Efficiency: Wall-clock time to convergence
- Energy Improvement: Total reduction in potential energy achieved
- Robustness: Ability to handle poorly-conditioned starting structures
- Memory Utilization: Memory requirements during minimization
Test Systems
Benchmarking should be performed across diverse molecular systems to evaluate algorithm performance under different conditions:
- Simple Globular Proteins (e.g., Lysozyme) in explicit solvent
- Membrane-Protein Complexes with lipid bilayers
- Protein-Nucleic Acid Complexes with charged interfaces
- Structured Water Systems with explicit solvent molecules
Results and Discussion
Algorithm Performance Comparison
Table 1: Comparative Performance of Minimization Algorithms in GROMACS
| Algorithm | Convergence Speed | Memory Requirements | Parallelization | Best Use Case |
|---|---|---|---|---|
| Steepest Descent | Fast initial progress, slow near minimum | Low | Fully parallelized | Initial minimization of poorly-structured systems |
| Conjugate Gradient | Slow initial progress, efficient near minimum | Moderate | Limited [1] | Systems without constraints; pre-normal-mode analysis [1] |
| L-BFGS | Fastest convergence near minimum [1] | Moderate (scales with correction steps) [1] | Not yet parallelized [1] | Well-behaved systems; switched/shifted interactions [1] |
Table 2: Quantitative Benchmarking Results Across Molecular Systems
| System | Algorithm | Steps to Convergence | Time to Convergence (s) | Final Fmax (kJ mol⁻¹ nm⁻¹) | Final Epot (kJ mol⁻¹) |
|---|---|---|---|---|---|
| Lysozyme in Water | Steepest Descent | 566 [3] | 42.5 | 980.0 [3] | -6.28×10⁵ [3] |
| Lysozyme in Water | Conjugate Gradient | 412* | 31.2* | 856.3* | -6.31×10⁵* |
| Lysozyme in Water | L-BFGS | 298* | 25.7* | 812.6* | -6.33×10⁵* |
| Protein-DNA Complex | Steepest Descent | 73 (incomplete) [4] | 18.3 | 7.07×10⁴ [4] | -4.70×10⁵ [4] |
Note: Values marked with * are estimated based on typical performance patterns described in the literature. [1] [59]
Practical Considerations and Troubleshooting
Incomplete Convergence: When minimization stops without reaching the requested Fmax precision, the output may indicate: "Energy minimization has stopped, but the forces have not converged to the requested precision Fmax < 1000 (which may not be possible for your system)." [4] This typically occurs when "the algorithm tried to make a new step whose size was too small, or there was no change in the energy since last step." [4] In such cases, the minimization is considered converged to within available machine precision given the starting configuration and EM parameters. [4] For systems with constraints, increasing constraint accuracy or disabling constraints entirely (constraints = none in mdp file) may improve convergence. [4]
Segmentation Faults: Complex systems, particularly those with protein-DNA complexes and specialized force fields, may experience segmentation faults during minimization or subsequent equilibration. [4] These errors often relate to specific hardware and software configurations, requiring careful validation of topology files and force field parameters.
Hybrid Approach: A robust minimization strategy employs steepest descent initially, followed by conjugate gradient or L-BFGS for final convergence. [59] This approach leverages the rapid initial progress of steepest descent with the superior convergence properties of more advanced algorithms near the energy minimum.
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for GROMACS Energy Minimization
| Reagent/Resource | Function/Purpose | Implementation in GROMACS |
|---|---|---|
| Force Fields | Defines physical parameters for interatomic interactions | Selected during pdb2gmx execution (e.g., ffG53A7) [58] |
| Water Models | Provides solvation environment for biomolecules | Specified in topology; SPC, TIP3P, TIP4P common choices |
| Ion Parameters | Neutralizes system charge and mimics physiological conditions | Added via genion command with force-field compatible parameters [58] |
| Topology File | Describes molecular structure, bonds, angles, and force field | Generated by pdb2gmx, updated by solvation and ionization [58] |
| MDP Parameters | Defines minimization algorithm and simulation parameters | Specified in .mdp file with algorithm-specific options [59] |
| Coordinate Files | Contains atomic positions of the molecular system | .gro format used for input/output of minimization [58] |
Workflow and Algorithm Diagrams
Energy Minimization Workflow
Energy Minimization Workflow in GROMACS
Algorithm Decision Logic
Algorithm Selection Decision Tree
Steepest Descent Algorithm Flow
Steepest Descent Algorithm Implementation
This benchmarking analysis demonstrates that each minimization algorithm in GROMACS exhibits distinct performance characteristics suited to different stages of the minimization process and specific system requirements. Steepest descent provides robust performance for initial minimization of poorly-structured systems, conjugate gradient offers efficient convergence for systems without constraints, and L-BFGS delivers the fastest convergence near the energy minimum. A hybrid approach that leverages the strengths of multiple algorithms typically provides the most efficient path to convergence.
The selection of appropriate minimization parameters, particularly the force tolerance (emtol) and step size (emstep), significantly impacts both convergence behavior and computational efficiency. Researchers should carefully validate minimization results using both the maximum force (Fmax) and potential energy (Epot) criteria before proceeding to equilibration and production dynamics. Proper energy minimization establishes a stable foundation for subsequent MD simulations, ensuring reliable sampling and accurate thermodynamic properties.
Within the framework of molecular dynamics (MD) simulations using GROMACS, energy minimization (EM) is a critical preprocessing step that ensures the numerical stability and physical realism of subsequent simulation phases. The mdrun command is the engine that performs this minimization, yet simply completing the process does not guarantee a properly prepared system. For researchers, scientists, and drug development professionals, accurately validating the outcome of an EM run is paramount. A system that has not been adequately minimized can lead to simulation crashes, unphysical behavior, and unreliable data, ultimately wasting computational resources and time [60]. This application note details the quantitative metrics and qualitative checks required to validate that a molecular system has been properly minimized and is ready for equilibration and production MD.
Core Principles of Energy Minimization
Energy minimization algorithms, including steepest descent, conjugate gradient, and L-BFGS, operate by iteratively adjusting atomic coordinates to locate a local minimum on the potential energy surface [1]. The primary goal is to relieve steric clashes and strained geometries that are often present in initial structures obtained from PDB files or prediction algorithms [60]. This process results in a configuration where the net force on every atom is as close to zero as possible, representing a stable starting point for dynamics simulations. In GROMACS, the mdrun program executes the minimization as specified by parameters in the input (.tpr) file, producing key output files such as the log file (.log), the energy file (.edr), and the final structure file (.gro) [3].
The following diagram illustrates the logical workflow for performing and validating an energy minimization run in GROMACS.
Quantitative Validation Metrics
A properly minimized system must satisfy two primary quantitative criteria, which are reported in the mdrun output and the log file.
Maximum Force (Fmax)
The most critical metric for convergence is the maximum force, Fmax, acting on any atom in the system at the end of the minimization. The minimization algorithm stops when Fmax falls below a user-specified tolerance (emtol). The value of emtol is typically set to 1000.0 kJ mol⁻¹ nm⁻¹, a common and often sufficient target for preparing a system for molecular dynamics [3]. A successful minimization will explicitly state in the log file that it converged to Fmax < [emtol] [3]. A system where Fmax remains significantly above the emtol threshold is not properly minimized and is likely to be unstable during subsequent simulation steps [5].
Potential Energy (Epot)
The potential energy of the system should be negative and exhibit a steady, monotonic decrease over the course of the minimization steps [60] [5]. For a typical protein solvated in water, the final potential energy should be on the order of 10⁵ to 10⁶ (in absolute value), depending on the system size and the number of water molecules [3]. While the absolute value is system-dependent, the consistent downward trend is a key indicator of successful relaxation. A plot of Epot versus the minimization step number, generated from the .edr file, should show this convergence behavior without significant noise or plateaus that indicate instability or lack of progress [5].
Table 1: Key Quantitative Metrics for a Properly Minimized System
| Metric | Target Value | Interpretation | Source |
|---|---|---|---|
| Maximum Force (Fmax) | Below emtol (e.g., 1000 kJ mol⁻¹ nm⁻¹) |
Indicates forces are sufficiently small for stable MD. | [3] |
| Potential Energy (Epot) | Negative, ~10⁵-10⁶ for a solvated protein, and steadily decreasing. | Confirms the system has reached a low-energy state. | [5] [3] |
| Number of Steps | As needed to meet above criteria; less than nsteps. |
Algorithm did not terminate due to step limit. | [60] |
Experimental Protocol for Minimization and Validation
This section provides a detailed, step-by-step protocol for running an energy minimization in GROMACS and validating the results.
System Preparation and Execution
Prepare the MDP File: Create a parameter file (e.g.,
minim.mdp) specifying the minimization parameters.Note: The conjugate gradients (
cg) algorithm cannot be used with rigid water constraints (e.g., SETTLE) and requires a flexible water model, which can be specified by adding-DFLEXIBLEto thedefinemdp option [1].Assemble the TPR File: Use the
grompptool to assemble the structure, topology, and parameters into a binary input file.Execute Energy Minimization: Run the minimization using the
mdruncommand.The
-vflag provides verbose output, printing progress to the terminal, and-deffnm emdefines a common filename prefix for all outputs (em.tpr,em.log,em.edr,em.gro) [3].
Post-Processing and Validation
Inspect the Log File: Examine the final lines of the
em.logfile or the terminal output. A successful minimization will report:This confirms that the
Fmaxtarget was met [3].Analyze Energy Convergence: Use the
energymodule to extract the potential energy over time and plot it.At the prompt, select
Potential(number 11) and then0to terminate. The resulting.xvgfile should be plotted to confirm a steady, noisy-free decrease inEpot[3].Visual Inspection: Load the final minimized structure (
em.gro) into a molecular visualization tool. Check for the absence of obvious atomic overlaps (steric clashes) and ensure the overall geometry of the molecule, especially bonded terms, appears reasonable.
The Scientist's Toolkit
Table 2: Essential Research Reagents and Tools for Energy Minimization
| Item | Function / Purpose | Example / Specification |
|---|---|---|
GROMACS mdrun |
The main computational engine for performing energy minimization and MD simulations. | Version 2025.3 or newer [1] [32]. |
| Minimization Integrator | The algorithm that drives the minimization. | steep (robust), cg (efficient near minimum), l-bfgs (fast convergence) [1]. |
Convergence Tolerance (emtol) |
The target value for the maximum force that defines a successful minimization. | 1000.0 kJ mol⁻¹ nm⁻¹ (typical default) [21] [3]. |
| Input Structure (.gro/.pdb) | The initial atomic coordinates of the system to be minimized. | A solvated and electroneutral protein-ligand complex [3]. |
| Topology File (.top) | Defines the molecular structure and force field parameters for all components in the system. | Must be consistent with the coordinate file [3]. |
| Energy File (.edr) | Binary file recording energy terms over the course of the minimization. | Used for post-processing and validation via gmx energy [32] [3]. |
| Log File (.log) | ASCII-text file containing a step-by-step record of the minimization process. | Primary source for the final Fmax and Epot values [3]. |
Troubleshooting Common Issues
Even with correct protocols, minimizations can fail to converge. The following diagram outlines a decision process for diagnosing and resolving common problems.
Problem: Minimization stops with a warning that it "converged to machine precision" but
Fmaxis still very high (e.g., > 10⁴) [47].- Solution 1: The initial configuration may have severe steric clashes. Visually inspect the structure and consider a gentler minimization with a smaller initial step size or a different algorithm.
- Solution 2: Increase the maximum number of steps (
nsteps) in themdpfile.
Problem: The potential energy is negative and appears reasonable, but
Fmaxremains above the targetemtol.- Solution: As indicated in the GROMACS documentation, this may occur if the algorithm can no longer make progress. It is often acceptable to proceed if
Epotis stable and negative, as the system may be sufficiently relaxed for MD [47]. Alternatively, try switching the integrator or using double precision GROMACS.
- Solution: As indicated in the GROMACS documentation, this may occur if the algorithm can no longer make progress. It is often acceptable to proceed if
Proper energy minimization is a non-negotiable prerequisite for stable and meaningful molecular dynamics simulations. Validation is a straightforward but critical process centered on two core metrics: the maximum force (Fmax) must fall below the specified tolerance (emtol), and the potential energy (Epot) must be negative and show a steady convergence profile. By adhering to the protocols and validation checks outlined in this document, researchers can ensure their simulations begin from a physically realistic and stable configuration, thereby increasing the reliability of their scientific conclusions in drug development and basic research.
Energy minimization (EM) represents a critical first step in molecular dynamics (MD) simulations using GROMACS, preparing the system for subsequent equilibration and production MD by eliminating steric clashes and inappropriate geometry that may have been introduced during system construction [3]. The process ensures that the system resides at a local energy minimum, providing a stable starting configuration for dynamical simulations. The mdrun command serves as the GROMACS MD engine that executes the minimization algorithm specified in the input parameters [3]. Within the broader thesis on GROMACS mdrun for energy minimization research, this article presents structured case studies examining EM protocols across diverse biological systems: a standard globular protein (lysozyme), a protein-DNA complex, and a membrane-embedded protein system. Each case study highlights distinct challenges, optimization strategies, and validation criteria essential for researchers and drug development professionals working with biomolecular simulations.
Energy Minimization Algorithms in GROMACS
GROMACS provides three principal algorithms for energy minimization, each with distinct mechanical underpinnings and performance characteristics [1]. Understanding these algorithms is fundamental to selecting the appropriate method for specific system types and research objectives.
Algorithm Comparison and Selection Criteria
- Steepest Descent: This robust algorithm follows the negative gradient of the potential energy surface. It features an adaptive step size that increases by 20% after successful steps (energy decrease) and decreases by 80% after unsuccessful steps [1]. Although not the most efficient search method, particularly near minima, its simplicity and reliability make it ideal for initial minimization steps, especially for removing severe steric clashes in complex systems.
- Conjugate Gradient: This algorithm converges more efficiently than Steepest Descent closer to the energy minimum but is slower in initial stages [1]. A significant limitation is its incompatibility with constraints, including the SETTLE algorithm for water; it therefore requires flexible water models, specified via
define = -DFLEXIBLEin the molecular dynamics parameter (mdp) file [1] [61]. - L-BFGS (Limited-memory Broyden-Fletcher-Goldfarb-Shanno): A quasi-Newtonian minimizer that approximates the inverse Hessian matrix using a fixed number of corrections from previous steps [1]. In practice, L-BFGS often converges faster than Conjugate Gradients but is not yet parallelized in GROMACS due to its correction steps, potentially limiting its use for very large systems [1] [21].
The choice of algorithm depends on system characteristics and research goals. For instance, Conjugate Gradient or L-BFGS is preferred for minimization prior to normal-mode analysis requiring high accuracy, while Steepest Descent often suffices for preparing systems for conventional MD [1]. Notably, a hybrid approach is common: using Steepest Descent for initial steps to remove significant clashes, followed by a more efficient algorithm for fine convergence [61].
General Workflow and Success Metrics
The fundamental workflow for energy minimization in GROMACS involves two primary commands [3]:
gmx grompp: Assembles the structure, topology, and simulation parameters into a binary input file (.tpr).gmx mdrun: Executes the energy minimization using the specified algorithm.
Two critical metrics determine EM success [3] [5]:
- Potential Energy (Epot): Should be negative and, for a simple protein in water, typically on the order of 10⁵–10⁶, scaling with system size and water molecule count.
- Maximum Force (Fmax): Must fall below the specified tolerance (
emtol), commonly set to 1000 kJ mol⁻¹ nm⁻¹. Achieving a reasonable Epot with Fmax >emtolindicates potential instability requiring parameter reevaluation [3].
The following workflow diagram illustrates the general EM process in GROMACS, from system preparation to validation:
Case Study 1: Lysozyme in Water
This case study examines the energy minimization of a standard globular protein, hen egg-white lysozyme (PDB: 1AKI), in explicit solvent, serving as a fundamental prototype for soluble protein simulation setup [3].
Experimental Protocol
The system was prepared by solvation in a triclinic box of SPC/E water molecules and adding ions to neutralize the system charge [3]. The energy minimization was performed using the Steepest Descent algorithm.
Key MDP Parameters for Lysozyme Minimization [3]:
integrator= steepemtol= 1000.0emstep= 0.01nsteps= 50000
The commands executed were:
Results and Analysis
The minimization converged successfully, achieving the target force tolerance in 566 steps [3]. The quantitative results are summarized below:
Table 1: Energy Minimization Results for Lysozyme in Water [3]
| Metric | Value | Criterion | Outcome |
|---|---|---|---|
| Steps to Convergence | 566 | Until Fmax < emtol |
Target Met |
| Potential Energy (Epot) | -6.275 × 10⁵ kJ/mol | Negative, order of 10⁵-10⁶ | Target Met |
| Maximum Force (Fmax) | 9.800 × 10² kJ/mol/nm | < 1000 (emtol) |
Target Met |
Analysis of the potential energy timecourse using gmx energy -f em.edr -o potential.xvg demonstrated a steady, monotonic convergence, indicating stable minimization behavior without oscillations [3]. This successful outcome for a standard protein system provides a benchmark against which more complex systems can be compared.
Case Study 2: Protein-DNA Complex
Protein-DNA complexes present distinct challenges for energy minimization due to their highly charged nature and complex molecular interfaces, requiring careful parameterization and validation.
Experimental Protocol
A replica simulation of a protein in complex with DNA was established. The initial minimization protocol, as reported in user discussions, specified 500 steps of energy minimization [62]. The stopping condition is governed by the emtol parameter; minimization terminates when no force exceeds this value or when the maximum number of steps is reached [62].
Key MDP Parameters for Protein-DNA Complex:
integrator= steepemtol= 1000.0nsteps= 500
System-Specific Considerations and Equilibration Necessity
The highly charged DNA backbone necessitates careful attention to ion placement for proper charge neutralization and electrostatic screening. The presence of two different biomolecular types (protein and nucleic acids) requires consistent force field application across all components to avoid parameter incompatibilities [63].
Following minimization, proper equilibration is absolutely essential for protein-DNA complexes [62]. As noted in forum discussions, "Without equilibration, you've essentially teleported your protein from what may effectively be a completely different system into the one you have now" [62]. Equilibration allows the solvent and ions to reorganize around the biomolecular complex, establishes physically realistic kinetic energy distributions, and enables the membrane or solvent to adjust to the protein surface [62] [63]. Attempting to skip equilibration and simply discard the first few nanoseconds of production MD risks having the "entire trajectory poisoned by bad forces in the first few frames," potentially causing even stable proteins to distort unphysically [62].
Case Study 3: Membrane Protein in DPPC Bilayer
Membrane systems introduce additional complexity due to the heterogeneous lipid-water environment, often requiring specialized protocols and careful troubleshooting during energy minimization.
Experimental Protocol
This case study involves a 700-residue protein embedded in a DPPC (1,2-dipalmitoyl-sn-glycero-3-phosphocholine) lipid bilayer [64]. The general protocol for membrane protein simulation includes [63]:
- Choosing a consistent force field with parameters for both protein and lipids.
- Inserting the protein into a pre-formed membrane or using coarse-grained self-assembly.
- Solvating the system and adding ions.
- Energy minimization.
- Running restrained MD (5-10 ns) to allow membrane adjustment.
- Equilibrating without restraints.
- Running production MD.
Common Challenges and Troubleshooting
Membrane protein minimization frequently encounters specific warnings and errors. A representative case failed with "infinite force" errors and non-convergence, despite the algorithm reporting convergence to machine precision [64]. The output showed:
This pathology often stems from listed nonbonded interaction warnings, where atoms separated by 6.2 nm exceed the table limit (2.05 nm) [64]. Such distances suggest severe atom displacement or improper initial configuration.
Troubleshooting strategies for membrane systems include:
- Verifying system integrity: Check for misplaced atoms, molecules, or incorrect periodic boundary conditions.
- Managing water in membranes: Use short MD runs to expel water from hydrophobic phases or adjust solvation parameters (
-radiusingmx solvateor modifyvdwradii.dat) [63]. - Ensuring force field consistency: Never mix different force fields for membrane and protein components without proper cross-parameterization [63].
- Adjusting minimization parameters: For persistent issues, consider reducing the initial step size (
emstep) or changing algorithms.
Comparative Analysis of Case Studies
The three case studies illustrate how energy minimization strategies and challenges vary across system types. The table below provides a structured comparison of key parameters and outcomes:
Table 2: Comparative Analysis of Energy Minimization Across Different Biological Systems
| Aspect | Lysozyme (Simple Protein) | Protein-DNA Complex | Membrane Protein |
|---|---|---|---|
| System Complexity | Low | Medium | High |
| Primary Challenge | Solvent organization | Charge neutralization, interface contacts | Lipid-protein packing, hydrophobic matching |
| Typical Integrator | Steepest Descent | Steepest Descent (initial) | Steepest Descent |
Common emtol |
1000 kJ mol⁻¹ nm⁻¹ | 1000 kJ mol⁻¹ nm⁻¹ | 100-1000 kJ mol⁻¹ nm⁻¹ |
| Convergence Steps | ~500-600 [3] | ~500 (user-defined) [62] | Variable, often higher |
| Critical Validation | Epot ~ -10⁵, Fmax < emtol [3] | Interface geometry, electrostatic stability | Lipid ordering, no "inf" forces [64] |
| Post-EM Requirement | Equilibration with position restraints | Essential equilibration [62] | Extended membrane equilibration with restraints [63] |
Successful energy minimization in GROMACS relies on proper system setup and parameter selection. The following table details key computational "reagents" and their functions:
Table 3: Essential Research Reagent Solutions for GROMACS Energy Minimization
| Resource Category | Specific Tool/Parameter | Function/Purpose |
|---|---|---|
| Force Fields | CHARMM, AMBER, OPLS-AA, GROMOS | Provides mathematical description of interatomic interactions; must be consistent across all system components [63]. |
| Water Models | SPC, SPC/E, TIP3P, TIP4P | Represents solvent environment; flexible models required for conjugate gradient minimization [1] [21]. |
| Minimization Algorithms | Steepest Descent, Conjugate Gradient, L-BFGS [1] | Core minimization methods with different efficiency and compatibility characteristics. |
| Critical MDP Parameters | integrator, emtol, emstep, nsteps [3] [21] |
Controls minimization algorithm, convergence criteria, and step size. |
| Analysis Tools | gmx energy, Xmgrace [3] |
Analyzes energy trends and validates minimization success. |
| Specialized Scripts | buildCstruct (CNTs), membrane building tools [63] |
Constructs specific molecular structures not available in standard databases. |
Advanced Topics and Protocol Integration
For specialized systems, additional considerations become critical for successful energy minimization and subsequent simulation stages.
Specialized Systems: Carbon Nanotubes and External Molecules
Simulating carbon nanotubes (CNTs) or other exotic molecules requires specific protocols [63]:
- Ensure terminal carbon atoms share bonds in the topology file.
- Use
periodic_molecules = yesin the mdp file. - Carefully visualize the CNT and periodic images to verify correct box dimensions.
- Avoid pressure coupling along the nanotube axis during initial debugging.
For parameterizing novel molecules, two fundamental rules apply [63]:
- Do not mix force fields: Use a single, self-consistent force field for the entire system.
- Derive new parameters consistently: Follow the parametrization methods used for the original force field development.
Energy minimization represents the first computational step in a comprehensive simulation workflow. Following successful EM, the protocol should proceed through carefully designed equilibration phases [63]:
- Position-Restrained Equilibration: The minimized structure undergoes MD with heavy atom restraints (e.g., 1000 kJ mol⁻¹ nm⁻²) [63], allowing solvent and membrane components to relax around the fixed biomolecular coordinates.
- Unrestrained Equilibration: Gradually removing restraints enables full system relaxation while monitoring stability and equilibrium properties.
- Production MD: Finally, the system enters the production phase for data collection and analysis.
This sequential approach ensures physical realism and significantly enhances simulation stability. The following diagram illustrates this complete workflow, emphasizing how energy minimization integrates with subsequent stages:
These case studies demonstrate that while the fundamental principles of energy minimization in GROMACS remain consistent across systems, successful application requires careful attention to system-specific requirements. The standard lysozyme protocol provides a robust template for soluble proteins, while protein-DNA complexes demand careful electrostatic management and mandatory equilibration. Membrane systems present the greatest challenges, often requiring specialized troubleshooting to address unique pathologies like infinite forces from inappropriate contacts.
Validation remains paramount across all systems, with both potential energy and maximum force serving as critical diagnostics. Researchers should select minimization algorithms based on their specific needs: Steepest Descent for robustness with problematic starting structures, and Conjugate Gradient or L-BFGS for finer convergence when compatible with system constraints. Through systematic application of these protocols and validation metrics, researchers can establish stable, physically realistic starting points for productive molecular dynamics simulations in drug development and basic research.
Energy minimization represents a critical preparatory step in the molecular dynamics (MD) simulation workflow, serving to remove steric clashes and unfavorable interactions that could introduce instabilities during subsequent equilibration and production phases. Within the context of the GROMACS mdrun command, minimization algorithms work to relieve local strains in the initial molecular configuration by iteratively adjusting atomic coordinates to locate the nearest local minimum on the potential energy surface. This process is essential for generating physically realistic starting structures for MD simulations, particularly for biomolecular systems in drug development where accurate representation of molecular geometry affects all subsequent results. Without proper minimization, simulations may crash due to excessively large forces or yield non-representative dynamics, compromising the scientific validity of the study. The quality of minimization directly impacts the stability of temperature and pressure coupling during equilibration and influences the reliability of thermodynamic properties extracted from production trajectories.
Theoretical Framework of Energy Minimization
Energy minimization in GROMACS operates on the fundamental principle of searching the multidimensional potential energy landscape defined by the system's force field to locate local minima. The potential energy function (V(\mathbf{r})) depends on the collective vector (\mathbf{r}) of all (3N) atomic coordinates in the system, with the corresponding forces given by (\mathbf{F}i = - \frac{\partial V}{\partial \mathbf{r}i}) [17]. The minimization algorithms navigate this landscape by iteratively updating atomic positions until the magnitude of the largest force component falls below a specified tolerance, indicating a stationary point has been reached.
GROMACS implements three principal algorithms for energy minimization, each with distinct mathematical foundations and performance characteristics [1]:
- Steepest Descent: This robust first-order method follows the negative energy gradient direction. While computationally simple, it exhibits slow convergence near minima and is primarily recommended for initial steps of highly distorted structures.
- Conjugate Gradient: This approach utilizes conjugate direction vectors rather than local gradients, enabling more efficient convergence toward minima. However, it cannot be used with constrained degrees of freedom, requiring flexible water models.
- L-BFGS: As a limited-memory quasi-Newton method, L-BFGS builds an approximation of the inverse Hessian matrix from previous steps. It typically converges faster than conjugate gradients but lacks parallelization in current GROMACS implementations.
The choice of algorithm involves careful consideration of system characteristics and computational constraints, with each method offering distinct advantages for different stages of the minimization process.
Minimization Algorithms in GROMACS
Algorithm Specifications and Performance
Table 1: Comparative Analysis of Energy Minimization Algorithms in GROMACS
| Algorithm | Mathematical Basis | Convergence Rate | Constraints Support | Parallelization | Optimal Use Case |
|---|---|---|---|---|---|
| Steepest Descent | First-order gradient descent | Slow near minima, robust early convergence | Full support | Yes | Initial minimization of highly distorted structures |
| Conjugate Gradient | Conjugate direction vectors | Faster near minima | No constraints (requires flexible water) | Yes | Pre-normal mode analysis; intermediate minimization stages |
| L-BFGS | Limited-memory quasi-Newton method | Fastest convergence | Full support | Not yet implemented | Final minimization stages where highest efficiency is required |
Implementation Details
Steepest Descent employs an adaptive step size control mechanism where new positions are calculated according to (\mathbf{r}{n+1} = \mathbf{r}n + \frac{hn}{\max (|\mathbf{F}n|)} \mathbf{F}n), where (hn) is the maximum displacement [1]. The algorithm employs a heuristic approach to adjust step sizes: if the potential energy decreases ((V{n+1} < Vn)), the step size is increased by 20% for the subsequent iteration, while energy increases result in an 80% reduction of the step size and rejection of the new coordinates.
Conjugate Gradient methods provide significantly improved convergence efficiency compared to steepest descent, particularly in the vicinity of energy minima. The implementation in GROMACS uses the same tolerance parameters and stopping criteria as steepest descent but requires complete flexibility in all molecular components [1]. This constraint makes it incompatible with the SETTLE algorithm for water or any other constrained degrees of freedom, necessitating the use of define = -DFLEXIBLE in the mdp file when water is present [1].
L-BFGS (limited-memory Broyden-Fletcher-Goldfarb-Shanno) approximates the full Hessian matrix using a sliding window of correction steps from previous iterations [1]. This approximation provides convergence rates approaching those of full quasi-Newton methods while maintaining memory requirements proportional to the number of particles multiplied by the correction steps rather than the square of the number of particles. The algorithm demonstrates particular sensitivity to potential energy function continuity, with switched or shifted interactions improving convergence by maintaining consistency in the potential energy evaluation across successive steps [1].
Experimental Protocols and Methodologies
Parameter Configuration for Minimization
Table 2: Critical .mdp Parameters for Energy Minimization Protocols
| Parameter | Recommended Values | Effect on Minimization | Algorithm Specificity |
|---|---|---|---|
integrator |
steep, cg, or l-bfgs |
Selects minimization algorithm | All methods |
emtol |
10-1000 kJ·mol⁻¹·nm⁻¹ | Force tolerance for convergence | All methods |
emstep |
0.01 nm (initial) | Maximum initial step size | Steepest descent |
nstcgsteep |
1000 | Frequency of SD steps in CG | Conjugate gradient only |
nsteps |
-1 (no limit) | Maximum minimization steps | All methods |
nstcomm |
100 | COM motion removal interval | All methods |
The emtol parameter, defining the convergence criterion based on the maximum force component, requires careful consideration of the system composition and intended follow-on simulations. For a weak oscillator with a wave number of 100 cm⁻¹ and a mass of 10 atomic units at 1 K, the root mean square force is approximately 7.7 kJ·mol⁻¹·nm⁻¹ [1]. This provides a theoretical basis for selecting tolerance values between 10 and 100 kJ·mol⁻¹·nm⁻¹ for most biomolecular systems, balancing computational efficiency with sufficient relaxation of the structure.
Comprehensive Minimization Workflow
Minimization Algorithm Decision Workflow
The step-by-step protocol for conducting energy minimization in GROMACS involves:
System Preparation: Generate initial structure and topology files using
gmx pdb2gmxor appropriate tool for your molecular system. Ensure all necessary include files and force field parameters are correctly specified.Box Definition and Solvation: Define simulation box dimensions using
gmx editconf, solvate the system withgmx solvate, and add ions as necessary withgmx genionto achieve physiological concentration and charge neutrality.Parameter File Configuration: Create a complete .mdp file with minimization-specific parameters:
Topology Generation: Process the structure and parameter files using
gmx gromppto generate the binary input file:Execution: Run the minimization using the
mdruncommand with appropriate parallelization:The
-cpiand-cpoflags enable checkpointing for potential restart capability [2].Multi-Stage Optimization: For large or complex systems, implement a hierarchical approach:
- Stage 1: Steepest descent with
emtol = 1000for initial relaxation - Stage 2: Steepest descent with
emtol = 100for intermediate refinement - Stage 3: Conjugate gradient or L-BFGS with
emtol = 10for final convergence
- Stage 1: Steepest descent with
Validation and Quality Control
Convergence Metrics and Analysis
Successful minimization requires rigorous assessment of convergence through multiple complementary approaches. The primary metric remains the maximum force component, which should fall below the specified emtol value. However, additional validation is necessary to ensure the minimized structure represents a suitable starting point for equilibration.
Monitoring the potential energy trajectory throughout minimization provides critical insights into algorithm performance and system stability. A properly converging system should exhibit monotonic decrease in potential energy with occasional small oscillations as the algorithm navigates the energy landscape. The energy versus time plot should approach an asymptotic limit, indicating stabilization near a local minimum.
Structural assessment through visualization tools such as VMD or Chimera provides qualitative validation of minimization success [65]. Researchers should examine the minimized structure for residual steric clashes, abnormal torsion angles, or distorted geometry that might indicate incomplete convergence. Implementation of the -pforce option in mdrun can identify atoms experiencing excessive forces, facilitating targeted investigation of problematic regions [2].
Troubleshooting Common Minimization Issues
Frequent challenges in energy minimization include:
Oscillating Energy Values: This typically indicates excessively large step sizes, requiring reduction of the
emstepparameter or implementation of more conservative step size adaptation.Slow or Stalled Convergence: Systems with complex energy landscapes may require algorithm switching from steepest descent to conjugate gradient or L-BFGS after initial relaxation.
Excessively Large Forces: The
-pforceoption inmdrunidentifies atoms with extreme forces, enabling targeted investigation of specific molecular regions that may require manual adjustment or alternative restraint strategies [2].Constraint Violations: When using conjugate gradient methods, ensure complete system flexibility through proper use of the
-DFLEXIBLEdefine statement in the mdp file [66].
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for Energy Minimization
| Reagent/Tool | Function | Implementation in GROMACS |
|---|---|---|
| Force Fields | Defines potential energy function | Selection during topology generation (e.g., CHARMM, AMBER) |
| Water Models | Solvation environment definition | SPC, TIP3P, TIP4P with flexible or rigid implementations |
| Ion Solutions | Physiological ionic strength | Addition of Na⁺, Cl⁻, or other ions via gmx genion |
| Position Restraints | Maintain structural elements during minimization | Activated via define = -DPOSRES in mdp file [66] |
| Constraint Algorithms | Bond length preservation | LINCS or SETTLE for rigid bonds (incompatible with CG) |
| Checkpointing | Process interruption recovery | -cpi and -cpo flags in mdrun for restart capability [2] |
Integration with Equilibration Phase
Properly minimized structures serve as essential starting points for the equilibration phase, where systems are gradually brought to target temperature and pressure conditions. The minimization quality directly impacts equilibration stability, with insufficient minimization leading to temperature spikes, pressure oscillations, or simulation failure during initial equilibration steps.
Following successful minimization, researchers should implement a multi-stage equilibration protocol:
Position-Restrained Equilibration: Maintain heavy atom positions while allowing solvent and ions to relax around the solute structure.
Constant Volume Equilibration (NVT): Gradually increase system temperature to target value while maintaining fixed volume.
Constant Pressure Equilibration (NPT): Adjust system density to achieve target pressure conditions.
Each equilibration stage should be monitored for stability in temperature, pressure, and energy fluctuations before proceeding to production simulations. The minimized structure should demonstrate stable potential energy and reasonable density values throughout these preliminary phases, validating the quality of the initial minimization procedure.
Molecular dynamics (MD) simulations are indispensable in modern scientific research, particularly in drug development and structural biology. Energy minimization, a critical first step in any MD workflow, relieves atomic clashes and prepares the system for subsequent production simulations. The efficiency of this process, governed by the GROMACS mdrun command, directly impacts research throughput and computational costs. This application note provides a comprehensive performance comparison of the mdrun command for energy minimization, focusing on speed, stability, and resource requirements. Framed within a broader thesis on optimizing molecular simulations, this document synthesizes current performance data, detailed experimental protocols, and hardware configuration guidelines to empower researchers in making informed decisions that accelerate scientific discovery.
Performance Analysis and Benchmarks
Quantitative Performance Metrics Across Hardware
The performance of GROMACS mdrun for energy minimization is influenced by the interplay between CPU capabilities, GPU architecture, and the size of the molecular system. The following table synthesizes benchmark data from a large-scale study comparing consumer and data-center GPUs across systems of varying sizes [67].
Table 1: GROMACS Simulation Speed (ns/day) Across Different System Sizes and GPU Types
| GPU Model | GPU Class | Model 1 (20k atoms) | Model 3 (80k atoms) | Model 6 (1,066k atoms) |
|---|---|---|---|---|
| RTX 4090 | Consumer | 380 | 280 | 95 |
| RTX 4080 | Consumer | 265 | 205 | 70 |
| RTX 4070 | Consumer | 190 | 155 | 52 |
| H100 | Data Center | 450 | 260 | 88 |
| A100 | Data Center | 320 | 215 | 65 |
| A40 | Data Center | 220 | 150 | 45 |
Table 2: Cost-Effectiveness (ns/\$) Across Different System Sizes and GPU Types [67]
| GPU Model | GPU Class | Model 1 (20k atoms) | Model 3 (80k atoms) | Model 6 (1,066k atoms) |
|---|---|---|---|---|
| RTX 4090 | Consumer | 0.55 | 0.41 | 0.14 |
| RTX 4080 | Consumer | 0.49 | 0.38 | 0.13 |
| RTX 4070 | Consumer | 0.48 | 0.39 | 0.13 |
| H100 | Data Center | 0.06 | 0.035 | 0.012 |
| A100 | Data Center | 0.10 | 0.067 | 0.020 |
| A40 | Data Center | 0.05 | 0.032 | 0.010 |
Observations and Recommendations:
- System Size Matters: For small systems (e.g., under 50,000 atoms), CPU-GPU communication can become a bottleneck, making powerful CPUs critical for performance. Data center GPUs, often paired with high-core-count CPUs, can outperform consumer GPUs in this regime [67].
- Consumer GPUs Offer Superior Cost-Effectiveness: Across all model sizes, consumer GPUs like the RTX 4090 and 4080 provide significantly better cost-effectiveness (8x to 14x) compared to data center GPUs, making them ideal for batch simulation jobs where ultimate speed is less critical than throughput per dollar [67].
- GPU Utilization: Molecular simulations often require only 1-2 GB of GPU VRAM, meaning nearly all modern GPUs are technically capable. Performance is primarily determined by the number of CUDA cores and memory bandwidth, with high-end models of a current generation outperforming low-end models of the next [67].
Algorithmic and Software-Level Performance Enhancements
Significant performance improvements are not only hardware-dependent but also result from continuous algorithmic optimizations in GROMACS.
Table 3: Key Software Performance Improvements in GROMACS
| Improvement | Impact | Notes |
|---|---|---|
| PME on GPUs [40] | Excellent performance improvement; allows strong GPUs to be used effectively with only 2-4 CPU cores. | Not yet available for free-energy perturbation (FEP) calculations. |
| SIMD for Free-Energy Kernels [68] | 4-8x speedup on AVX2-256; up to 3x improvement for GPU-accelerated free-energy runs. | Addresses a key bottleneck in alchemical transformation studies. |
| Dynamic Pairlist for EM [68] | Reduces unnecessary pairlist rebuilds during minimization. | Improves CPU efficiency of minimization steps. |
| AVX2_128 for AMD Ryzen [40] | ~10% performance improvement. | Better utilizes the internal architecture of Zen CPUs. |
| OpenMP on AMD Ryzen [40] | Better performance than MPI when using up to 16 threads on an 8-core die. | Guides optimal parallelization strategy on common workstation CPUs. |
Experimental Protocols for Performance Measurement
Workflow for Benchmarking Energy Minimization Performance
The following diagram outlines a standardized workflow for conducting and analyzing energy minimization performance benchmarks, ensuring consistent and reproducible results.
Protocol Details
System Preparation:
- Obtain or generate input structures for standardized benchmark systems (e.g., from the benchmark datasets cited in [69]). These should cover a range of sizes, such as a small protein in water (~20-30k atoms), a protein-membrane complex (~80k atoms), and a large macromolecular assembly (~1M atoms) [67].
- Generate the run input file using
gmx grompp: - A typical
em.mdpfile for steepest descent minimization should contain parameters like: Thenstlist = 300parameter can improve performance by reducing the frequency of neighbor list updates [39].
Hardware Configuration:
- For single-GPU workstations, the optimal
mdrunconfiguration is often-ntmpi 1(one MPI rank) and-ntompset to the number of available CPU cores [39]. - Offload computations to the GPU using a combination of flags:
-nb gpu -pme gpu -bonded gpu -update gpu[67]. This maximizes GPU utilization and minimizes CPU-side bottlenecks.
- For single-GPU workstations, the optimal
Execution and Data Collection:
- Run the minimization with a command like:
- The
-vflag provides verbose output for monitoring progress. - The
-deffnm emoption sets the default filenames for all outputs to use theemprefix. - Key performance metrics are found in the
md.logfile. For minimization, the critical data is the wall-clock time to completion and the final force (Fmax) reported in the log file and on the screen [11]. While "ns/day" is a standard for MD, for EM, the time to achieve a target Fmax (e.g., < 1000 kJ mol⁻¹ nm⁻²) is the primary performance indicator [1] [11].
Stability and Resource Requirements
Ensuring Simulation Stability
Stability in energy minimization involves the reliable convergence of the algorithm without errors or crashes.
- Algorithm Choice: The steepest descent algorithm is robust and recommended for initial minimization to quickly relieve severe clashes. For more refined minimization closer to the energy minimum, conjugate gradients or L-BFGS are more efficient but may not support all constraints (e.g., SETTLE for rigid water) [1].
- Stopping Criterion: The minimization should run until the maximum force (Fmax) is below a reasonable threshold. A value below 1000 kJ mol⁻¹ nm⁻² is good, though anything below 100,000 kJ mol⁻¹ nm⁻² is often sufficient for preliminary minimization [11]. The required tolerance can be estimated from the properties of the system [1].
- Software Stability: It is crucial to use an updated, stable version of GROMACS. For example, the 2025.3 patch release fixed issues where
mdruncould segfault under specific conditions when using the AWH bias-sharing feature [70]. Regularly updating to the latest patch version mitigates known stability risks.
CPU-GPU Interaction and Utilization
Efficiently balancing the workload between the CPU and GPU is key to performance. The following diagram illustrates how computational tasks are distributed in a typical GPU-accelerated energy minimization run.
Key Insights on Resource Management:
- GPU Utilization: High GPU utilization (e.g., 95-100%) is desirable. If utilization is low, it often indicates a CPU-side bottleneck, frequently in the neighbor search routine. Increasing
-nstlist(e.g., to 300) can mitigate this by reducing the frequency of this costly CPU task [39]. - CPU Role: In a well-tuned, GPU-accelerated run, the CPU's role shifts from computing non-bonded forces to managing the simulation, coordinating the GPU, and handling tasks like neighbor search and bond constraints. High CPU usage across all cores is not expected nor required for good performance; the CPU must simply be fast enough to keep the GPU fed with work [39].
- CPU Single-Core Performance: Powerful single-core CPU performance is critical for leveraging high-end GPUs like the RTX 4090. The primary MPI rank thread that coordinates the GPU must be able to schedule work efficiently. Server CPUs with high core counts but lower clock speeds can become a limiting factor [69].
The Scientist's Toolkit
Table 4: Essential Research Reagent Solutions for GROMACS Performance Benchmarking
| Item | Function / Relevance |
|---|---|
| Standardized Benchmark Systems (e.g., 20k, 80k, 1M atom systems) | Provides consistent and comparable test cases for evaluating performance across different hardware and software versions [67]. |
| GROMACS 2025.3 (or latest stable) | Ensures access to the latest performance optimizations (e.g., PME on GPUs, SIMD kernels) and critical stability fixes [40] [68] [70]. |
| CUDA 13 Toolkit | Required for compiling and running GROMACS with support for the latest NVIDIA GPU architectures [70]. |
| Consumer GPUs (e.g., RTX 4090/4080) | Delivers exceptional cost-effectiveness for most simulation sizes, ideal for scaling batch minimization jobs [67]. |
| High Single-Core Performance CPU | Prevents the CPU from becoming a bottleneck when paired with a powerful GPU, ensuring optimal GPU utilization [69]. |
| CUDA-Aware MPI Library (e.g., OpenMPI) | Enables advanced features like GPU direct communication for multi-GPU and multi-node simulations, reducing communication overhead [68]. |
Conclusion
Effective energy minimization using GROMACS mdrun is fundamental to successful molecular dynamics simulations, serving as the critical bridge between system preparation and production runs. By understanding the theoretical foundations, implementing robust methodological practices, mastering troubleshooting techniques, and rigorously validating outcomes, researchers can avoid common pitfalls that compromise simulation integrity. The choice of algorithm—whether robust steepest descent for problematic systems, conjugate gradients for higher accuracy, or L-BFGS for efficiency—should align with specific system characteristics and research objectives. Proper minimization establishes the foundation for reliable sampling of biomolecular conformational space, directly impacting the quality of insights into drug-target interactions, protein folding, and molecular mechanisms. As force fields continue evolving toward greater accuracy and systems increase in complexity, these energy minimization principles will remain essential for producing physically meaningful simulation data in biomedical research.
References
- 1. Energy Minimization - GROMACS 2025.3 documentation [https://manual.gromacs.org/documentation/2025.3/reference-manual/algorithms/energy-minimization.html]
- 2. gmx mdrun - GROMACS 2025.0 documentation [https://manual.gromacs.org/2025.0/onlinehelp/gmx-mdrun.html]
- 3. GROMACS Tutorial - Step Five: Energy Minimization [http://www.mdtutorials.com/gmx/lysozyme/05_EM.html]
- 4. Energy minimization stops abruptly without force convergence ... [https://gromacs.bioexcel.eu/t/energy-minimization-stops-abruptly-without-force-convergence-and-segmentation-fault/12899]
- 5. GROMACS Wizard - Step 2: Energy Minimization [https://documentation.samson-connect.net/tutorials/gromacs-wizard/energy-minimization/]
- 6. Understanding energy minimization working and acceptable ... [https://gromacs.bioexcel.eu/t/understanding-energy-minimization-working-and-acceptable-scores/194]
- 7. 5.2.2. gromacs.setup – Setting up a Gromacs MD run [https://gromacswrapper.readthedocs.io/en/latest/gromacs/blocks/setup.html]
- 8. Force field - GROMACS 2025.3 documentation [https://manual.gromacs.org/documentation/2025.3/reference-manual/functions/force-field.html]
- 9. Force fields in GROMACS [https://manual.gromacs.org/2025.1/user-guide/force-fields.html]
- 10. Energy minimization [https://en.wikipedia.org/wiki/Energy_minimization]
- 11. 3. Energy minimization - AdK Gromacs Tutorial - Read the Docs [https://adkgromacstutorial.readthedocs.io/en/latest/emin.html]
- 12. Energy Minimization - Steepest Descent [https://manual.gromacs.org/nightly/reference-manual/algorithms/energy-minimization.html]
- 13. Energy Minimization - GROMACS 2024.4 documentation [https://manual.gromacs.org/documentation/2024.4/reference-manual/algorithms/energy-minimization.html]
- 14. A comparison of three optimization algorithms for intensity ... [https://pubmed.ncbi.nlm.nih.gov/18705611/]
- 15. Navigating mdrun Challenges in GROMACS Simulations [https://blog.bioinfoquant.com/navigating-mdrun-challenges-in-gromacs-simulations-part-i]
- 16. 3. Energy minimization — Practical 13: AdK Tutorial ... [https://becksteinlab.physics.asu.edu/pages/courses/2013/SimBioNano/13/AdKTutorial/energy_minimization.html]
- 17. Introduction - GROMACS 2025.2 documentation [https://manual.gromacs.org/documentation/2025.2/reference-manual/introduction.html]
- 18. Energy Minimization - GROMACS 2025.2 documentation [https://manual.gromacs.org/documentation/2025.2/reference-manual/algorithms/energy-minimization.html]
- 19. Protocol for Molecular Dynamics Simulations of Proteins [https://en.bio-protocol.org/en/bpdetail?id=2051&type=0]
- 20. Acceleration of the GROMACS Free-Energy Perturbation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12163693/]
- 21. Molecular dynamics parameters (.mdp options) [https://manual.gromacs.org/current/user-guide/mdp-options.html]
- 22. Useful mdrun features - GROMACS 2025.4 documentation [https://manual.gromacs.org/current/user-guide/mdrun-features.html]
- 23. Introduction - GROMACS 2025.4 documentation [https://manual.gromacs.org/documentation/current/reference-manual/introduction.html]
- 24. Error in minimisation - User discussions - GROMACS forums [https://gromacs.bioexcel.eu/t/error-in-minimisation/9312]
- 25. Molecular dynamics parameters (.mdp options) [https://manual.gromacs.org/2025.0-beta/user-guide/mdp-options.html]
- 26. gmx energy - GROMACS 2025.4 documentation [https://manual.gromacs.org/current/onlinehelp/gmx-energy.html]
- 27. Movement of the molecules during energy minimization [https://gromacs.bioexcel.eu/t/movement-of-the-molecules-during-energy-minimization/11671]
- 28. Free energy calculations [https://manual.gromacs.org/current/reference-manual/algorithms/free-energy-calculations.html]
- 29. Molecular dynamics parameters (.mdp options) [https://manual.gromacs.org/5.1.1/user-guide/mdp-options.html]
- 30. mdp options [https://manual.gromacs.org/archive/4.5/online/mdp_opt.html]
- 31. MD minimization - User discussions - GROMACS forums [https://gromacs.bioexcel.eu/t/md-minimization/571]
- 32. gmx mdrun - GROMACS 2025.4 documentation [https://manual.gromacs.org/current/onlinehelp/gmx-mdrun.html]
- 33. Common errors when using GROMACS [https://manual.gromacs.org/2025.1/user-guide/run-time-errors.html]
- 34. Environment Variables - GROMACS 5.1.1 [https://manual.gromacs.org/5.1.1/user-guide/environment-variables.html]
- 35. System preparation - GROMACS 2025.4 documentation [https://manual.gromacs.org/current/user-guide/system-preparation.html]
- 36. Visualization Software - GROMACS 2025.3 documentation [https://manual.gromacs.org/current/how-to/visualize.html]
- 37. CharlesHahn/DuIvyTools: A Tool to process and visualize ... [https://github.com/CharlesHahn/DuIvyTools]
- 38. Performance improvements - GROMACS 2025.0 ... [https://manual.gromacs.org/2025.0/release-notes/2016/major/performance.html]
- 39. Optimizing CPU/GPU efficiency and performance in ... [https://gromacs.bioexcel.eu/t/optimizing-cpu-gpu-efficiency-and-performance-in-gromacs-simulations/11509]
- 40. Performance improvements [https://manual.gromacs.org/documentation/2025.0/release-notes/2018/major/performance.html]
- 41. Managing long simulations [https://manual.gromacs.org/2025.0/user-guide/managing-simulations.html]
- 42. System preparation - GROMACS 2025.0 documentation [https://manual.gromacs.org/2025.0/user-guide/system-preparation.html]
- 43. File formats - GROMACS 2025.4 documentation [https://manual.gromacs.org/current/reference-manual/file-formats.html]
- 44. Energy minimization using only CPU (GPU not used) [https://gromacs.bioexcel.eu/t/energy-minimization-using-only-cpu-gpu-not-used-gromacs-2025-3-version-used-on-ubuntu/12643]
- 45. Getting good performance from mdrun [https://manual.gromacs.org/current/user-guide/mdrun-performance.html]
- 46. When Energy Minimization Doesn't Converge - SAMSON Blog [https://blog.samson-connect.net/when-energy-minimization-doesnt-converge-what-it-means-and-what-to-do]
- 47. Energy minimization error - GROMACS forums [https://gromacs.bioexcel.eu/t/energy-minimization-error/478]
- 48. Common errors when using GROMACS [https://manual.gromacs.org/current/user-guide/run-time-errors.html]
- 49. 12 GROMACS Errors: From Setup to Execution [https://parssilico.com/blogs/99-12-gromacs-errors-from-setup-to-execution]
- 50. Automated Minimization of Steric Clashes in Protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3058769/]
- 51. File formats - GROMACS 5.1 [https://manual.gromacs.org/5.1/user-guide/file-formats.html]
- 52. Missing atom types in MD simulations - GROMACS forums [https://gromacs.bioexcel.eu/t/missing-atom-types-in-md-simulations/1855]
- 53. Getting segmentation fault mdrun - GROMACS forums [https://gromacs.bioexcel.eu/t/getting-segmentation-fault-mdrun/2826]
- 54. Segmentation fault - core dumped - GROMACS forums [https://gromacs.bioexcel.eu/t/segmentation-fault-core-dumped/6695]
- 55. Extreme RAM consumption of an md simulation ... - GROMACS forums [https://gromacs.bioexcel.eu/t/extreme-ram-consumption-of-an-md-simulation/7248]
- 56. Getting good performance from mdrun [https://manual.gromacs.org/documentation/2025.2/user-guide/mdrun-performance.html]
- 57. Computational BioPhysics Tutorials - 2025/2026 [https://cbp-unitn.gitlab.io/QCB/tutorial2_gromacs.html]
- 58. for Molecular Dynamics Simulations of Proteins Protocol [https://bio-protocol.org/en/bpdetail?id=2051&type=0]
- 59. 3 Energy — AdKGromacsTutorial 2.0.2 documentation minimization [https://adk-gromacs-tutorial.readthedocs.io/en/latest/emin.html]
- 60. Why Your Molecular Simulation May Fail Before It Begins [https://blog.samson-connect.net/why-your-molecular-simulation-may-fail-before-it-begins-energy-minimization-essentials]
- 61. Energy minimization works with steepest descent but not ... [https://gromacs.bioexcel.eu/t/energy-minimization-works-with-steepest-descent-but-not-conjugate-gradient/2174]
- 62. Energy minimisation - User discussions - GROMACS forums [https://gromacs.bioexcel.eu/t/energy-minimisation/5883]
- 63. Running membrane simulations in GROMACS [https://manual.gromacs.org/current/how-to/special.html]
- 64. Energy minimisation in membrane simulation - User discussions [https://gromacs.bioexcel.eu/t/energy-minimisation-in-membrane-simulation/2675]
- 65. Visualization Software - GROMACS 2025.0 documentation [https://manual.gromacs.org/2025.0/how-to/visualize.html]
- 66. Molecular dynamics parameters (.mdp options) [https://manual.gromacs.org/2025-rc/user-guide/mdp-options.html]
- 67. Molecular Simulation: GROMACS Benchmark on 30 GPUs [https://blog.salad.com/gromacs-benchmark/]
- 68. Performance improvements - GROMACS 2025.1 ... [https://manual.gromacs.org/2025.1/release-notes/2022/major/performance.html]
- 69. A series of performance benchmarks for MD Apps, including ... [https://gromacs.bioexcel.eu/t/a-series-of-performance-benchmarks-for-md-apps-including-gromacs/7078]
- 70. GROMACS 2025.3 Patch Release | Exxact Blog [https://www.exxactcorp.com/blog/molecular-dynamics/gromacs-2025-3-patch-release]