A Practical Guide to Protein-Ligand Complex Preparation for Energy Minimization
This article provides a comprehensive, step-by-step guide for researchers and drug development professionals on preparing protein-ligand complexes for energy minimization, a critical step in structure-based drug design.
A Practical Guide to Protein-Ligand Complex Preparation for Energy Minimization
Abstract
This article provides a comprehensive, step-by-step guide for researchers and drug development professionals on preparing protein-ligand complexes for energy minimization, a critical step in structure-based drug design. It covers the foundational principles of molecular interactions, detailed methodological workflows using common software tools like MOE and Schrödinger's Glide, strategies for troubleshooting common issues such as steric clashes and incorrect protonation states, and modern validation techniques including the use of high-quality datasets and deep learning models. By integrating both classical and AI-driven approaches, this guide aims to equip scientists with the knowledge to generate reliable and physically accurate models for virtual screening and lead optimization.
Understanding the Physics of Protein-Ligand Interactions for Stable Complexes
The Role of Minimization in Computer-Aided Drug Design (CADD)
In computer-aided drug design (CADD), the concept of a perfectly rigid protein-ligand complex is a theoretical abstraction; real-world molecular systems are dynamic and must be optimized for computational analysis. Energy minimization, also referred to as geometry optimization, is the fundamental computational process of adjusting the atomic coordinates of a molecular system to find a low-energy, and thus more stable, conformation [1]. This process transforms a structure, which may be a protein, ligand, or complex, into a state that is statistically favored and more likely to correspond to its natural state [1]. The imperative for minimization arises from the fact that initial molecular structures, whether derived from X-ray crystallography, NMR, or computational modeling, often contain steric clashes, distorted bond geometries, and other high-energy interactions that are artifacts of the model-building process rather than reflections of biological reality. Without minimization, these inaccuracies can severely compromise the results of subsequent computational analyses, such as molecular docking or molecular dynamics simulations, leading to false predictions.
Theoretical frameworks for ligand binding have evolved significantly from the early lock-and-key model to the induced-fit and population-shift models [2]. The lock-and-key model, which posits a rigid complementarity between the protein and ligand, has been largely superseded by models that acknowledge protein flexibility. The induced-fit model suggests that ligand binding induces conformational changes in the protein, while the population-shift model posits that the protein exists in an equilibrium of multiple conformations, with the ligand selectively stabilizing a subset of these [2]. Energy minimization is a practical computational tool that mimics aspects of these processes, allowing both the ligand and the protein to adapt to a more mutually complementary, low-energy arrangement, thereby simulating the local induced-fit effect [1] [2].
Theoretical Foundations of Molecular Energy Minimization
The Potential Energy Surface
The energy of a molecule is a function of the nuclear coordinates of its constituent atoms. The way in which this energy varies with the coordinates is described as the potential energy surface (PES) or hyper surface [3]. Stable states of molecular systems correspond to the minima on this multidimensional surface. The global minimum represents the most stable configuration, while local minima represent metastable states [3]. The objective of energy minimization is to locate one of these minima, starting from a non-equilibrium geometry. The process involves the mathematical procedure of optimization to move atoms so as to reduce the net forces on them—the gradients of the potential energy—until they become negligible [3].
Core Algorithms for Energy Minimization
Several iterative algorithms are employed to navigate the PES, each with a characteristic balance of computational cost and efficiency. The general update formula for these methods is x_new = x_old + correction, where the "correction" term varies [3].
- Steepest Descent: This method moves the atomic coordinates in the direction opposite to the largest gradient at the initial point. While robust for relieving severe steric clashes in initial structures, it can be slow to converge near the minimum as it tends to oscillate [3].
- Conjugate Gradient: To overcome the inefficiencies of Steepest Descent, the Conjugate Gradient method incorporates information from previous search directions. This reduces oscillation and allows for faster convergence to the minimum, making it a popular choice for many applications [3].
- Newton-Raphson: This is a more sophisticated and computationally expensive method per step, as it requires the calculation of second derivatives (the Hessian matrix). It converges quadratically near the minimum and is therefore very efficient for final stages of minimization when high precision is required [3].
Table 1: Comparison of Primary Energy Minimization Algorithms.
| Algorithm | Key Principle | Computational Cost | Convergence Speed | Typical Use Case |
|---|---|---|---|---|
| Steepest Descent | Moves atoms opposite the largest force (gradient). | Low per step | Fast initial progress, slow near minimum | Initial stabilization of poorly structured models. |
| Conjugate Gradient | Uses historical gradient data to avoid oscillation. | Moderate per step | Faster than Steepest Descent | General-purpose minimization after initial relaxation. |
| Newton-Raphson | Uses first and second derivatives (Hessian) for precise steps. | High per step (due to Hessian) | Very fast near minimum | Final, high-precision refinement of structures. |
Practical Implementation in a Drug Discovery Workflow
Preparation of the Protein-Ligand Complex
The first step in any minimization protocol is the careful preparation of the initial protein-ligand complex structure, typically obtained from the Protein Data Bank (PDB) [4]. This preparation involves several critical steps:
- Structure Cleaning: Removing extraneous water molecules, ions, and cofactors not essential for the binding interaction, though strategically placed water molecules can be retained.
- Protonation: Adding hydrogen atoms to the structure, which are often not resolved in X-ray structures. This includes assigning physiologically relevant protonation states and tautomers to amino acid side chains (e.g., His, Asp, Glu) and the ligand, a step that is highly dependent on the local pH [5] [4].
- Force Field Parameter Assignment: The molecular system must be described within a force field—a collection of formulas and parameters used to simulate atomic systems [1]. Tools like AutoSMILES in YASARA automate this process by performing pH-dependent bond order assignment, semi-empirical charge calculations, and parameter refinement, which is crucial for simulating a wide range of ligands [1]. Compatible force fields include various versions of AMBER, CHARMM, and YASARA's own force fields like YAMBER and YASARA2 [1] [4].
Table 2: Essential Research Reagent Solutions for Minimization and Preparation.
| Tool / Resource | Type | Primary Function in Preparation/Minimization |
|---|---|---|
| YASARA AutoSMILES [1] | Software Module | Automated force field parameter assignment and refinement for ligands. |
| CGenFF / Antechamber [4] | Parameterization Tool | Generates force field parameters for small molecules for CHARMM and AMBER, respectively. |
| PDB [4] | Database | Source for initial 3D structures of protein-ligand complexes. |
| AMBER, CHARMM, YAMBER [1] [4] | Force Field | Defines the energy function and parameters for molecular mechanics calculations. |
Configuring the Minimization Experiment
Once the system is prepared, the minimization experiment must be configured. A key strategic decision is the degree of flexibility allowed for the protein.
- Rigid Backbone Minimization: In this approach, the protein backbone's heavy atoms are held fixed. Only the ligand and possibly the side chains of the protein's binding site residues are allowed to move. This method is computationally less expensive and is often used for high-throughput virtual screening or when the protein structure is considered highly reliable [1].
- Flexible Backbone Minimization: Here, the protein's backbone is also allowed to adjust. This simulates a more realistic induced fit, where both the ligand and the target structure adapt to each other, which can be critical for identifying novel binding pockets or accommodating larger ligands [1]. This method is more computationally intensive but can provide deeper insights, especially when dealing with apo (ligand-free) protein structures.
The following workflow diagram outlines the key decision points and steps in a standard minimization protocol for preparing a protein-ligand complex.
Applications and Impact on Drug Discovery
Energy minimization is not merely a preparatory step; it is an integral part of the molecular modeling workflow that enables several key applications in drug discovery.
- Pose Refinement and Validation: Especially for smaller, fragment-like molecules, predicting the correct binding mode is challenging. A subsequent energy minimization step following docking can provide additional confidence by refining the ligand's position and optimizing its interactions with the target, ensuring that the best plausible pose has been selected for further design steps [1].
- Resolving Steric Clashes and Expanding Binding Sites: Minimization can be employed when a binding site is too narrow to host a ligand. By temporarily tolerating initial clashes and then performing energy minimization, both the ligand and the protein are allowed to adapt, creating more space and simulating an induced fit [1]. This can be used to generate space in subpockets or explore potential rotamers that expand the binding site of apo structures [1].
- Revealing Novel Interactions: The process of optimization can lead to the emergence of new, favorable interactions with side chains, the backbone, water molecules, or metals that were not apparent in the initial model. These observations can positively impact the estimated ligand score and provide valuable insights for medicinal chemistry optimization [1].
The following diagram illustrates how energy minimization is embedded within a broader CADD pipeline, highlighting its role in connecting different computational techniques.
Advanced Considerations and Future Outlook
While fundamental, energy minimization has limitations. It typically finds the nearest local minimum on the PES, which may not be the global minimum or the biologically most relevant state [3]. To overcome this, researchers use more advanced sampling techniques.
- Molecular Dynamics (MD) Simulations: MD simulations model the physical movements of atoms over time by numerically solving Newton's equations of motion. This allows the system to overcome small energy barriers and sample a wider range of conformational states, providing a dynamic picture of protein-ligand interactions beyond a single minimized snapshot [2] [4].
- Accounting for Global Flexibility: Sophisticated CADD techniques now explicitly account for full protein flexibility by using multiple protein structures. These can be derived from experimental sources (e.g., multiple X-ray structures or NMR ensembles) or computational methods like MD simulations. Protocols such as the relaxed complex scheme—where ligands are docked into an ensemble of protein structures—or dynamic pharmacophore generation more comprehensively capture the reality of protein dynamics as described by the population-shift model [2].
The continued integration of energy minimization as a core component within these more extensive and computationally demanding workflows ensures that it remains an indispensable tool for achieving high-quality, reliable results in structure-based drug design.
In the realm of structural biology and rational drug design, a profound understanding of non-covalent interactions is indispensable. These interactions, while weaker than covalent bonds, are the fundamental forces governing the three-dimensional structure of proteins, the specific binding of ligands, and the functional dynamics of biological macromolecules [6] [7]. The preparation of a protein-ligand complex for computational minimization and simulation is a critical step whose success is entirely contingent upon a correct physical representation of these forces. An inaccurate model of non-covalent interactions will lead to unrealistic minimized structures, flawed binding affinity predictions, and ultimately, a failed research outcome. This whitepaper provides an in-depth technical guide to the key non-covalent interactions—hydrogen bonds, ionic, van der Waals, and hydrophobic effects—framed within the practical context of preparing a protein-ligand complex for energy minimization. It is structured to equip researchers with the necessary knowledge to make informed decisions during system setup, to select appropriate force field parameters, and to interpret the results of minimization protocols with a critical understanding of the underlying molecular forces.
Non-covalent interactions are electromagnetic in nature and differ from covalent bonds primarily in that they do not involve the sharing of electrons [6]. The chemical energy released during their formation is typically on the order of 1–5 kcal/mol, which is significantly weaker than a covalent bond (on the order of 100 kcal/mol) but becomes highly significant in aggregate [6] [8]. The following sections detail the primary non-covalent interactions relevant to protein-ligand complexes.
Table 1: Key Characteristics of Non-Covalent Interactions
| Interaction Type | Strength (kcal/mol) | Distance Dependence | Directionality | Primary Contributor to |
|---|---|---|---|---|
| Ionic | 1-5 (in vacuum); highly screen-dependent [6] | 1/r² | Low | Surface salt bridges, ligand recognition [9] |
| Hydrogen Bond | 1-5 (can reach 40 in strong bonds) [6] | ~1/r⁶ | High | Secondary structure (α-helices, β-sheets), ligand specificity [9] |
| van der Waals | 1-2 (per atom pair) [8] | ~1/r⁶ | Low | Close-range packing, steric complementarity [6] |
| Hydrophobic Effect | Not a direct force; ΔG driven by ΔS [6] | N/A | N/A | Core protein folding, burial of non-polar surfaces [7] |
Hydrogen Bonds
A hydrogen bond is a strong, highly directional non-covalent interaction between a partially positive hydrogen atom (donor) bound to a highly electronegative atom (e.g., O, N), and a partially negative acceptor atom, typically oxygen or nitrogen, possessing a lone pair of electrons [6] [9]. In proteins, hydrogen bonds are the principal force behind the stability of secondary structures such as α-helices and β-strands [9]. In ligand binding, they are a key determinant of specificity.
Experimental Consideration for Minimization: During the preparation of a protein-ligand complex, it is critical to ensure the correct protonation states of potential donor and acceptor groups. The hydrogen bonding geometry should be checked post-minimization, as optimal interaction energy occurs at specific donor-acceptor distances and angles. A well-positioned hydrogen bond can contribute ~5 kJ/mol (~1.2 kcal/mol) of stabilization in a solvent like chloroform, but this value is highly sensitive to the dielectric constant of the environment [6].
Ionic Interactions (Salt Bridges)
Ionic interactions involve the electrostatic attraction between two permanently charged groups of opposite signs, such as the negative carboxylate group of aspartate or glutamate and the positive ammonium group of lysine [6] [9]. These interactions can be strong in the hydrophobic core of a protein where the dielectric constant is low but are significantly weakened in aqueous environments due to the high dielectric constant of water and competitive solvation by ions [7].
Experimental Consideration for Minimization: The strength of an ionic interaction is inversely proportional to the square of the distance between the charges and is heavily influenced by the dielectric properties of the assigned force field. A single salt bridge in a protein's interior can contribute ~5 kJ/mol of attraction at intermediate ion strength [6]. Proper assignment of partial charges on the ligand and protein is paramount for accurately modeling these interactions during minimization.
van der Waals Forces
van der Waals forces are a subset of electrostatic interactions involving permanent or induced dipoles and can be broken down into three types: permanent dipole-dipole (Keesom force), dipole-induced dipole (Debye force), and induced dipole-induced dipole (London dispersion forces) [6]. Dispersion forces are present between all atoms and arise from correlated fluctuations of electron clouds, creating transient dipoles [8]. Although weak for an individual atom pair, the cumulative effect of many van der Waals contacts across a protein-ligand interface is a major driver of binding affinity and is essential for achieving close molecular packing [6].
Experimental Consideration for Minimization: The potential energy of van der Waals interactions follows a Lennard-Jones potential, with a steep repulsive wall at short distances and a favorable attractive region at optimal van der Waals contact distances [8]. Minimization algorithms must carefully navigate this energy landscape to avoid unrealistic atomic clashes while optimizing the multitude of favorable dispersive contacts.
Hydrophobic Effect
The hydrophobic effect is not a direct attractive force but rather an entropy-driven phenomenon. It describes the tendency of non-polar substances to aggregate in an aqueous solution to minimize the ordered "cage" of water molecules that would otherwise form around them [6] [7]. This release of ordered water into the bulk solvent increases the overall entropy of the system, making the association favorable. It is the primary driver of protein folding, leading to the formation of a hydrophobic core, and is a critical component of ligand binding, especially when non-polar regions of the ligand are buried in hydrophobic pockets on the protein [7].
Experimental Consideration for Minimization: The hydrophobic effect is implicitly modeled in most classical force fields through a combination of van der Waals parameters and a surface-area-dependent term, or explicitly in water models. Understanding that this is a major contributor to the driving force for binding, but is not a single parameter in a minimization, is crucial.
Experimental and Computational Methodologies for Investigation
The investigation of protein-ligand binding, driven by non-covalent interactions, employs a suite of biophysical and computational techniques [10]. Integrating these methods provides a comprehensive picture of the binding event.
Table 2: Key Methodologies for Studying Protein-Ligand Interactions
| Method Category | Example Techniques | Information Provided | Utility in Minimization Context |
|---|---|---|---|
| Thermal/Thermodynamic | Isothermal Titration Calorimetry (ITC), Thermal Shift Assay (TSA/CETSA) [10] | Binding affinity (Kd), enthalpy (ΔH), entropy (ΔS), complex stability | Provides experimental benchmarks for validating computational models and scoring functions. |
| Spectroscopic | Circular Dichroism (CD), Nuclear Magnetic Resonance (NMR), Surface Plasmon Resonance (SPR) [10] | Structural changes, binding kinetics (kon, koff), residue-specific interaction data | Offers insights into conformational selection and structural integrity post-minimization. |
| 3D Structure Determination | X-ray Crystallography, Cryo-Electron Microscopy (Cryo-EM) [11] [10] | Atomic-resolution 3D structure of the complex | The primary source for initial atomic coordinates for minimization; the "ground truth" for pose validation. |
| Mass Spectrometry | Native ESI-MS [10] | Detection and quantification of non-covalent complexes | Confirms the existence and stoichiometry of the complex under study. |
| Prediction & Simulation | Molecular Docking, Molecular Dynamics (MD) [10] | Binding pose prediction, dynamics of interactions, binding free energies | The core computational methods for which system minimization is a critical preparatory step. |
A Workflow for High-Quality Structure Preparation
The accuracy of any minimization or simulation is predicated on the quality of the initial structural model. A robust workflow for preparing a protein-ligand complex is essential. Recent research highlights common issues in widely-used datasets like PDBbind, such as incorrect bond orders, poor protonation states, and steric clashes, which can compromise downstream applications [12]. The following workflow, inspired by the HiQBind-WF, outlines a semi-automated, high-fidelity preparation process.
Diagram 1: Structure Preparation Workflow
Detailed Protocol for Workflow Execution:
Data Curation & Filtering:
- Input: Obtain the protein-ligand complex structure in PDB format from the RCSB PDB [12].
- Splitting: Split the structure into three components: the protein, the ligand(s) of interest, and additives (ions, solvents, co-factors) [12].
- Filtering: Apply a series of filters to reject structures that are unsuitable for minimization of non-covalent complexes. This includes:
- Removing ligands that are covalently bonded to the protein.
- Excluding ligands with rare elements not well-parameterized in standard force fields.
- Identifying and potentially rejecting complexes with severe steric clashes [12].
Structure Repair:
- Ligand Fixing (LigandFixer): Process the ligand structure to ensure correctness. This involves:
- Assigning correct bond orders and aromaticity.
- Determining a reasonable protonation state at the physiological pH of interest [12].
- Protein Fixing (ProteinFixer): Process the protein structure to add missing atoms (e.g., in side chains like ASN, GLN) and, if necessary, model missing loops or residues [12].
- Ligand Fixing (LigandFixer): Process the ligand structure to ensure correctness. This involves:
Complex Assembly & Refinement:
- Recombination: Recombine the fixed protein and ligand structures.
- Hydrogen Addition: Add hydrogen atoms to both the protein and the ligand in the context of the complex. This is a critical step, as it ensures that hydrogen bonds are modeled correctly between the protein and ligand, rather than independently [12].
- Constrained Energy Minimization: Perform a light, constrained energy minimization to resolve minor steric clashes introduced during hydrogen addition and to relax the structure into a local energy minimum, ready for subsequent production simulation or analysis [12].
Successful research into protein-ligand interactions relies on a foundation of key databases, software, and reagents.
Table 3: Essential Research Resources for Protein-Ligand Studies
| Resource Name | Type | Primary Function | Relevance to Non-Covalent Interactions |
|---|---|---|---|
| RCSB Protein Data Bank (PDB) [11] | Database | Repository for 3D structural data of proteins and nucleic acids. | Source of initial atomic coordinates for protein-ligand complexes, revealing geometry of non-covalent contacts. |
| PDBbind [12] | Curated Database | Collection of protein-ligand complexes with experimentally measured binding affinities. | Provides a benchmark for training and testing scoring functions that predict binding strength from non-covalent interactions. |
| BindingDB [12] | Database | Public database of measured binding affinities, focusing on drug-target interactions. | Source of experimental binding data for validation. |
| Molecular Dynamics Software (e.g., GROMACS, AMBER, NAMD) | Software | Simulates the physical movements of atoms and molecules over time. | Models the dynamics and stability of non-covalent interactions in a solvated, physiological environment. |
| Docking Software (e.g., AutoDock, Glide) | Software | Predicts the preferred orientation of a ligand bound to a protein. | Relies on scoring functions to evaluate poses based on estimated contributions from non-covalent forces. |
| CETSA / TSA Reagents [10] | Experimental Kit | Cellular or purified protein, thermal stability assay buffers. | Experimentally measures target engagement and stabilization of a protein by a ligand via non-covalent binding. |
| ITC Instrumentation [10] | Instrument | Measures heat change upon binding. | Directly quantifies the thermodynamic parameters (ΔH, Ka) of a binding event, deconvoluting enthalpic contributions. |
Non-covalent interactions are the subtle yet powerful language of molecular recognition in biological systems. A meticulous understanding of hydrogen bonds, ionic interactions, van der Waals forces, and the hydrophobic effect is not merely academic; it is a practical necessity for researchers preparing protein-ligand complexes for computational minimization. By applying rigorous structural preparation workflows, leveraging high-quality curated datasets, and utilizing the appropriate experimental and computational tools, scientists can ensure their models accurately reflect physical reality. This rigorous approach is fundamental to advancing reliable drug discovery and deepening our comprehension of biomolecular function.
The binding affinity between a protein and a ligand is quantifiably expressed as the change in Gibbs free energy (ΔG), a fundamental parameter in drug discovery and structural biology. This thermodynamic quantity results from the combined, often opposing, contributions of enthalpy (ΔH) and entropy (ΔS), as described by the equation ΔG = ΔH - TΔS. A pervasive and frequently confounding phenomenon known as enthalpy-entropy compensation (EEC) is often observed in protein-ligand interactions, wherein favorable enthalpic gains are offset by entropic penalties, and vice-versa. This compensation can frustrate rational drug design efforts aimed at improving binding affinity. This whitepaper provides an in-depth technical examination of EEC, its statistical prevalence, and its profound implications for preparing and analyzing protein-ligand complexes, with a specific focus on the context of minimization research. We further detail advanced computational and experimental methodologies for accurately characterizing binding thermodynamics, providing a foundational guide for researchers in the field.
The formation of a protein-ligand complex is a spontaneous process governed by a negative change in the Gibbs free energy (ΔG < 0). The magnitude of ΔG directly determines the binding affinity, with more negative values indicating tighter binding [13] [14]. This free energy change is partitioned into two components: the enthalpy change (ΔH), representing the heat released or absorbed during binding (primarily from the formation and breaking of non-covalent interactions), and the entropy change (TΔS), which reflects the change in the system's disorder, including the ligand, protein, and solvent water molecules [15].
The interplay between these components is formalized by the central equation: ΔG = ΔH - TΔS A key challenge in optimizing binding affinity arises from the phenomenon of enthalpy-entropy compensation (EEC), where a more favorable (negative) ΔH is counterbalanced by a less favorable (negative) TΔS, resulting in a minimal net improvement in ΔG [16] [15]. For researchers preparing protein-ligand complexes for minimization, understanding and anticipating EEC is critical, as modifications intended to strengthen interactions may not yield the expected gains in affinity due to this compensatory effect.
The Principle of Enthalpy-Entropy Compensation
Fundamental Concept and Mathematical Basis
Enthalpy-entropy compensation describes a linear relationship between the ΔH and ΔS terms for a series of related reactions or, in this context, for a series of protein-ligand interactions [16]. This relationship can be expressed as: ΔH = α + βΔS where α is a constant and β is the compensation temperature, often possessing dimensions of temperature (e.g., the isokinetic or isoequilibrium temperature) [16]. When this relationship holds, the substitution into the Gibbs free energy equation yields: ΔG = α - (T - β) ΔS This formulation makes it clear that at the temperature T = β, the ΔG value becomes constant (ΔG = α) for all members of the reaction series, independent of ΔS [16]. This represents a perfect, and theoretically frustrating, form of compensation.
Prevalence and Statistical Evidence in Protein-Ligand Interactions
Empirical evidence suggests that EEC is widespread in biomolecular interactions. A meta-analysis of 171 protein-ligand interactions across 32 diverse proteins revealed a strong linear correlation between ΔH and -TΔS, with a slope near unity [17]. This correlation is visually evident when data is plotted on a ΔH versus -TΔS graph, often forming a diagonal band [15] [17].
Statistical analysis of large datasets provides further insight. The distribution of binding affinities for over 3,000 protein-ligand complexes reveals a Gaussian distribution centered around a ΔG of approximately -36.5 kJ/mol (-8.7 kcal/mol), with the majority of values falling between -46 and -26 kJ/mol (-11.0 and -6.2 kcal/mol) [18]. This narrow range of observed ΔG values, despite vastly wider variations in the individual ΔH and TΔS components, is a direct consequence of EEC. The following table summarizes key statistical findings from large-scale analyses:
Table 1: Statistical Evidence for EEC from Large-Scale Data Analyses
| Dataset Description | Number of Complexes | Key Finding | Reference |
|---|---|---|---|
| Diverse protein-ligand interactions | 171 | Strong linear correlation between ΔH and -TΔS across 32 unrelated proteins. | [17] |
| Protein-ligand affinities from PDB | 3,025 | ΔG values are normally distributed around -36.5 kJ/mol, a narrow range implying EEC. | [18] |
| Meta-analysis from BindingDB | ~100 | Plot of ΔH vs. TΔS showed a slope of nearly unity, suggesting severe compensation. | [15] |
The mechanistic origins of EEC, while not fully unified, are often attributed to the properties of water and hydrogen bonding [16] [15], conformational changes in the protein and ligand upon binding, and alterations in solvent dynamics. From an evolutionary perspective, it has been proposed that EEC may act as a homeostatic mechanism, allowing proteins to maintain binding affinities adaptive to changes in ligand availability and environmental conditions [18] [19].
Experimental and Computational Methodologies
Accurately determining the thermodynamic parameters of protein-ligand binding is a cornerstone of biophysical research. The following section details key experimental and computational approaches, with an emphasis on the protocols and workflows relevant for rigorous characterization.
Experimental Determination via Isothermal Titration Calorimetry (ITC)
Protocol Overview: ITC is the gold standard for directly measuring the thermodynamics of binding in solution. In a single experiment, it determines the association constant (K~a~), stoichiometry (n), and enthalpy change (ΔH) [15] [17].
Detailed Methodology:
- Sample Preparation: Both the protein and ligand are dissolved in identical buffers to avoid heat effects from buffer mismatch. Samples are thoroughly degassed to prevent bubble formation in the instrument cell.
- Instrument Setup: The protein solution is loaded into the sample cell. The ligand solution is loaded into the injection syringe. A reference cell is filled with water or dialysate.
- Titration Experiment: The ligand is injected in a series of small aliquots into the protein solution. After each injection, the instrument measures the heat required to maintain the sample cell at the same temperature as the reference cell.
- Data Analysis: The resulting isotherm (a plot of heat released or absorbed per injection versus the molar ratio) is fit using a nonlinear regression model to obtain K~a~, n, and ΔH.
- Derivation of Parameters: The Gibbs free energy is calculated as ΔG = -RT ln(K~a~), and the entropic contribution is derived from the relationship TΔS = ΔH - ΔG.
Critical Considerations: ITC measurements are constrained by an "affinity window," typically best suited for K~d~ values between nanomolar and micromolar ranges. Weaker or tighter binding requires specialized techniques like competition assays [17]. While ITC provides direct ΔH measurements, the correlation between ΔH and TΔS calculated from ΔG can be influenced by this affinity window and must be interpreted with statistical care [15] [17].
Computational Determination Using Molecular Dynamics (MD)
Computational methods offer a powerful, atomistic view of the binding process and its thermodynamics. Enhanced sampling methods are often required to overcome the high energy barriers associated with ligand binding and unbinding.
A. Dissociation Parallel Cascade Selection MD (dPaCS-MD) with Markov State Models (MSM) Protocol Overview: This method generates multiple unbinding pathways and uses them to construct a kinetic model for calculating the binding free energy profile [20].
Detailed Workflow:
- System Setup: The protein-ligand complex is solvated in a water box, ions are added to neutralize the system, and force field parameters are assigned to the ligand.
- dPaCS-MD Simulation: Cycles of multiple parallel, short MD simulations are run. In each cycle, initial structures for the next round are selected based on a collective variable (CV), typically the distance between the protein and ligand, favoring structures with longer separation distances.
- MSM Construction: The ensemble of short trajectories is analyzed. Conformations are clustered into discrete states based on geometric criteria (e.g., protein-ligand distance, specific interaction patterns). A transition matrix is built to estimate the probabilities of moving between states.
- Free Energy Calculation: The MSM's stationary distribution is used to compute the free energy profile along the dissociation coordinate. The standard binding free energy (ΔG°) is derived from this profile [20].
Table 2: Performance of dPaCS-MD/MSM on Benchmark Systems
| Protein-Ligand Complex | Computed ΔG (kcal/mol) | Experimental ΔG (kcal/mol) | Reference |
|---|---|---|---|
| Trypsin/Benzamidine | -6.1 ± 0.1 | -6.4 to -7.3 | [20] |
| FKBP/FK506 | -13.6 ± 1.6 | -12.9 | [20] |
| Adenosine A2A Receptor/T4E | -14.3 ± 1.2 | -13.2 | [20] |
B. Absolute Binding Free Energy (ABFE) Calculations with Non-Equilibrium Approaches Protocol Overview: These alchemical methods compute the absolute ΔG by non-physically decoupling the ligand from its environment in the bound and unbound states [21].
Detailed Workflow:
- System Preparation: Similar to dPaCS-MD, the bound and unbound (ligand in solvent) states are prepared with appropriate solvation and force fields.
- Equilibrium Sampling: Equilibrium MD simulations are performed for the two physical end states (fully coupled ligand in protein and in solvent).
- Non-Equilibrium Transitions: Rapid "switching" simulations are initiated from these equilibrated states, during which the Hamiltonian is morphed to decouple the ligand over a short time (e.g., 500 ps). The work required for each transition is recorded.
- Free Energy Estimation: The free energy difference is recovered from the distribution of work values using the Crooks Fluctuation Theorem (for bi-directional transitions) or Jarzynski's equality (for uni-directional transitions) [21]. This approach has been shown to achieve accuracy comparable to sophisticated equilibrium FEP methods enhanced by Hamiltonian replica exchange.
The diagram below illustrates the logical relationship and workflow between the primary methodologies discussed for determining binding thermodynamics.
Diagram 1: Methodologies for Determining Binding Thermodynamics
The Scientist's Toolkit: Essential Research Reagents and Solutions
The following table catalogues critical reagents, software, and data resources essential for conducting research on protein-ligand thermodynamics and minimization.
Table 3: Essential Research Reagents and Resources
| Category / Item | Specifications / Examples | Primary Function / Application |
|---|---|---|
| Experimental Reagents | ||
| Purified Protein | >95% homogeneity, known concentration and activity. | The target macromolecule for binding studies. |
| Ligand Compound | High-purity, soluble in aqueous buffer. | The small molecule whose binding is being characterized. |
| ITC Buffer | Phosphate-buffered saline (PBS) or other non-interacting buffers. | Provides a consistent chemical environment; must be matched for protein and ligand. |
| Software & Force Fields | ||
| Molecular Dynamics Engines | AMBER, GROMACS, NAMD. | Performs the numerical integration of Newton's equations of motion for the molecular system. |
| Enhanced Sampling Tools | Plumed (plugin for MD codes). | Implements advanced sampling algorithms like metadynamics or umbrella sampling. |
| Force Fields | AMBER ff14SB/GAFF, CHARMM36. | Defines the potential energy function and parameters for proteins, ligands, and solvent. |
| Analysis & Modeling | MSMBuilder, PyEMMA. | Constructs and analyzes Markov State Models from MD trajectory data. |
| Databases | ||
| Protein Data Bank (PDB) | https://www.rcsb.org/ | Repository for 3D structural data of proteins and nucleic acids. |
| BindingDB | https://www.bindingdb.org/ | Public database of measured binding affinities, focusing on drug targets. |
| PDBbind | http://www.pdbbind.org.cn/ | A curated database of experimentally measured binding affinities for biomolecular complexes in the PDB. |
Implications for Protein-Ligand Minimization Research
The principles of EEC and binding thermodynamics have direct and consequential implications for preparing and analyzing protein-ligand complexes, particularly in the context of energy minimization and structure refinement.
Interpreting Minimization Outcomes: Energy minimization algorithms often focus on optimizing enthalpic contributions, such as improving van der Waals contacts, electrostatic interactions, and hydrogen bonding networks. However, due to EEC, these enthalpic gains may not translate directly to a more stable complex in a physiological context because of accompanying entropic losses (e.g., reduced conformational flexibility of the ligand or protein). Researchers must therefore use scoring functions or analysis protocols that consider both enthalpic and entropic components, rather than relying solely on potential energy.
Guiding Rational Ligand Design: The phenomenon of EEC explains why certain ligand modifications, such as the introduction of a hydrogen bond donor, sometimes fail to improve binding affinity as expected. The enthalpic benefit of the new bond can be compensated by the entropic cost of restricting the motion of the protein, ligand, or surrounding water molecules [15]. Consequently, minimization protocols on such modified complexes should be analyzed for changes in flexibility and solvation, not just interaction energy.
Informing the Choice of Scoring Functions: For virtual screening and pose prediction, standard docking scores are often poor proxies for binding affinity because they are typically enthalpy-dominated and lack a true treatment of entropy and solvation [22]. The development of machine-learning-based scoring functions trained on experimental data (e.g., ΔG from ITC) and advanced end-state methods like alchemical free energy calculations are becoming increasingly important for achieving predictive accuracy in drug discovery campaigns [21] [22].
The Gibbs free energy provides the definitive metric for the stability of a protein-ligand complex. The widespread occurrence of enthalpy-entropy compensation presents a significant challenge, implying that the optimization of binding affinity is a multi-dimensional problem that cannot be solved by focusing on enthalpy alone. For researchers engaged in minimization studies, this necessitates a paradigm that integrates robust computational methods—capable of capturing both energetic and entropic effects—with high-quality experimental validation. Moving forward, strategies that combine detailed structural analysis from minimization with rigorous thermodynamic profiling from ITC and advanced free energy calculations will be essential to circumvent the hurdles imposed by compensation and to enable the rational design of high-affinity ligands.
Molecular recognition, the specific interaction between biological macromolecules and their ligands, is fundamental to all biological processes, including signaling, catalysis, and gene regulation [23]. Understanding these mechanisms is particularly crucial for structure-based drug design, where elucidating protein-ligand interactions guides the development of therapeutic compounds [24]. The conceptual framework for understanding molecular recognition has evolved significantly from early static models to modern dynamic interpretations that account for protein flexibility and conformational heterogeneity.
This whitepaper provides a comprehensive overview of the primary molecular recognition models, their theoretical foundations, and their practical implications for preparing protein-ligand complexes in minimization research. We present quantitative comparisons of these models, detailed experimental methodologies for their study, and specific protocols relevant to computational drug discovery. By integrating historical perspectives with cutting-edge approaches like deep learning-driven dynamic docking, we aim to equip researchers with the theoretical knowledge and practical frameworks necessary to advance their work in structural biology and rational drug design.
Theoretical Foundations of Molecular Recognition
Evolution of Molecular Recognition Models
Three primary models have emerged to explain the structural basis of molecular recognition, each representing a significant paradigm shift in our understanding of biomolecular interactions.
1. Lock-and-Key Model (Fischer, 1894)
- Premise: The protein (lock) and ligand (key) possess complementary, pre-formed geometries that fit perfectly without conformational adjustments [24] [25].
- Theoretical Basis: An entropy-dominated binding process where rigid bodies associate with minimal reorganization energy [24].
- Limitations: Oversimplifies biological systems by neglecting inherent protein dynamics and conformational flexibility.
2. Induced Fit Model (Koshland, 1958)
- Premise: Ligand binding induces conformational changes in the protein structure to achieve optimal complementarity [23] [24].
- Theoretical Basis: The initial binding event triggers structural rearrangements that enhance binding affinity through improved surface complementarity and interaction networks.
- Distinction: Unlike lock-and-key, this model posits that the bound conformation does not significantly pre-exist in the unliganded protein ensemble [23].
3. Conformational Selection Model (Monod et al., 1965)
- Premise: Multiple protein conformations pre-exist in dynamic equilibrium, with ligands selectively binding to and stabilizing complementary conformations [23] [25].
- Theoretical Basis: Rooted in energy landscape theory, where proteins sample multiple conformational substates, and ligand binding causes a population shift toward favorable conformations [23].
- Extension: Often operates in a hybrid mechanism where initial conformational selection is followed by induced fit optimizations [25].
Table 1: Comparative Analysis of Molecular Recognition Models
| Feature | Lock-and-Key | Induced Fit | Conformational Selection |
|---|---|---|---|
| Protein Flexibility | Rigid | Adaptive | Pre-existing ensemble |
| Conformational Change | None | Post-binding | Pre-equilibrium |
| Ligand Specificity | High | Modifiable | Selective |
| Thermodynamic Control | Entropy-driven | Enthalpy-driven | Population shift |
| Temporal Order | Simultaneous | Conformation follows binding | Binding follows conformation |
| Experimental Evidence | Static structures | Kinetics, structural biology | NMR, kinetics, MD simulations |
Energy Landscape Theory and Conformational Ensembles
The conformational selection model derives from the energy landscape theory of protein folding and function [23]. According to this framework, a protein samples an ensemble of conformational substates in dynamic equilibrium, with the populations of these states following statistical thermodynamic distributions [23]. The heights of energy barriers between substates determine the timescales of conformational exchange.
Within this paradigm, ligands may interact not only with the lowest energy conformational state but also with higher energy substates that are transiently populated in solution [23]. The binding interaction doesn't 'induce' a new conformation but rather redistributes the relative populations of conformational states that already pre-exist through a process of population shift [23].
Diagram 1: Energy landscape of molecular recognition. Proteins exist as ensembles of conformations (P1, P2). Ligands can bind different states via lock-and-key, induced fit, or conformational selection pathways.
Practical Implications for Protein-Ligand Complex Preparation
Computational Challenges in Molecular Docking
Traditional molecular docking methods often treat proteins as rigid structures due to computational constraints, despite the recognized importance of protein dynamics [26]. This approach presents significant limitations, as AlphaFold-predicted apoprotein structures frequently yield ligand pose predictions that poorly align with experimental holo-structures [26]. The fundamental challenge lies in the fact that binding-competent conformations may be rarely populated in the unliganded ensemble, creating a sampling problem for both computational and experimental approaches.
Key Challenges in Accounting for Protein Flexibility:
- Rare Transitions: Biologically relevant conformational changes often involve rare transitions between metastable states with high energy barriers [26].
- Computational Cost: Molecular dynamics (MD) simulations can propose appropriate conformations but are computationally demanding for capturing rare events [26].
- Cryptic Pockets: Binding pockets may not be apparent in static structures or apo conformations, requiring conformational changes for accessibility [26].
Advanced Docking Methodologies
Recent advances address these challenges through innovative computational approaches:
DynamicBind: A Deep Generative Model for Dynamic Docking DynamicBind employs an SE(3)-equivariant geometric diffusion network to construct a smooth energy landscape that facilitates efficient transitions between equilibrium states [26]. Unlike traditional docking, DynamicBind adjusts protein conformation from initial AlphaFold predictions to holo-like states while simultaneously optimizing ligand position and orientation [26].
Table 2: Performance Comparison of Docking Methods on PDBbind and MDT Test Sets
| Method | Type | Ligand RMSD <2Å (%) | Ligand RMSD <5Å (%) | Success Rate (Stringent) |
|---|---|---|---|---|
| DynamicBind | Dynamic docking | 33-39 | 65-68 | 0.33 |
| DiffDock | Deep learning | - | - | 0.19 |
| GNINA | Force field-based | - | - | - |
| GLIDE | Force field-based | - | - | - |
| VINA | Force field-based | - | - | - |
Key Innovations in DynamicBind:
- Morph-like Transformation: Unlike traditional diffusion models that apply Gaussian noise, DynamicBind uses morph-like transformations between AlphaFold-predicted and holo conformations during training, maintaining chemical constraints while learning biologically relevant transitions [26].
- Simultaneous Optimization: The model progressively translates and rotates both ligand and protein residues while modifying torsional angles over 20 iterations [26].
- Funneled Energy Landscape: Creates a minimally frustrated energy landscape that lowers free energy barriers between biologically relevant states [26].
Ensemble Docking with AlphaFold2 and MD Refinements Recent benchmarks demonstrate that AlphaFold2 (AF2) models perform comparably to experimental structures in docking protocols targeting protein-protein interactions [27]. Integration of molecular dynamics simulations and ensemble generation algorithms like AlphaFlow can further refine both native and AF2 models, though outcomes vary significantly across conformations [27].
Diagram 2: DynamicBind dynamic docking workflow. The process transforms apo protein structures into holo-like conformations through simultaneous optimization of protein and ligand coordinates.
Experimental Protocols for Studying Recognition Mechanisms
Distinguishing Conformational Selection from Induced Fit
Kinetic and thermodynamic analyses can differentiate between conformational selection and induced fit mechanisms:
Stop-Flow Kinetics Protocol
- Objective: Measure binding rates under pseudo-first-order conditions with varying ligand concentrations.
- Principle: In conformational selection, the rate of complex formation depends nonlinearly on total protein concentration and linearly on the concentration of the higher-energy conformation [23]. Induced fit typically shows linear dependence on ligand concentration.
- Procedure:
- Rapidly mix protein and ligand solutions in a stopped-flow instrument.
- Monitor signal changes (fluorescence, absorbance) over time.
- Fit observed rates at multiple ligand concentrations to distinguish mechanisms.
NMR Relaxation Dispersion Techniques
- Application: Characterize weakly populated higher-energy states (as low as 1% population) in the conformational ensemble [23].
- Timescale: Sensitive to μs-ms conformational exchange processes.
- Information Content: Provides kinetic and thermodynamic parameters for exchange processes and structural constraints for higher-energy states [23].
Molecular Dynamics Simulations for Conformational Sampling
System Setup and Equilibration
- Initial Structure Preparation: Obtain protein structures from PDB or AlphaFold predictions. For calreticulin family studies, focus on the carbohydrate recognition domain (CRD) [25].
- Solvation and Ion Addition: Place the protein in an appropriate water model (TIP3P, TIP4P) and add ions to physiological concentration.
- Energy Minimization: Use steepest descent followed by conjugate gradient algorithms to remove steric clashes.
- Equilibration: Perform gradual heating from 0K to 300K with positional restraints on protein heavy atoms, followed by restraint-free equilibration.
Production Simulation and Analysis
- Trajectory Generation: Run unrestrained MD simulations for timescales sufficient to capture relevant conformational transitions (typically 100ns-1μs).
- Conformational Clustering: Use algorithms (k-means, hierarchical) to identify distinct conformational states sampled during simulations.
- Binding Affinity Calculations: Employ molecular mechanics Poisson-Boltzmann surface area (MMPBSA) or similar methods to identify favorable binding conformations [25].
- Interaction Analysis: Quantify noncovalent bonds (hydrogen bonds, hydrophobic contacts, electrostatic interactions) contributing to binding specificity.
The Scientist's Toolkit: Essential Research Reagents and Solutions
Table 3: Key Computational Tools for Molecular Recognition Studies
| Tool Category | Example Software | Primary Function | Application in Recognition Studies |
|---|---|---|---|
| Molecular Visualization | ACD/ChemSketch [28] | Chemical structure drawing and visualization | Ligand preparation and communication of structural insights |
| Docking & Screening | VLifeMDS [28], Phase [28] | Structure-based virtual screening, pharmacophore modeling | Pose prediction, lead identification through structure-based approaches |
| Structure Prediction | AlphaFold2 [27] | Protein structure prediction from sequence | Generation of apo structures for docking when experimental structures unavailable |
| Molecular Dynamics | OpenEye Scientific [29] | MD simulations, free energy calculations | Conformational sampling, binding affinity predictions |
| Specialized Analysis | CDD Vault [28] | Data management and analysis | Organization of biological and chemical data from binding studies |
| Dynamic Docking | DynamicBind [26] | Flexible protein-ligand docking | Accommodating large conformational changes during docking |
The evolving understanding of molecular recognition—from rigid lock-and-key complementarity to dynamic conformational selection and hybrid models—has profound implications for preparing protein-ligand complexes in minimization research. The historical dominance of the induced fit hypothesis is increasingly supplemented by evidence for conformational selection and population shift mechanisms across diverse biological systems [23].
For computational researchers, this paradigm shift necessitates moving beyond rigid docking approaches toward methods that explicitly account for protein flexibility and conformational ensembles. Advanced tools like DynamicBind demonstrate the feasibility of predicting ligand-specific conformations from unbound structures without requiring holo-structures or extensive sampling [26]. Similarly, integration of AlphaFold2 predictions with molecular dynamics refinements provides promising avenues for structure-based drug design, particularly for challenging targets like protein-protein interfaces [27].
The most comprehensive understanding emerges from hybrid models where initial conformational selection is followed by induced fit optimization [25]. This hierarchical mechanism leverages the advantages of both paradigms: the specificity of conformational selection and the precision of induced fit. As computational methodologies continue to advance, incorporating this nuanced understanding of molecular recognition will be essential for accelerating drug discovery and expanding the druggable genome.
Why Proper Preparation is Crucial for Accurate Binding Affinity Prediction
Accurate prediction of protein-ligand binding affinity is a cornerstone of computational drug discovery, yet its success is profoundly dependent on rigorous preparatory steps. This whitepaper details how proper preparation—encompassing protein construct design, structural data curation, and force field parameterization—serves as the critical foundation for reliable affinity predictions. When preparation is overlooked, even sophisticated computational methods fail due to issues like data bias, poor structural modeling, and inadequate sampling. By synthesizing recent advances in machine learning and molecular dynamics, this guide provides researchers with a structured framework to navigate the intricacies of system setup, thereby enhancing the accuracy and generalizability of binding affinity studies within minimization research.
In structure-based drug design, the accuracy of binding affinity predictions is inextricably linked to the quality of the initial preparation. The process of "minimization" in computational research refers to the energy optimization of a molecular system, but this optimization can only succeed if the initial structural model is biologically relevant and computationally tractable. Protein-ligand complex preparation involves a series of deliberate choices, from the initial protein construct design to the treatment of solvent molecules and ions, each of which can significantly influence the predicted binding free energy. As recent studies have highlighted, the failure of many machine learning models to generalize to novel targets often stems not from algorithmic deficiencies but from inadequate data curation and structural biases in training datasets [30].
The central thesis of this guide is that proper preparation creates a physically realistic starting point that enables accurate sampling of the protein-ligand interaction landscape. Without this foundation, predictions may appear accurate on benchmark sets due to data leakage but fail dramatically in real-world drug discovery applications. The following sections dissect the key preparatory stages, provide quantitative comparisons of different approaches, and offer detailed protocols to ensure researchers can build reliable models for minimization research.
Protein System Preparation
Construct Design and Engineering
The process begins with selecting an appropriate protein construct for study. About 75% of the over 100,000 structures in the Protein Data Bank contain ligands, reflecting their importance in structural studies [31]. However, studying full-length proteins is often unnecessary and can hinder crystallization and computational analysis.
Key Considerations for Construct Design:
- Domain Identification: Focus on the ligand-binding domain rather than the full-length protein. For example, in studying the BCR-Abl oncogene, the kinase domain (less than 300 residues) was sufficient to elucidate the inhibition mechanism of imatinib, rather than working with the full approximately 200 kDa protein [31].
- Terminal Handling: Avoid terminating constructs in the middle of secondary structure elements. Follow predicted domain boundaries and secondary structure elements to maintain structural integrity [31].
- Construct Parallelization: Empirical data from structural genomics consortia suggest designing 10-20 different protein constructs per target, as less than half typically yield samples suitable for crystallization studies [31].
Table 1: Statistical Analysis of Successful Construct Design Strategies from Structural Genomics Initiatives
| Strategy | Success Rate | Recommended Number | Key Factor |
|---|---|---|---|
| Multiple terminal variants | ~45% yield | 10-20 constructs | Variation of start/end points |
| Tag implementation | Increased purification success | 2-3 expression vectors | N/C-terminal histidine tags |
| Domain-focused design | Higher than full-length | 2-4 boundary combinations | PFAM-annotated domains |
Handling Protein Flexibility and Conformational States
Proteins are dynamic entities that sample multiple conformational states, and ligands often bind to specific ones. Traditional docking methods frequently treat proteins as rigid bodies, which severely limits their accuracy when the binding site undergoes conformational changes upon ligand binding [26].
Critical Aspects of Flexibility:
- State-Specific Binding: Many drugs bind to specific protein conformations. For example, imatinib binds to and stabilizes the inactive form of the Abl kinase [31].
- Apo-Holo Differences: AlphaFold-predicted structures often represent apo conformations that may differ significantly from ligand-bound holo states, leading to inaccurate docking results [26].
- Cryptic Pockets: Some binding sites only become apparent when the protein undergoes specific conformational changes, requiring methods that can sample these transitions [26].
Advanced methods like DynamicBind address this by employing "dynamic docking" that adjusts the protein conformation from its initial AlphaFold prediction to a holo-like state, efficiently sampling large conformational changes such as the DFG-in to DFG-out transition in kinases [26].
Data Curation and Bias Mitigation
The Data Leakage Problem in Public Databases
A critical but often overlooked preparatory step is the rigorous curation of training and benchmark data. Recent research has revealed substantial train-test data leakage between the widely used PDBbind database and the Comparative Assessment of Scoring Functions (CASF) benchmark, severely inflating the reported performance of many machine learning models [30].
Quantifying Data Leakage:
- Structural Similarity: Approximately 49% of CASF complexes have exceptionally similar counterparts in the PDBbind training set, sharing similar ligands, protein structures, and binding conformations [30].
- Performance Inflation: Models trained on standard splits can achieve competitive performance through simple memorization rather than genuine learning of protein-ligand interactions [30].
Table 2: Impact of Data Cleaning on Model Performance (CASF2016 Benchmark)
| Model | Original PDBbind R | CleanSplit R | Performance Drop | Note |
|---|---|---|---|---|
| GenScore | 0.816 (reported) | 0.683 | -16.3% | Physics-based GNN |
| Pafnucy | 0.789 (reported) | 0.652 | -17.4% | 3D CNN-based |
| GEMS | 0.831 | 0.819 | -1.4% | Proposed solution |
| Simple Search Algorithm | 0.716 | N/A | N/A | 5-nearest neighbors |
Implementing Clean Data Splits
The PDBbind CleanSplit protocol addresses data leakage through a multimodal filtering algorithm that assesses:
- Protein similarity using TM-scores
- Ligand similarity using Tanimoto scores
- Binding conformation similarity using pocket-aligned ligand RMSD
This approach removes not only direct duplicates but also complexes with similar interaction patterns, even when proteins have low sequence identity [30]. Implementing such careful splits during experimental setup is crucial for obtaining realistic performance estimates.
Molecular Representation and Feature Engineering
Input Feature Selection
The choice of input features significantly impacts model performance and interpretation. Different deep learning architectures utilize varying feature sets, each with distinct advantages:
Common Feature Categories:
- Structure-based features: 3D grids (e.g., in Pafnucy) or molecular graphs that capture spatial relationships
- Sequence-based features: Protein sequences and ligand SMILES strings (e.g., in DeepDTA, CAPLA)
- Hybrid approaches: Combine structural and sequence information (e.g., in DLSSAffinity, EGNA) [32]
Ensembling Diverse Representations
Research demonstrates that model ensembling with different feature combinations significantly improves generalization. The EBA (Ensemble Binding Affinity) method combines 13 deep learning models trained on various input feature combinations, achieving a Pearson correlation of 0.914 on the CASF2016 benchmark [32].
The most effective ensembles incorporate features that capture both short-range and long-range interactions, including a novel angle-based feature vector for direct protein-ligand interactions [32].
Force Fields and Scoring Functions
Physical Energy Functions
Physics-based scoring functions typically combine multiple energy terms using functional forms derived from force fields:
ΔG ≈ ΔHgas + ΔGsolvent - TΔS
Where:
- ΔHgas represents gas-phase enthalpy computed using force fields or neural network potentials
- ΔGsolvent accounts for solvation effects, often decomposed into polar and non-polar components
- -TΔS represents the entropic penalty upon binding [33]
Machine Learning Potentials
Recent approaches combine physical energy functions with machine learning. For example, AK-Score2 integrates three independent neural network models with physics-based scoring, demonstrating that this hybrid approach outperforms either method alone [34].
However, attempts to simply replace forcefield-based enthalpy calculations with neural network potentials have shown limitations, as relatively small percentage errors in large energy terms can produce errors far too large for meaningful binding affinity prediction [33].
Experimental Protocols and Workflows
Standardized Preparation Workflow
Diagram 1: Protein-Ligand Preparation Workflow (76 characters)
Advanced Dynamic Docking Protocol
For targets with significant flexibility, dynamic docking approaches like DynamicBind offer superior performance:
Diagram 2: Dynamic Docking Process (67 characters)
This protocol achieves a success rate of 33-39% (ligand RMSD < 2Å) on challenging test sets, significantly outperforming rigid docking methods [26].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents and Computational Tools for Binding Affinity Studies
| Tool/Reagent | Function | Application Context |
|---|---|---|
| AlphaFold2/3 | Protein structure prediction | Generating apo protein structures when experimental structures are unavailable [26] |
| RDKit | Cheminformatics toolkit | Ligand conformation generation, molecular descriptor calculation [34] [26] |
| ESM-2 | Protein language model | Extracting structural and phylogenetic information from protein sequences [35] |
| AutoDock-GPU | Molecular docking | Generating conformational decoys for training [34] |
| PDBbind Database | Curated protein-ligand complexes | Training and benchmarking affinity prediction models [35] [30] |
| CleanSplit Protocol | Data curation algorithm | Removing data leakage for robust benchmarking [30] |
| DynamicBind | Dynamic docking tool | Handling large conformational changes [26] |
| GEMS | Graph neural network | Affinity prediction with strong generalization [30] |
Proper preparation of protein-ligand complexes is not merely a preliminary step but the decisive factor in accurate binding affinity prediction. This whitepaper has systematically addressed the key preparatory stages—from intelligent construct design and rigorous data curation to appropriate molecular representation and method selection. The quantitative evidence presented demonstrates that investments in careful preparation yield substantial returns in prediction accuracy and model generalizability.
As the field advances, several emerging trends will further emphasize the importance of preparation: the integration of experimental ensembles from NMR and molecular dynamics, the development of better data curation standards, and the creation of hybrid models that leverage both physical principles and machine learning. By adopting the rigorous preparatory frameworks outlined in this guide, researchers can significantly enhance the reliability of their binding affinity predictions, ultimately accelerating the discovery of novel therapeutic agents.
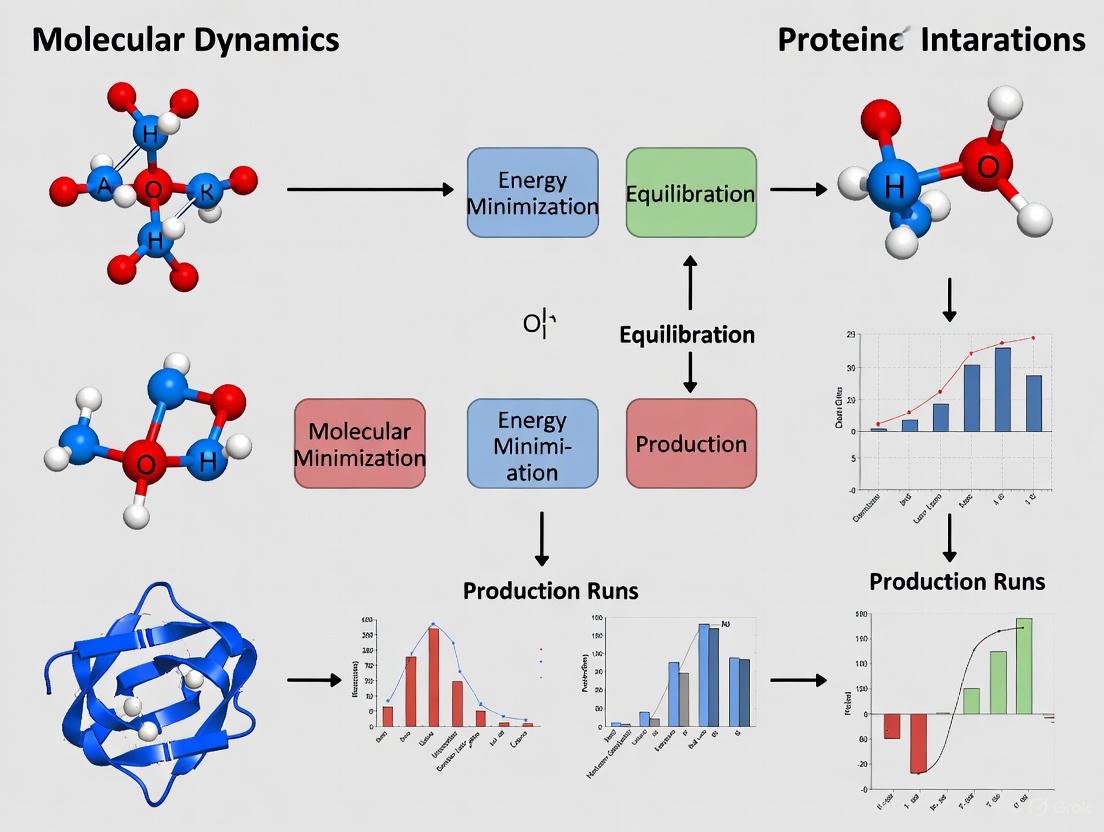
A Step-by-Step Workflow for Complex Preparation and Minimization
The Protein Data Bank (PDB) serves as the primary repository for three-dimensional structural data of proteins and nucleic acids, essential for initiating research on protein-ligand complexes. For researchers embarking on minimization studies, the initial acquisition and preparation of the system from the PDB establishes the foundation for all subsequent computational analyses. This process requires careful selection of appropriate structures, understanding of available file formats, and implementation of rigorous quality control measures to ensure the reliability of the starting structural model. The RCSB PDB provides multiple programmatic access points for downloading structural data, supporting both manual retrieval of individual structures and batch downloading for large-scale studies [36] [37].
Within the context of preparing a protein-ligand complex for minimization research, this initial step is critical because the quality of the starting structure directly influences the accuracy of energy minimization, molecular dynamics simulations, and subsequent binding affinity calculations. Current challenges in the field include addressing structural artifacts present in widely-used datasets such as PDBbind, which may contain errors in ligand bond orders, missing protein atoms, and steric clashes that can compromise computational results [12]. Recent workflows like HiQBind-WF have been developed specifically to address these concerns through systematic data cleaning and structural preparation protocols [12].
Data Acquisition Methodologies
PDB Structure Retrieval Protocols
The RCSB PDB offers multiple methods for structure retrieval, catering to different research needs. For individual structures, researchers can download files directly through HTTPS URLs using standardized naming conventions. The programmatic access service provides direct download URLs for various file formats, including PDBx/mmCIF, XML, BinaryCIF, and legacy PDB format [37]. These files can be retrieved in compressed or uncompressed forms depending on application requirements.
For large-scale studies requiring multiple structures, the batch download service is recommended. Researchers can utilize provided shell scripts to efficiently download large datasets without manual intervention [36]. The rsync protocol offers an alternative method for maintaining local copies of the entire archive or specific subsets, with scripts like rsyncPDB.sh facilitating automated synchronization [37].
The directory structure of the PDB archive organizes entries based on the middle two characters of the PDB ID. For example, the structure with PDB ID 100d is located in the directory '00' following the path pattern pub/pdb/data/structures/divided/pdb/00/ [37]. This organization system is consistent across the archive and should be understood for programmatic access.
File Format Selection Guidelines
Choosing the appropriate file format depends on the specific research application and software requirements. The table below summarizes the available formats and their appropriate use cases:
Table 1: PDB File Format Comparison for Protein-Ligand Complex Studies
| Format | Extension | Features | Recommended Use Cases |
|---|---|---|---|
| PDBx/mmCIF | .cif | Comprehensive data representation, includes rich metadata | Primary recommendation for new studies, suitable for all structure types |
| Legacy PDB | .pdb | Traditional format, limited support for large complexes | Compatibility with older software; avoid for large structures |
| PDBML/XML | .xml | Extensive metadata, machine-readable | Data mining, detailed annotation extraction |
| BinaryCIF | .bcif | Compact binary format, fast loading | Web applications, large-scale structural bioinformatics |
The PDBx/mmCIF format is particularly recommended for protein-ligand complex studies as it overcomes limitations of the legacy PDB format regarding atom count, residue types, and annotation completeness [37]. Additionally, biological assembly files should be retrieved when studying functional complexes rather than the asymmetric unit, as biological assemblies represent the physiologically relevant quaternary structure.
Structural Quality Assessment and Validation
Identifying and Addressing Common Structural Artifacts
Recent studies have highlighted significant quality issues in widely used protein-ligand datasets. The HiQBind workflow identified several categories of structural artifacts in PDBbind v2020 that necessitate correction before minimization studies [12]:
- Ligand-related issues: Incorrect bond orders, improper protonation states, and unrealistic aromaticity in ligand structures.
- Protein-related issues: Missing heavy atoms and side chains in protein residues, particularly in flexible loop regions.
- Complex-related issues: Severe steric clashes between protein and ligand atoms, and inappropriate treatment of covalently bound ligands.
These artifacts substantially impact the accuracy of scoring functions and molecular dynamics simulations. Implementation of systematic quality control measures is therefore essential during the initial acquisition phase.
Quality Assessment Protocol
The following protocol provides a systematic approach for evaluating newly acquired PDB structures:
- Experimental Validation: Consult validation reports available for each PDB entry, assessing global quality metrics including Ramachandran plot outliers, side-chain rotamer outliers, and clash scores.
- Ligand Validation: Examine electron density maps when available (from structure factors) to confirm ligand placement and geometry.
- Completeness Check: Identify missing residues or atoms in the protein structure, particularly in binding site regions.
- Biological Relevance: Verify that the biological assembly correctly represents the functional oligomeric state.
- Consistency Assessment: Cross-reference with similar structures in the PDB to identify unusual features or potential errors.
The HiQBind workflow demonstrates that automated algorithms can correct many common issues, including adding missing protein atoms with tools like ProteinFixer and correcting ligand chemistry with LigandFixer [12].
Workflow for System Acquisition and Preparation
The acquisition and preparation of protein-ligand complexes from the PDB requires a methodical approach to ensure data quality. The following workflow outlines the key decision points and processing steps:
Diagram 1: PDB Acquisition and Preparation Workflow
This workflow emphasizes the importance of quality assessment and systematic correction of structural artifacts, which aligns with recent research demonstrating significant improvements in computational results when using curated datasets [12].
Research Reagent Solutions for Structural Preparation
The following essential tools and resources facilitate effective acquisition and preparation of protein-ligand complexes from the PDB:
Table 2: Essential Research Reagents for PDB Acquisition and Preparation
| Tool/Resource | Type | Primary Function | Application in Preparation |
|---|---|---|---|
| RCSB PDB API | Web Service | Programmatic structure access | Automated retrieval of PDB entries and metadata |
| HiQBind-WF | Computational Workflow | Data cleaning and curation | Corrects structural artifacts in protein-ligand complexes |
| PyMOL | Molecular Visualization | Structure analysis and rendering | Visual quality assessment and preparation verification |
| PDBx/mmCIF Tools | Software Library | Format processing | Handles next-generation PDB file formats |
| Chemical Component Dictionary | Reference Database | Chemical ontology | Validates ligand chemistry and identity |
These tools collectively address the major challenges in initial system acquisition, from automated data retrieval through structural validation and correction. The integration of these resources enables researchers to establish robust, reproducible workflows for preparing protein-ligand complexes for minimization research.
The initial acquisition and preparation of protein-ligand complexes from the PDB represents a critical first step in minimization research that significantly influences all subsequent computational analyses. By implementing rigorous retrieval protocols, selecting appropriate file formats, conducting thorough quality assessments, and applying systematic corrections to structural artifacts, researchers can establish reliable foundation for their studies. Recent advancements in automated curation workflows like HiQBind-WF demonstrate that addressing data quality issues at this initial stage substantially improves the accuracy and reliability of computational drug discovery pipelines. As structural biology continues to generate increasingly complex data, maintaining rigorous standards for initial system preparation remains essential for advancing protein-ligand interaction research.
The accuracy of structure-based drug design projects, including molecular dynamics and binding affinity calculations, is fundamentally dependent on the quality of the initial protein model [38]. Experimentally derived structures from sources like the Protein Data Bank (PDB) frequently contain common deficiencies that, if left uncorrected, can compromise the validity of all subsequent simulations and analyses [38] [39]. This guide details the critical steps of adding missing atoms and residues, assigning correct protonation states, and optimizing the hydrogen bond network to create a reliable, all-atom protein structure suitable for minimization research. A meticulously prepared structure is not merely a preliminary step but a prerequisite for obtaining scientifically sound results in computational drug discovery [40].
Core Concepts and Quantitative Benchmarks
The Impact of Electrostatics on Stability
Electrostatic interactions, reflected in the perturbation of residue pKa values, are a significant contributor to protein stability and function [41]. The determination of macroscopic pKa values for all ionizable groups in a hyperstable ribbon–helix–helix protein via NMR spectroscopy provides a quantitative measure of these interactions. The study found that residues involved in salt bridges (four Asp and one Lys) exhibited pKa values shifted to favor the charged state, stabilizing the protein. Conversely, the pKa values for residues C40 and Y47, located in the hydrophobic dimer interface, were shifted beyond 13.7. The energy required for this large shift accounts for about two-thirds of the total protein stability, underscoring the critical importance of both electrostatic and hydrophobic interactions [41].
Benchmarking Preparation and Energy Calculations
Validating preparation workflows and subsequent energy calculations requires reliable benchmark sets. The PLA15 benchmark set, for instance, was developed to validate predictions of protein-ligand interaction energies using high-level quantum chemical methods [42]. Furthermore, the CCDC/Astex test set provides a manually curated collection of 305 protein-ligand complexes, pruned for diversity and suitability for validating interaction prediction algorithms [43].
Advanced methods for calculating binding free energies, such as dPaCS-MD/MSM (dissociation Parallel Cascade Selection Molecular Dynamics with Markov State Model analysis), have demonstrated strong agreement with experimental values across diverse systems [20]. The table below shows a benchmark of this method against experimental data for three protein-ligand complexes.
Table 1: Benchmarking Binding Free Energy Calculations using dPaCS-MD/MSM [20]
| Complex | Calculated ΔG° (kcal/mol) | Experimental ΔGexp (kcal/mol) |
|---|---|---|
| Trypsin/benzamidine | -6.1 ± 0.1 | -6.4 / -7.3 |
| FKBP/FK506 | -13.6 ± 1.6 | -12.9 |
| Adenosine A2A receptor/T4E | -14.3 ± 1.2 | -13.2 |
Methodology and Experimental Protocols
A Workflow for Comprehensive Protein Preparation
A systematic, step-by-step approach ensures all common structural issues are addressed. The following diagram outlines a robust protein preparation workflow, integrating tasks from initial import to final optimization.
Detailed Experimental Protocols
Protocol 1: Standardized Protein Preparation and Validation
This protocol can be executed manually or using automated pipelines in software suites like Schrödinger's Protein Preparation Workflow or SAMSON [38] [40].
Initial Import and Cleaning:
- Import the protein structure from a local database or the PDB website [38].
- Remove co-crystallized water molecules at your discretion, though they can be critical in some binding sites and should be evaluated carefully [38] [40].
- Strip unwanted ligands, co-factors, and monatomic ions not involved in the binding interaction of interest [40].
- Remove alternate locations by keeping only the highest-occupancy atoms to create a single, defined conformation [40].
Completing the Protein Structure:
- Add missing hydrogen atoms based on standard residue types at a specified pH (typically 7.0) or based on valence for non-standard residues [38] [40].
- Add missing heavy atoms and side chains identified in the structure [38].
- Add missing loops or residues based on SEQRES records in the PDB file, often using homology modeling or ab initio loop prediction methods integrated into tools like PDBFixer [40].
Refinement and Optimization:
- Assign protonation states for all ionizable residues (see Protocol 2) [38] [41].
- Correct flipped residues, such as the heavy atoms in arginine, glutamine, and histidine side chains [38].
- Optimize the hydrogen bond network using a systematic, cluster-based approach to reorient side-chain amides, hydroxyls, and other groups [38].
- Perform a restrained energy minimization to relax the structure. This step allows hydrogen atoms to move freely while applying restraints to heavy atoms to relieve steric clashes and strained bonds and angles without significantly altering the experimental conformation [38].
Protocol 2: Determining Protonation States of Ionizable Residues
The protonation state of residues like histidine, aspartic acid, and glutamic acid is pH-dependent and critical for modeling electrostatic interactions [41].
Experimental Determination via NMR Spectroscopy (Gold Standard):
- Monitor chemical shifts: Acquire NMR spectra of the protein across a wide pH range (e.g., 0 to 12.5) [41].
- Site-specific monitoring: Track the chemical shifts of atoms in each ionizable amino acid that are strongly dependent on the degree of protonation [41].
- Global fitting: Perform a global analysis of multiple NMR signals per residue to account for the effects of neighboring residues and determine macroscopic pKa values [41].
Computational Prediction (Common Practice):
- Use integrated preparation tools: Software like Schrödinger's Protein Preparation Wizard can automatically determine the most likely protonation states for histidine residues and other ionizable groups at a given pH [38].
- Evaluate energy penalties: These tools can also calculate the energy penalties associated with alternate protonation states, allowing researchers to make informed decisions [38].
- Manual inspection and adjustment: For critical residues in the active site, manually set the protonation state based on the local hydrogen-bonding network and literature evidence [38] [40].
The Scientist's Toolkit: Essential Research Reagents and Software
Table 2: Key Software Tools for Protein Preparation and Validation
| Tool Name | Type / Category | Primary Function in Preparation |
|---|---|---|
| Schrödinger Protein Prep | Integrated Workflow | Aggregates and automates correction of common structural problems, protonation state assignment, and optimization [38]. |
| SAMSON/PDBFixer | Structure Editing & Fixing | Removes alternates, adds missing atoms/residues, adds hydrogens, and builds solvent boxes [40]. |
| SwissDock | Docking Server | Online tool for docking that requires a pre-prepared protein target; includes basic preparation steps [44]. |
| GROMACS | Molecular Dynamics Engine | Performs energy minimization and dynamics simulations using prepared structures and force fields (AMBER, CHARMM, OPLS) [45]. |
| Antechamber/GAFF | Parameterization Tool | Generates force field parameters for small molecule ligands (HET groups) compatible with protein force fields [45] [20]. |
The meticulous preparation of a protein structure by adding missing atoms, completing residues, and assigning physiologically relevant protonation states is a non-negotiable step in the pipeline of structure-based drug design. By following the detailed workflows and methodologies outlined in this guide, researchers can transform a raw, incomplete experimental model into a robust, all-atom starting structure. This diligence ensures that subsequent computational analyses—from molecular docking and dynamics to binding free energy calculations—are built upon a solid foundation, thereby maximizing the reliability and predictive power of the research outcomes.
Ligand preparation is a critical, foundational step in the pipeline of preparing a protein-ligand complex for minimization research and structure-based drug design. The accuracy of subsequent steps—including molecular docking, molecular dynamics simulations, and free energy calculations—is profoundly dependent on the chemical and electronic correctness of the small molecule ligand. An improperly prepared ligand, with incorrect bond orders, suboptimal tautomeric states, or inaccurate partial charge distributions, can lead to flawed binding mode predictions and unreliable structure-activity relationships, ultimately misdirecting lead optimization efforts [46] [47].
This technical guide details the core aspects of ligand preparation, focusing on the correction of bond orders, the enumeration of tautomers, and the assignment of partial charges. The objective is to transform a raw, often imperfect, input ligand structure into a high-quality, biologically relevant 3D model that accurately represents the state of the molecule under physiological conditions. Adherence to the protocols described herein ensures that the ligand model is a solid foundation for rigorous computational research [48].
Core Concepts in Ligand Preparation
The process of ligand preparation addresses several key chemical concepts to ensure the model is both geometrically and electronically sound.
- Bond Orders define the connectivity and electron distribution between atoms. Correct assignment is essential for defining molecular geometry, reactivity, and interaction capabilities. Raw structural files from sources like the PDB may contain ambiguous or incorrect bond orders, which must be corrected using chemical knowledge databases [47].
- Tautomers are constitutional isomers of organic compounds that readily interconvert by the formal migration of a hydrogen atom, accompanied by a switch of adjacent single and double bonds. Common tautomeric systems include keto-enol, lactam-lactim, and amine-imine pairs. The dominant tautomer in an aqueous environment may not be the one that optimally complements the protein's binding site, making the exploration of plausible states a necessity [48].
- Partial Charges are non-integer charges assigned to atoms within a molecule, representing the uneven distribution of electron density. They are critical for accurately modeling electrostatic interactions, such as hydrogen bonding and salt bridges, with the protein target. Charges can be assigned using empirical methods, force field parameters, or quantum mechanical calculations [46] [24].
Methodologies and Experimental Protocols
A robust ligand preparation protocol is methodical and accounts for the chemical variability of small molecules. The following workflow and detailed methodologies are designed to ensure comprehensive preparation.
The following diagram illustrates the sequential workflow for a complete ligand preparation process, integrating the key procedures described in this guide:
Protocol for Correcting Bond Orders
Incorrect bond orders are a common issue in structures retrieved from databases, which can distort the ligand's geometry and electronic properties.
- Principle: Use a combination of chemical perception algorithms and curated databases to assign integer bond orders (single, double, triple, aromatic) based on atomic coordinates and connectivity.
- Detailed Procedure:
- Structure Import: Load the ligand structure file (e.g., SDF, MOL2) into the preparation software.
- Chemical Database Lookup: The software should reference a chemical database such as the Chemical Components Dictionary (CCD) to assign bond orders. A SMARTS pattern is generated for the ligand to facilitate this lookup [47].
- Valence Validation: Check for atoms with valence violations. Atoms with incorrect valences indicate potential errors in bond order assignment or protonation state.
- Aromaticity Perception: Apply an algorithm (e.g., Hückel's rule) to identify and standardize aromatic ring systems, ensuring consistent bond order and geometry.
- Manual Inspection and Curation: Visually inspect the resulting structure, particularly around metal coordination sites or unusual functional groups, to verify the automated assignments are chemically sensible.
Protocol for Generating Tautomers and Ionization States
The goal of this step is to generate a set of plausible low-energy tautomeric and protonation states that the ligand could adopt at a specified pH.
- Principle: Utilize tools like Epik to calculate the pKa values of ionizable groups and enumerate low-energy tautomers, weighting them based on their predicted population at the target pH [47] [48].
- Detailed Procedure:
- Set Simulation pH: Define the physiological pH for the system, typically pH 7.4. If the structure is from a crystallographic experiment with a known, significantly different pH, consider using the PDB pH value [47].
- pKa Prediction: The software calculates microscopic pKa values for all ionizable sites in the molecule.
- State Enumeration: Generate all possible tautomers and ionization states that are populated at the target pH. The software typically applies energy penalties to states that are less favorable.
- State Selection and Filtering: Retain all states within a specified energy window (e.g., 2-3 kcal/mol from the most stable state) for further analysis. This ensures coverage of relevant states without generating an unmanageably large number of structures.
Protocol for Assigning Partial Charges
Partial charges define the electrostatic landscape of the ligand and are crucial for modeling interactions.
- Principle: Assign atomic partial charges that are consistent with the chosen force field (e.g., OPLS4, GAFF) for subsequent molecular mechanics calculations. This can be done via pre-defined force field atom types or by calculating charges directly from the electron density.
- Detailed Procedure:
- Select Force Field: Choose the target force field for minimization and dynamics simulations (e.g., OPLS4 for Desmond MD simulations).
- Choose Charge Method:
- Force Field Assignment: The simplest method, where charges are assigned based on the ligand's atom types according to the force field's pre-parameterized library.
- Semi-Empirical Calculations: Use fast quantum mechanical methods like AM1 or PM3 to calculate the electron density and derive charges (e.g., AM1-BCC charges). This is more accurate for molecules not fully represented in the force field library.
- Quantum Mechanics (QM): For the highest accuracy, particularly for metal-binding ligands or unusual chemistries, perform DFT calculations (e.g., with Jaguar) to derive electrostatic potential (ESP)-fitted charges [49] [50].
- Charge Generation: Execute the chosen calculation to assign the partial charges to each atom of the ligand.
- Charge Validation: Check that the sum of all partial charges equals the total formal charge of the molecule.
Quantitative Data and Analysis
The following table summarizes the common methods for assigning partial charges, along with their typical applications and computational trade-offs, which are critical for planning research.
Table 1: Comparison of Partial Charge Assignment Methods
| Method | Basis of Calculation | Typical Application | Computational Cost | Key Considerations |
|---|---|---|---|---|
| Force Field Library | Pre-defined parameters based on atom type and bonding environment. | High-throughput screening, standard drug-like molecules. | Very Low | Fast but may lack accuracy for novel functional groups or complex electronic effects. |
| Semi-Empirical (e.g., AM1-BCC) | Approximate quantum mechanical calculation. | Lead optimization, general use when force field parameters are insufficient. | Low to Medium | A good balance of speed and accuracy; widely used for preparing ligands for docking and MD. |
| Quantum Mechanical (QM) DFT | Ab initio density functional theory calculation. | Metal-coordinating ligands, covalent inhibitors, final validation of key compounds. | High to Very High | Highly accurate but computationally expensive; essential for modeling polarization effects [50]. |
The Scientist's Toolkit: Essential Research Reagents and Software
Successful ligand preparation relies on a suite of specialized software tools that automate and validate the complex procedures described above.
Table 2: Key Software Solutions for Ligand Preparation
| Software Tool | Primary Function in Ligand Preparation | Key Features |
|---|---|---|
| Schrödinger LigPrep | Integrated preparation of ligands for downstream workflows. | Generates 3D structures from 1D/2D inputs; enumerates tautomers and ionization states with Epik; assigns force field-compatible charges; filters structures based on user-defined criteria [47] [48]. |
| Chemical Computing Group MOE | All-in-one molecular modeling and ligand preparation. | Structure-based drug design; includes tools for correcting bond orders and valences, protonation state assignment, and energy minimization. |
| Open Source Tools (e.g., RDKit) | Programmatic and scriptable ligand preparation. | A versatile cheminformatics toolkit; can be used to standardize molecules, generate tautomers, and calculate descriptors; requires more setup but offers high customization [26]. |
| Cresset Flare | Protein-ligand modeling with advanced electrostatic preparation. | Uses extended electron distributions (XED) for a more accurate treatment of electrostatics and lone pairs; includes QM and MM/GBSA methods for charge assignment and scoring [49]. |
Proper ligand preparation is a non-negotiable prerequisite for reliable computational research on protein-ligand complexes. By meticulously correcting bond orders, evaluating relevant tautomeric and ionization states, and assigning accurate partial charges, researchers ensure that their starting models have the highest possible fidelity. This diligence directly translates to more predictive docking results, more stable molecular dynamics simulations, and more meaningful insights during lead optimization. Neglecting this step undermines the validity of all subsequent minimization and analysis, while mastering it lays the groundwork for successful, reproducible structure-based drug discovery.
The assembly of a protein-ligand complex is a meticulously orchestrated process where additives—ions, solvent molecules, and co-factors—play indispensable roles in stabilizing the complex and modulating its biological function. Within the context of preparing a protein-ligand complex for minimization research, understanding and controlling these components is paramount, as they significantly influence the complex's conformational state, thermodynamic stability, and ultimate biological relevance. The solvent environment, particularly the presence of specific ions, is not merely a background matrix but an active participant in binding energetics. Research has demonstrated that the salt-dependence of a bimolecular association often stems from specific ion binding at functional sites rather than a general screening effect, with profound implications for interpreting binding free energies [51] [52]. Similarly, co-factors can act as essential structural or catalytic elements that lock proteins into ligand-receptive conformations. This section provides an in-depth technical guide to the strategic use of additives during complex assembly, framing them not as passive spectators but as critical determinants of success in structural studies and energy minimization protocols. The methodologies outlined herein—co-crystallization and ligand soaking—enable the precise integration of these additives into the protein-ligand complex, forming a robust foundation for subsequent computational analysis [53].
Key Additives and Their Functional Roles
Ions Ions interact with proteins through specific binding or non-specific screening effects. Specific ion binding involves direct coordination at active sites, which can competitively inhibit ligand binding or stabilize particular protein conformations. The energetics of ion-protein interactions can be substantial; for instance, studies on RNase Sa demonstrated that the observed salt-dependence of nucleotide binding was primarily due to competitive ion binding rather than electrostatic screening, with full thermodynamic profiles obtainable for these ion-protein interactions [52]. Non-specific electrostatic screening reduces the effective charge-charge interactions between a protein and ligand according to the Debye-Hückel theory, modulating binding affinity based on ionic strength.
Solvent Molecules Water molecules frequently occupy specific, well-defined positions in protein structures, often forming bridging hydrogen-bond networks between the protein and ligand. These structured water molecules can be crucial for ligand recognition and binding affinity. Displacing unstructured, high-energy water molecules from hydrophobic pockets can provide a significant thermodynamic driving force for ligand binding (hydrophobic effect). The solvent channels within protein crystals, which can constitute up to 80% of the crystal volume, allow for the free diffusion of ligands and additives to their functional binding sites [53].
Co-factors Co-factors are organic or inorganic non-protein molecules required for a protein's biological activity. Metalloenzymes require metal ions (e.g., Zn²⁺, Mg²⁺, Fe²⁺) for catalytic activity, where the metal often participates directly in the reaction mechanism. Small organic molecules, known as coenzymes (e.g., NADH, FAD, coenzyme A), act as carriers of specific functional groups or electrons, enabling catalysis that would not be possible with amino acid side chains alone.
Table 1: Quantitative Effects of Additives on Protein-Ligand Interactions
| Additive Type | Specific Example | Effect on Stability/Function | Experimental Evidence |
|---|---|---|---|
| Ions (Specific) | NaCl, K₂SO₄ binding to RNase Sa active site | Competes with nucleotide inhibitor (3'GMP) binding; increases protein melting temperature (Tₘ) | DSC and ITC showed competitive inhibition model; Tₘ increase consistent with ion binding stabilizing native state [52] |
| Ions (Screening) | Variable ionic strength buffers | Weaken charge-charge interactions via Debye-Hückel screening | Commonly observed salt-dependence of bimolecular associations [51] |
| Solvent | Structured water molecules | Forms bridging hydrogen-bond networks; contributes to binding affinity and specificity | X-ray crystallography identifies specific water positions; thermodynamic measurements quantify contributions |
| Co-factors | Metal ions (Zn²⁺, Mg²⁺), NADH, FAD | Essential for catalytic activity or structural integrity; enables reactions beyond protein capabilities | Activity assays show loss of function upon removal; structural studies identify binding motifs [54] |
Experimental Protocols for Complex Assembly with Additives
Co-crystallization with Additives
Co-crystallization involves incubating the protein with its ligand and necessary additives in solution prior to the crystallization experiment, resulting in the formation of protein-ligand complex crystals [53]. This method is particularly advantageous for capturing the correct ligand-binding position, as crystal packing tends to favor the active site [53].
Detailed Methodology:
Protein-Ligand-Incubation:
- Prepare a homogeneous mixture of the purified protein (concentrated to 5–25 mg·mL⁻¹) with a 10–1000-fold molar excess of the ligand over its dissociation constant (Kd) [53].
- Include essential additives in the mixture:
- Ions: Add salts to provide appropriate ionic strength and specific ions required for function or stability (e.g., 100 mM NaCl in phosphate buffer) [53] [51].
- Co-factors: Include necessary co-factors (e.g., metal ions, NADH) at physiologically relevant concentrations.
- Reducing Agents: Add DTT to prevent oxidation of cysteine residues if required.
- Solubilizers: Use DMSO, surfactants, or cyclodextrins to maintain ligand solubility, especially for hydrophobic compounds [53].
- Incubate the mixture on ice or at an appropriate temperature for a sufficient period (30 minutes to several hours) to ensure complex formation.
Crystallization Setup:
- Utilize the vapor diffusion method (sitting or hanging drop) in 24-well or 96-well plates.
- Mix equal volumes (e.g., 0.1–0.5 µL) of the protein-ligand-additive complex solution with the crystallization reservoir solution.
- Use commercially available or home-made crystallization screens (e.g., SG1, Morpheus II) that vary precipients (e.g., PEG), pH, and salts [53].
- For challenging systems, employ microseeding to accelerate the process. Crush existing crystals (even microcrystals or spherulites) to create a seed stock. Introduce these microseeds into new crystallization drops to bypass nucleation and directly promote growth in the metastable zone [53].
Incubation and Harvesting:
- Seal the plates and incubate at a controlled temperature (4°C or 20°C).
- Monitor crystal growth regularly.
- Once crystals reach optimal size, harvest them using cryo-loops. Soak crystals briefly in a cryoprotectant solution (e.g., reservoir solution supplemented with 20–25% glycerol) before flash-freezing in liquid nitrogen for data collection [53].
Ligand Soaking into Pre-formed Crystals
Ligand soaking involves introducing a ligand solution directly into crystals of the apo-protein (protein without ligand) [53]. This method is often preferred for its simplicity and speed but requires careful optimization of soaking conditions.
Detailed Methodology:
Apo-Crystal Preparation:
- Grow apo-protein crystals using standard vapor diffusion techniques as described above, ensuring they are of high quality and well-diffracting.
Soaking Solution Preparation:
- Prepare a soaking solution containing the ligand at a high concentration (typically a significant excess over Kd), dissolved in the crystallization reservoir solution or a closely related stabilizing buffer.
- Include necessary additives in the soaking solution:
- Stabilizing Agents: Additives like polyethylene glycol (PEG) to maintain crystal stability.
- Solubilizers: DMSO at low concentrations (e.g., 1–5%) to keep the ligand in solution.
- Specific Ions/Co-factors: If the ligand binding is dependent on specific ions or co-factors, ensure they are present in the soaking solution [53] [52].
Soaking Procedure:
- Transfer a single crystal into a small drop (2–5 µL) of the soaking solution.
- Control the soaking time precisely, as it can range from a few seconds (for small ligands and nanocrystals) to several days (for replacement soaking methods) [53].
- Monitor the crystal for signs of cracking or dissolution, which may indicate large conformational changes or suboptimal soaking conditions.
Cryoprotection and Freezing:
- After soaking, briefly transfer the crystal to a cryoprotectant solution (e.g., reservoir solution with added glycerol or MPD).
- Flash-freeze the crystal in liquid nitrogen for X-ray data collection.
Table 2: Research Reagent Solutions for Complex Assembly
| Reagent/Category | Specific Examples | Function in Complex Assembly |
|---|---|---|
| Buffers & Salts | Sodium phosphate, Sodium acetate, NaCl, KCl, (NH₄)₂SO₄ | Maintains pH and ionic strength; provides specific ions for binding or stability; can competitively inhibit ligand binding [53] [52] |
| Precipitants | Polyethylene glycol (PEG) of various molecular weights (e.g., PEG 6000), Ammonium sulfate | Drives protein crystallization by excluding volume and promoting protein-protein interactions [53] |
| Stabilizers & Reducers | Dithiothreitol (DTT), EDTA | Prevents protein oxidation (DTT); chelates metal ions to test metal dependence or prevent degradation (EDTA) [53] |
| Solubilizers | Dimethyl sulfoxide (DMSO), Surfactants, Cyclodextrins | Dissolves and maintains solubility of hydrophobic ligands in aqueous crystallization or soaking conditions [53] |
| Co-factors | NADH, FAD, Metal ions (Zn²⁺, Mg²⁺), Nucleotides (ATP, GTP) | Essential for protein function; must be included to obtain biologically relevant complex structures [54] |
| Cryoprotectants | Glycerol, Ethylene glycol, MPD | Prevents ice crystal formation during flash-freezing for X-ray data collection [53] |
Workflow Visualization: Complex Assembly Methods
The following diagram illustrates the two primary experimental pathways for assembling protein-ligand complexes, highlighting the integration of additives at critical steps.
Diagram 1: Experimental pathways for assembling protein-ligand complexes via co-crystallization and soaking, showing key decision points involving additive use.
Quality Control and Data Validation
Ensuring the integrity of the assembled protein-ligand complex is critical before proceeding to minimization research. Several biophysical and computational techniques are employed for this purpose. Dynamic Light Scattering (DLS) is used to check for protein monodispersity and the absence of aggregates in the protein-ligand-additive solution before crystallization, ensuring a homogeneous sample [53]. Differential Scanning Fluorimetry (DSF) can monitor protein stability and confirm ligand binding by observing shifts in the protein's melting temperature (Tₘ) [53]. Isothermal Titration Calorimetry (ITC) provides a full thermodynamic profile (ΔG, ΔH, TΔS) of the binding interaction, which is crucial for validating the biological relevance of the complex and can reveal the energetic contribution of specific ion binding [52].
From a structural perspective, it is essential to validate the final complex against common artifacts. Tools like the HiQBind-WF workflow can be applied to check for and correct structural issues such as steric clashes (heavy atoms closer than 2 Å), incorrect ligand bond orders, protonation states, and missing protein atoms or residues [12] [55]. For complexes obtained via soaking, careful analysis of the electron density map (e.g., a |Fₒ| - |Fₒ| omit map) is necessary to confirm proper ligand placement and occupancy without inducing detrimental crystal packing distortions [53].
Energy minimization is a critical step in the computational preparation of protein-ligand complexes, serving to relieve atomic clashes, correct distorted geometries, and identify low-energy conformations that are statistically more likely to represent native biological states [1]. The reliability of subsequent analyses, including binding pose prediction and affinity estimation, is fundamentally dependent on the physico-chemical accuracy of this refinement process. This guide provides an in-depth examination of two cornerstone components of energy minimization: the selection of appropriate force fields and the implementation of solvation models, framing them within a comprehensive protocol for preparing protein-ligand complexes for research.
The strategic importance of these choices is underscored by performance benchmarks. For instance, in large-scale docking runs on benchmark sets, force-field-based optimization can significantly shift promising conformations toward native crystal structure geometries, with complex optimizations finishing in less than 500 milliseconds on average when leveraging modern computational architectures [56]. Furthermore, the accurate treatment of solvation dramatically impacts predictive accuracy, as water molecules are essential components of protein binding sites, mediating interactions and whose displacement by ligand parts critically affects the computed free energy of binding [57].
Core Concepts: Force Fields and Solvation
Molecular Mechanics Force Fields
A molecular mechanics force field is a mathematical construct that describes the potential energy of a molecular system as a function of its nuclear coordinates. It comprises a collection of analytical functions and associated parameters that model the energy associated with bonded interactions (bond stretching, angle bending, torsion rotations) and non-bonded interactions (van der Waals forces, electrostatic interactions) [58] [59]. The general form of a typical force field energy function is:
( E{\text{total}} = E{\text{bond}} + E{\text{angle}} + E{\text{torsion}} + E{\text{vdw}} + E{\text{electrostatic}} + E_{\text{solvation}} )
Where the individual terms represent energies from bond stretching, angle bending, torsional rotations, van der Waals interactions, electrostatic interactions, and solvation effects, respectively. Parameterization of these force fields can be derived from gas-phase ab initio calculations and experimental data, aiming for a representation that is both accurate and computationally efficient [58].
Implicit vs. Explicit Solvation
The treatment of solvent is a primary differentiator among minimization protocols. Explicit solvation models treat water molecules as individual entities, capable of forming specific, directional interactions like hydrogen bonds. While accurate, this approach is computationally demanding [57]. Implicit solvation models (or continuum models), in contrast, represent the solvent as a continuous medium with bulk properties like a dielectric constant, which is computationally efficient but misses the granularity of specific water-mediated interactions [58].
Modern hybrid approaches seek to bridge this gap. For example, tools like WaterMap use molecular dynamics (MD) simulations with explicit water to statistically characterize the thermodynamic properties of hydration sites, identifying which water molecules are energetically favorable to displace with a ligand [57]. Similarly, the 3D Reference Interaction Site Model (3D RISM) applies statistical mechanics and integral equation theory to describe solvent structure around a solute, offering a compromise between the accuracy of explicit solvent and the speed of implicit models [57].
Force Field Selection for Protein-Ligand Complexes
The choice of force field dictates the physical realism of the minimization. Selecting an appropriate one requires balancing accuracy, computational cost, and system-specific features like metal ions or post-translational modifications.
Table 1: Overview of Force Fields for Protein-Ligand Minimization
| Force Field Family | Representative Examples | Key Characteristics | Best Use Cases |
|---|---|---|---|
| AMBER | AMBER03, AMBER10, AMBER14, AMBER15FB [1] | Parameters derived from high-level QM calculations; excellent for proteins & nucleic acids. | General-purpose minimization of biological complexes; often the default choice. |
| YASARA | YAMBER2, YAMBER3, YASARA2 [1] | In-house developed; demonstrated high performance in CASP challenges. | Robust minimization within the YASARA/SeeSAR environment. |
| Polarizable Force Fields (PFF) | Polarizable Force Field (PFF) [58] | Incorporates electronic polarizability, allowing charge distributions to respond to environment. | Systems where charge transfer or strong polarization is critical. |
| General-Purpose | AMBER94, AMBER96, AMBER99, NOVA, NOVA2 [1] | Well-established, extensively validated parameters. | Legacy systems or for comparison with previous studies. |
Special Considerations: Polarizable Force Fields
Standard, non-polarizable force fields use fixed atomic partial charges, which is a significant simplification. Polarizable force fields (PFFs) address this by allowing the charge distribution of the protein and ligand to change in response to their electronic environment, which is crucial when moving from gas-phase to aqueous solvation [58]. A PFF, when coupled with a continuum solvation model, has been developed explicitly for modeling protein-ligand interactions, leading to a more accurate depiction of energetics and structural features in aqueous solvents [58]. While computationally more intensive, PFFs can provide superior accuracy for systems where electronic polarization plays a key role in binding.
Solvation Models in Energy Minimization
Water is not a passive spectator but an active contributor to protein-ligand binding. Its displacement by a ligand can be a major driver of binding affinity, contributing significantly to the thermodynamic signature of binding [60] [57]. Therefore, the selection of a solvation model is not merely a technical step but a strategic one.
Table 2: Comparison of Solvation Methodologies for Energy Minimization
| Solvation Methodology | Model Type | Theoretical Basis | Advantages | Limitations |
|---|---|---|---|---|
| Continuum Solvation | Implicit | Represents solvent as a dielectric continuum [58]. | Fast computation; analytical gradients enable efficient minimization [58]. | Poor description of specific, water-mediated interactions and hydrogen-bond directionality [57]. |
| Explicit Solvent | Explicit | Models individual water molecules [57]. | Atomistic detail; captures specific water networks and bridging interactions. | High computational cost; requires extensive sampling for convergence. |
| WaterMap | Explicit/Hybrid | Statistical mechanics analysis of MD trajectories [57]. | Provides thermodynamic signature (enthalpy, entropy) of hydration sites. | Based on a rigid protein assumption; not suitable for highly flexible binding sites [57]. |
| 3D RISM | Implicit/Statistical | Integral equation theory of liquids [57]. | Faster than explicit MD; captures granular solvent effects like hydration shells. | Can be less accurate than well-converged explicit solvent simulations [57]. |
| GIST | Explicit/Hybrid | Grid Inhomogeneous Solvation Theory from MD simulations [60]. | Calculates solvation thermodynamics for entire binding sites; can be calibrated with experimental ITC data. | Performance can be target-specific, requiring training for optimal use [60]. |
Practical Application of Solvation Models
In practice, a multi-stage approach is often beneficial. An initial, rapid minimization might use an implicit solvent model to roughly refine the structure, followed by a more detailed refinement using a method that accounts for explicit waters. For instance, one can place the ligand in the binding site, temporarily tolerate clashes, and then use energy minimization with an induced-fit approach (allowing both ligand and protein to adapt) to resolve steric conflicts and create a more realistic binding mode [1]. Tools like WaterMap and 3D RISM are particularly valuable in the design phase to identify conserved, high-energy water molecules that, if displaced by a suitable ligand group, could yield a favorable gain in binding free energy [57].
Integrated Workflow for Energy Minimization
The following diagram illustrates a recommended decision workflow for applying force fields and solvation models in the energy minimization of a protein-ligand complex.
Experimental Protocols
Protocol 1: High-Throughput Minimization with GPU Acceleration
This protocol is designed for speed and scalability, ideal for virtual screening or processing large datasets of protein-ligand complexes [56].
- Structure Preparation: Input structures are first curated using a workflow like HiQBind-WF to correct common artifacts, including fixing ligand bond orders, assigning reasonable protonation states, and adding missing protein atoms [12].
- Parameter Assignment: Force field parameters are automatically assigned to the ligand using a tool like AutoSMILES, which performs pH-dependent bond order assignment and semi-empirical charge calculations [1].
- Minimization Execution: The minimization kernel is executed on a Graphics Processor Unit (GPU). The procedure can be guided by pharmacophoric constraints, such as preferred dihedral angles for rotatable bonds and key hydrogen bond interactions that should be conserved [56].
- Output: The result is a refined protein-ligand complex with relieved steric clashes and improved geometry. This entire process can be completed in under 500 ms per complex on average hardware [56].
Protocol 2: Free Energy Assessment with Hybrid Solvation
This protocol leverages solvation thermodynamics for a more detailed analysis of binding, useful for explaining SAR trends or guiding lead optimization [60] [57].
- System Setup: A curated protein-ligand complex is solvated in a box of explicit water molecules for molecular dynamics (MD) simulation.
- Hydation Site Analysis: Run a MD simulation of the apo protein (without the ligand). Analyze the trajectory with Grid Inhomogeneous Solvation Theory (GIST) or WaterMap to map the location and thermodynamic stability (free energy, enthalpy, entropy) of hydration sites in the binding pocket [60] [57].
- Complex Minimization & Scoring: Minimize the protein-ligand complex in explicit solvent. The binding free energy can then be estimated using an end-point method like MM-GBSA, where the standard desolvation term is potentially replaced or corrected by the thermodynamic data from Step 2 [57].
- Interpretation: The analysis identifies which water displacements upon ligand binding were favorable (e.g., displacing a high-energy, unstable water) and which were costly, providing a thermodynamic rationale for binding affinity and informing future chemical design.
The Scientist's Toolkit
Table 3: Essential Software and Datasets for Energy Minimization R&D
| Tool/Resource | Type | Primary Function | Application in Minimization |
|---|---|---|---|
| YASARA/SeeSAR | Software Suite | Molecular modeling, visualization, and analysis [1]. | Integrated platform for running energy minimizations with flexible/rigid backbone options and multiple force fields. |
| AutoDock Vina | Docking Software | Protein-ligand docking and scoring [61] [59]. | Provides a popular scoring function; often used for pose generation prior to minimization. |
| PLIP | Analysis Tool | Protein-Ligand Interaction Profiler [62]. | Detects and characterizes non-covalent interactions (H-bonds, hydrophobic contacts, etc.) before and after minimization to validate results. |
| HiQBind-WF | Data Curation Workflow | Semi-automated pipeline for creating high-quality protein-ligand datasets [12]. | Corrects structural errors in raw PDB files (bad bonds, protonation, clashes) to ensure a reliable starting point for minimization. |
| MISATO | ML Dataset | Dataset of ~20,000 protein-ligand complexes with QM-refined structures and MD trajectories [63]. | Provides benchmark structures with improved ligand geometry and electronic properties for force field validation. |
| WaterMap | Solvation Analysis | Calculates thermodynamic properties of hydration sites from MD [57]. | Identifies displaceable, high-energy water molecules to guide ligand design for improved binding affinity. |
| PDBbind / BioLiP | Structural Database | Curated databases of protein-ligand complexes with binding affinity data [12] [63]. | Source of experimental structures and affinities for training, testing, and validating minimization protocols. |
The meticulous preparation of a protein-ligand complex through energy minimization is a foundational step in computational structure-based research. The choices outlined in this guide—between different force field families, and among a hierarchy of solvation models—are not merely technical preferences but are consequential scientific decisions that directly impact the physical realism and predictive power of subsequent computational analyses. By adopting a reasoned approach, such as using fast, implicit solvation for initial screening and reserving more sophisticated, thermodynamics-aware methods for key complexes, researchers can efficiently navigate the trade-offs between computational cost and predictive accuracy. As the field advances with larger, higher-quality datasets and more physically accurate models, these energy minimization protocols will continue to be a cornerstone of reliable and impactful computational drug discovery.
The preparation of a protein-ligand complex for energy minimization is a foundational step in structure-based drug design, directly influencing the reliability of subsequent computational analyses such as docking and molecular dynamics simulations. This process ensures that the starting three-dimensional structure is physically realistic, chemically accurate, and suitable for computational modeling. A properly prepared system minimizes artifacts and false positives, allowing researchers to generate meaningful hypotheses about binding modes and molecular interactions. This guide provides a detailed, practical protocol for preparing protein-ligand complexes using the Molecular Operating Environment (MOE) platform, with comparative insights from other widely used software tools. The objective is to equip researchers with a reproducible workflow that enhances the structural integrity of their computational models, thereby supporting broader research efforts in drug discovery and development.
Key Concepts and Terminology
- Force Field: A mathematical function and parameter set used to calculate the potential energy of a system of atoms. MOE supports several, including MMFF94x [64].
- Energy Minimization: A computational process that adjusts atomic coordinates to find a low-energy conformation of a molecule or complex, relieving steric strain and bad contacts.
- Protonation 3D: The process of adding hydrogen atoms to a molecular structure and assigning their most probable ionization and tautomeric states at a specific pH [64] [65].
- Active Site: The region of a protein, often a pocket or cleft, where a ligand binds and chemical catalysis occurs.
- Ligand Strain Energy: The energy penalty a ligand incurs by adopting a bound conformation that is higher in energy than its global minimum [66].
The overall process of preparing a protein-ligand complex for minimization involves sequential preparation of the ligand, preparation of the protein, and finally, the treatment of the combined complex. The following diagram illustrates this high-level workflow, highlighting the key stages and decision points.
Comparative Analysis of Software Platforms
While this tutorial focuses on MOE, several other software platforms offer robust capabilities for preparing protein-ligand complexes. The choice of platform often depends on the specific research goals, available computational resources, and the need for integration with downstream applications. The following table summarizes the key features and typical use cases for several prominent tools.
Table 1: Comparison of Software Platforms for Protein-Ligand Preparation
| Software Platform | Primary Docking Method | Key Strengths | Typical Docking Time | Best Suited For |
|---|---|---|---|---|
| MOE | Induced-Fit Docking [67] | Integrated workflow from preparation to analysis; active site analysis & interaction visualization [67] [64] | Varies by system size | Academic research & lead optimization [64] |
| Schrödinger (Glide) | HTVS, SP, XP [68] | High pose prediction accuracy & database enrichment [68] | ~10 sec/compound (SP) [68] | Virtual screening of large libraries [68] |
| Rowan | AutoDock Vina with ML correction [66] | Ligand strain energy estimation using neural network potentials [66] | Slower due to post-processing [66] | Hit-to-lead optimization where strain is a concern [66] |
| UCSF DOCK | Shape-based matching & scoring [69] | Free for academics; well-documented tutorial system [69] | Varies | Academic teaching and proof-of-concept studies [69] |
Detailed Experimental Protocol in MOE
Ligand Preparation
The initial stage involves preparing the small molecule ligand, which includes building or importing its structure, ensuring proper chemistry, and optimizing its geometry.
- Load or Build the Ligand Structure: Begin by importing a ligand structure file (e.g., PDB, SDF) or using the MOE Builder to draw the ligand from scratch [64].
- Add Partial Charges: Calculate the partial charges required by the force field.
- Navigate to
Compute->Partial Charges. - Select the appropriate force field, such as
MMFF94[64].
- Navigate to
- Energy Minimization in Solvent: Solvation critically affects a ligand's bioactive conformation. Minimize the ligand in an explicit solvent shell.
- Add Solvent: Go to
Edit->Build->Water Soak. Set theSoak Modeto "Sphere" and increase theSolvent Layer Widthto 10.0 Å [64]. - Minimize the Solvated System: Use
Compute->Energy Minimize. Select theMMFF94xforce field and set the gradient convergence criterion to 0.0001. Ensure the solvation model is set to "Gas Phase" because the water molecules are explicitly present [64]. - Isolate the Minimized Ligand: Post-minimization, save only the ligand by using the
Sequence Editorto select the ligand chain and saving the "Selected" chains [64].
- Add Solvent: Go to
Protein and Complex Preparation
With the ligand prepared, the next phase is to process the protein structure and combine it with the ligand to form the complex.
- Obtain and Prepare the Protein Structure:
- Protonate the Protein:
- This critical step adds hydrogen atoms and assigns correct protonation states. Use
Compute->Protonate 3D[64]. Alternatively, theLigXcommand can be used, which performs protonation, partial charge assignment, and a limited minimization while keeping the protein side chains rigid and the ligand flexible [64].
- This critical step adds hydrogen atoms and assigns correct protonation states. Use
- Assemble and Analyze the Complex:
- Load the cleaned protein file and the solvated-minimized ligand.
- Generate a 2D ligand-protein interaction diagram via
Ligand->Ligand Interactionsto analyze interaction types like hydrogen bonds and hydrophobic contacts [64]. - Visualize the binding site's electrostatics with
Surface->Surfaces and Maps->Electrostatic Map[64].
System Minimization
The final stage involves energy minimization of the entire protein-ligand complex to relieve any residual steric clashes introduced during the preparation process.
- Set Up the Force Field: Open
Window->Potential Setup. Load theMMFF94xforce field and select an appropriate solvation model (e.g., implicit solvent). Increase the cut-off distances to 10/12 Å for better accuracy [64]. - Run Minimization: Execute
Compute->Energy Minimize. Set theGradientto 0.0001 and clickOKto initiate the minimization process [64]. - Validate the Result: Record the final potential energy and its components (
Compute->Potential Energy). Inspect the minimized structure for unreasonable geometry or unwanted atom displacements [64].
The Scientist's Toolkit: Essential Research Reagents and Software
Successful computational research relies on a suite of software tools and resources. The following table details the essential "research reagents" for preparing protein-ligand complexes.
Table 2: Essential Tools and Resources for Protein-Ligand Complex Preparation
| Tool Name | Category | Primary Function | Access Information |
|---|---|---|---|
| MOE (Molecular Operating Environment) | Integrated Software Suite | All-in-one platform for molecular modeling, simulation, and drug design [67]. | Commercial software (ChemComp.com) [67]. |
| RCSB Protein Data Bank (PDB) | Structural Database | Primary repository for experimentally-determined 3D structures of proteins and nucleic acids [64]. | Freely available at PDB.org. |
| Avogadro or ChemDraw | Chemistry Software | Used to build, edit, and optimize 2D/3D chemical structures before importing into MOE [64]. | Avogadro is open-source; ChemDraw is commercial. |
| MMFF94x | Force Field | A common force field used within MOE for energy minimization of small molecules and proteins [64]. | Bundled with MOE. |
| Chimera/UCSF ChimeraX | Visualization & Prep Tool | Prepares structures for other docking suites (like DOCK) by adding hydrogens, missing atoms, and assigning charges [69]. | Freely available for academics. |
| LigParGen | Parameter Server | Generates OPLS-AA force field parameters and charges for organic ligands for use in GROMACS and NAMD simulations [70]. | Freely available web server. |
Troubleshooting and Best Practices
Even with a detailed protocol, users may encounter challenges. The following table addresses common issues and provides solutions.
Table 3: Common Issues and Recommended Solutions
| Problem | Potential Cause | Solution |
|---|---|---|
| Unrealistically high ligand strain in docked pose [66] | Docking algorithm forces ligand into a high-energy conformation. | Use post-docking strain assessment with neural network potentials or DFT, as implemented in platforms like Rowan [66]. |
| Warning messages during protein protonation/preparation | Non-standard residues or missing atoms in the crystal structure [69]. | Use the structure cleaning tools in MOE, Chimera, or ChimeraX to mutate incomplete residues (e.g., to ALA or GLY) to resolve missing side chains [69]. |
| Poor docking results or nonsensical poses | Inaccurate protonation states or incorrect active site definition. | Verify the protonation states of key residues (e.g., His, Asp, Glu) in the active site. Manually define the docking site based on a co-crystallized ligand. |
| Minimization fails to converge | System is too large, or the initial structure has severe steric clashes. | Increase the number of minimization iterations. For severe clashes, consider a multi-stage minimization, relaxing positional restraints gradually. |
Solving Common Preparation Errors and Optimizing for Performance
Identifying and Resolving Severe Steric Clashes and Voids
The preparation of a protein-ligand complex for minimization research represents a foundational step in computational structural biology and drug discovery. Within this process, identifying and resolving severe steric clashes and voids is paramount, as these structural artifacts can compromise the physiological relevance of computational models and lead to misleading results in downstream applications. Steric clashes, characterized by unphysical overlap of non-bonding atoms, introduce substantial energetic strain, while voids indicate suboptimal packing that fails to recapitulate the complementarity observed in native complexes [71]. These issues are particularly prevalent in homology models, low-resolution structures, and computationally generated complexes where approximate positioning of atoms is employed.
The broader thesis of preparing a protein-ligand complex for minimization research hinges on creating a structurally sound starting model that faithfully represents the physical system without artificial strain. This foundation enables subsequent energy minimization and molecular dynamics simulations to explore biologically relevant conformational states rather than attempting to recover from physically implausible starting conditions. The process of clash identification and resolution thus serves as a critical gatekeeper for ensuring the validity of computational structural biology workflows, directly impacting the reliability of virtual screening, binding mode prediction, and free energy calculations in drug discovery pipelines [72] [26].
Quantitative Definitions and Detection of Steric Artifacts
Defining Steric Clashes and Voids
A rigorous quantitative definition of steric clashes is essential for consistent identification and resolution. The Chiron protocol defines a steric clash as any atomic overlap resulting in Van der Waals repulsion energy greater than 0.3 kcal/mol (approximately 0.5 kBT), with specific exceptions for bonded atoms, disulfide bonds, hydrogen-bonded heavy atoms, and certain backbone interactions [71]. This energy-based threshold provides a more physically meaningful assessment than simple distance cutoffs, as it accounts for the varying repulsive contributions from different atom types.
The clash-energy of a protein structure is calculated as the sum of Van der Waals repulsion energy of all clashes present. To normalize for protein size, researchers define a clash-score as the clash-energy divided by the number of contacts examined, providing a standardized metric for comparing steric strain across different structures [71]. Statistical analysis of high-resolution crystal structures has established that an acceptable clash-score threshold is 0.02 kcal·mol⁻¹·contact⁻¹, representing approximately one standard deviation above the mean for quality experimental structures [71].
Voids represent the complementary problem—regions where packing fails to achieve the density observed in native structures. While less quantized than clashes, voids can be identified through computational assessment of packing density and computational solvent mapping, which reveals regions that should be occupied by protein atoms or structured water molecules but remain accessible to solvent probes.
Statistical Basis from High-Resolution Structures
Analysis of high-resolution protein structures provides the statistical foundation for distinguishing problematic artifacts from naturally tight packing. The distribution of clash-scores across 4,311 unique high-resolution chains reveals the permissible level of Van der Waals repulsion as a consequence of optimal biological packing [71]. This distribution establishes the benchmark against which model quality can be assessed, with deviations indicating potential refinement issues.
Table 1: Clash-Score Distribution in High-Resolution Protein Structures
| Statistical Measure | Clash-Score Value (kcal·mol⁻¹·contact⁻¹) | Interpretation |
|---|---|---|
| Mean | ~0.01 | Typical well-refined structure |
| Acceptable threshold | 0.02 | 1 standard deviation above mean (covers ~84% of high-quality structures) |
| Problematic threshold | >0.02 | Indicates likely refinement artifacts |
The same analysis demonstrated that low-resolution structures (2.5-3.5 Å) and homology models exhibit significantly worse clash-scores than high-resolution structures, confirming the need for specialized refinement protocols for these model types [71].
Detection Methodologies and Tools
Energy-Based Detection Protocols
The Chiron protocol employs a grid-based search algorithm to efficiently identify atomic overlaps while restricting computational cost. The method constructs a grid around the protein with cell dimensions larger than the largest Van der Waals interaction distance (~4.5 Å), then systematically checks for clashes between atoms in the same or adjacent cells [71]. This approach reduces search time and space by leveraging the local nature of steric artifacts. Van der Waals repulsion energy is calculated using non-bonded parameters from the CHARMM19 force field, which provides physically realistic repulsive potentials [71].
Specialized Software Implementations
Multiple software tools have been developed specifically for clash detection, each with distinct capabilities:
DSSR automatically checks for steric clashes in coordinate files and reports the minimum distance between heavy atoms of potentially clashing residues. This approach proved valuable in assessing predicted RNA models during CASP16, where severe clashes sometimes prevented structural analysis [73].
MolProbity and WHAT_CHECK employ distance-based clash criteria, typically using a cutoff of 0.4 Å overlap between Van der Waals radii. While simpler than energy-based approaches, these tools provide rapid assessment and have been widely adopted in structural biology pipelines [71].
DynamicBind incorporates clash assessment as part of its comprehensive docking evaluation, using both ligand RMSD and clash scores to determine prediction success. This integrated approach recognizes that even accurate pose predictions with severe clashes have limited utility in structure-based drug design [26].
Resolution Protocols and Methodologies
Discrete Molecular Dynamics (DMD) Approaches
The Chiron protocol utilizes Discrete Molecular Dynamics (DMD) simulations with CHARMM19 non-bonded potentials, EEF1 implicit solvation parameters, and geometry-based hydrogen bond potentials to resolve clashes with minimal backbone perturbation [71]. DMD employs square-well potentials instead of continuous potentials, solving ballistic equations of motion rather than Newtonian equations. This approach enables more rapid exploration of conformational space while maintaining physical realism.
The DMD minimization protocol employs temperature control through Anderson's thermostat with velocity rescaling rates of either 200 ps⁻¹ or 4 ps⁻¹ depending on the simulation stage [71]. The time unit of all-atom DMD simulations is approximately 50 femtoseconds, enabling efficient sampling while maintaining atomic-level precision. Benchmark studies demonstrate that this approach efficiently resolves severe clashes in low-resolution structures and homology models that challenge conventional minimization methods [71].
Molecular Mechanics Minimization
Traditional steepest descent or conjugate gradient minimization using all-atom molecular mechanics force fields represents the most widely used method for resolving clashes. The implementation in GROMACS using CHARMM force fields can be effective, particularly when combined with molecular dynamics simulations [71]. A typical protocol involves:
- Initial conjugate gradient minimization for 1000 steps with convergence criteria of maximum force < 200 kJ·mol⁻¹·nm⁻¹
- If clashes persist, 2 ps MD simulation at 240 K
- Equilibrium MD simulation for 2 ps at 300 K
- Use of OBC implicit solvation without cutoffs [71]
This approach benefits from rigorous physical potentials but may struggle with severe clashes that require more substantial conformational rearrangements.
Knowledge-Based and Advanced Sampling Methods
Rosetta employs knowledge-based potentials and small backbone moves to resolve clashes, particularly effective for smaller proteins (<250 residues) [71]. The "fast relax" protocol with 4 relaxation repeat steps per cycle or "constrained relax" with fixed backbone coordinates can be employed, typically running 100 independent iterations with unique random seeds to ensure adequate sampling [71].
DynamicBind represents a recent advance using deep equivariant generative models to construct smooth energy landscapes that promote efficient transitions between equilibrium states. This approach can recover ligand-specific conformations from unbound protein structures without extensive sampling, effectively resolving clashes through biologically relevant conformational changes [26].
Integration in Protein-Ligand Complex Preparation
Workflow for Comprehensive Structure Preparation
A robust workflow for preparing protein-ligand complexes incorporates clash identification and resolution at multiple stages to ensure structural integrity throughout the preparation process.
Method Selection Guidance
The optimal approach for resolving steric clashes depends on their severity and the system characteristics. Comparative studies inform method selection based on specific scenarios:
Table 2: Resolution Methods for Different Scenarios
| Scenario | Recommended Method | Performance Notes |
|---|---|---|
| Severe clashes in homology models | Chiron (DMD) | Robust where molecular mechanics fails [71] |
| Moderate clashes in experimental structures | Molecular mechanics minimization | Efficient for local adjustments [71] |
| Small proteins (<250 residues) | Rosetta relaxation | Knowledge-based potentials effective [71] |
| Large conformational changes needed | DynamicBind | Handles DFG-in/out transitions, cryptic pockets [26] |
| Integration with MD workflows | GROMACS minimization | Compatible with subsequent simulation [71] |
Assessment of Resolution Success
Effective clash resolution must balance steric alleviation with structural integrity. Successful resolution should achieve:
- Clash-score below 0.02 kcal·mol⁻¹·contact⁻¹ [71]
- Minimal backbone perturbation (typically <0.5 Å RMSD)
- Preservation of secondary structure elements
- Maintenance of binding pocket geometry
- Physically realistic side-chain rotamers
The refinement should be evaluated against both geometric quality metrics and functional relevance, particularly for ligand-binding sites where subtle distortions can significantly impact apparent affinity.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Research Reagent Solutions for Clash Identification and Resolution
| Tool/Resource | Type | Primary Function | Application Context |
|---|---|---|---|
| Chiron | Automated web server | Identifies and resolves clashes using DMD | homology models, low-resolution structures [71] |
| DSSR | Nucleic acid structure analysis | Detects steric clashes in nucleic acids | RNA/DNA structures and complexes [73] |
| MolProbity | Structure validation | Quality control including clash detection | Experimental structure determination [71] |
| GROMACS | Molecular dynamics | Energy minimization and MD simulation | Physics-based refinement [71] [74] |
| Rosetta | Protein modeling | Knowledge-based refinement | Small protein refinement [71] |
| DynamicBind | Deep generative model | Dynamic docking with conformational adjustment | Ligand-specific conformation prediction [26] |
| CHARMM-GUI | Web-based interface | System setup for MD simulations | High-throughput refinement pipelines [72] |
| AutoDock Vina | Molecular docking | Pose prediction and scoring | Initial complex generation [72] |
Advanced Considerations and Future Directions
Integration with AI-Based Structure Prediction
The emergence of AlphaFold2 and related AI-based structure prediction methods has transformed structural biology but introduced new challenges for steric assessment. AF2-predicted structures often differ from ligand-bound conformations, creating potential clashes when docking ligands to apoprotein predictions [26] [27]. DynamicBind addresses this by simultaneously adjusting protein conformation and ligand placement, effectively resolving clashes through biologically realistic conformational changes [26]. This approach demonstrates the growing integration between prediction and refinement, where clashes are not merely artifacts but indicators of incorrect conformational states.
High-Throughput Applications
Advances in computational power and methodology now enable clash resolution in high-throughput pipelines. The implementation of automated workflows using CHARMM-GUI for system preparation enables batch processing of multiple protein-ligand complexes [72]. These pipelines can incorporate MD simulation as a refinement step, with ligand stability during simulation (assessed through RMSD) serving as a proxy for binding mode validity [72]. This approach has demonstrated 22% improvement in ROC AUC compared to docking alone, highlighting the value of incorporating physical refinement into virtual screening [72].
Method Selection Framework
Choosing the appropriate method requires consideration of multiple factors, which can be conceptualized as a decision pathway:
The identification and resolution of severe steric clashes and voids represents a critical component in preparing protein-ligand complexes for minimization research. By applying quantitative definitions derived from high-resolution structures, employing specialized detection methodologies, and implementing appropriate resolution protocols, researchers can ensure the physical realism of computational models. The continuing development of methods like DMD-based refinement and deep generative models addresses increasingly challenging cases, including large conformational changes and cryptic pocket opening. As structural biology continues to evolve toward more complex systems and integrative modeling, robust clash identification and resolution will remain essential for generating biologically meaningful computational models that reliably advance our understanding of protein-ligand interactions and facilitate drug discovery.
Addressing Incorrect Ligand Protonation States and Tautomers
Preparing a protein-ligand complex for molecular minimization is a critical step in structure-based drug design. The accuracy of this preparation, particularly in assigning the correct ligand protonation states and tautomers, directly determines the reliability of subsequent energy calculations and binding mode predictions. This guide details the core concepts, methodologies, and tools essential for addressing these conformational uncertainties within the context of minimization research.
The calculation of hydrogen positions is a common preprocessing step when working with crystal structures of protein-ligand complexes, as an explicit description of hydrogen atoms is generally needed to analyze binding modes or calculate associated binding energies [75]. Due to insufficient resolution, the vast majority of entries in the Protein Data Bank (PDB) only contain coordinates of non-hydrogen atoms, necessitating automated procedures for the placement of hydrogen atoms [75]. This problem is non-trivial due to the large number of degrees of freedom resulting from different chemical moieties and a high degree of mutual dependence.
Protonation states refer to the specific arrangement of protons around ionizable sites in a molecule, which can change with the pH of the environment [76]. Tautomers are structural isomers that interconvert by the migration of a proton, often accompanied by a rearrangement of single and double bonds [77]. The most common form is prototropic tautomerism, such as keto-enol equilibria [77]. For a minimization protocol, an incorrect initial assignment can lead to several critical failures:
- Erroneous Electrostatic Potentials: The forces governing the minimization are derived from a molecular mechanics force field. An incorrect charge state fundamentally misrepresents the electrostatic component of the interaction energy.
- Non-optimal Hydrogen Bonding: A protonation or tautomeric state that is unstable in the binding site can prevent the formation of key hydrogen bonds or introduce unfavorable interactions during energy minimization.
- Conformational Trapping: The minimization algorithm may converge to a local energy minimum that is structurally unrealistic, leading to incorrect conclusions about the binding mode.
Experimental evidence strongly suggests that protonation states frequently change from the unbound to the bound state, and that most binding events are pH-dependent, implying a net uptake or release of protons [78]. Furthermore, analyses indicate that approximately 29% of drugs exhibit tautomerism, underscoring the prevalence of this phenomenon in pharmaceutical compounds [77].
Key Concepts and Impacts
The Role of Protonation States in Receptor-Ligand Interactions
Electrostatics is a dominant force in the initial stages of receptor-ligand recognition and binding. The protonation state of a molecule dictates its charge distribution, which in turn governs long-range electrostatic steering and short-range interaction patterns [78]. The formation of a complex can substantially alter the microenvironment of an ionizable group, shifting its pKa and stabilizing a protonation state that is different from its preferred state in solution [78]. For example, a study on the ERα nuclear receptor binding to its DNA response element showed that binding was coupled to proton uptake, suggesting that two ionizable residues at the interface changed their protonation states upon complex formation [78].
Consequences of Tautomerism on Drug Properties and Binding
Tautomerism can significantly influence a drug's physicochemical properties, its interaction with the biological target, and its pharmacokinetic profile [79] [77]. Identifying the different tautomers of drugs and exploring their importance in the mechanisms of drug action are integral components of current drug discovery [77].
Pharmacodynamics: The active tautomer is responsible for the desired pharmacological effect. For instance, the antibiotic erythromycin exists in three tautomeric forms (one ketone and two cyclic hemiketals), but only the ketonic form is active and recognized by ribosomes to inhibit protein synthesis [79]. Dosing must account for the presence of inactive tautomers.
Pharmacokinetics: Tautomerism can affect a molecule's absorption, distribution, metabolism, and excretion (ADME). For example, different tautomers of a drug can exhibit varying solubility, membrane permeability, and metabolic rates [79]. Warfarin, an anticoagulant with over 40 possible tautomers, demonstrates how different forms may be metabolized differently, directly impacting its pharmacodynamic effect [79].
Table 1: Impact of Tautomerism on Drug Properties with Examples
| Aspect | Impact of Tautomerism | Example Drug |
|---|---|---|
| Drug Action (Pharmacodynamics) | Different tautomers can have vastly different potencies and efficacies. | Erythromycin (only ketone tautomer is active) [79] |
| Metabolism | Specific tautomers may be preferentially metabolized, leading to drug-drug interactions or detoxification. | Lapatinib (nitroso tautomer leads to mechanism-based inhibition) [79] |
| Distribution | Tautomers can exhibit different binding affinities to serum proteins like Human Serum Albumin (HSA). | Warfarin (ionized open-chain tautomer binds to HSA) [79] |
| Solid-State Properties (Preformulation) | Leads to desmotropy (tautomeric polymorphism), where different solid forms have different physicochemical properties. | Piroxicam (exists in zwitterionic form in the solid state) [77] |
Computational Methodologies and Tools
A robust chemical model is needed to describe effects such as tautomerism and ionization consistently [75]. Computational tools for predicting protonation states and tautomers generally fall into three categories: empirical rule-based, machine learning (ML)-based, and physics-based methods.
Empirical and Machine Learning Methods: Tools like Epik (Schrödinger) use rapid approaches based on linear free-energy relationships (Hammett-Taft LFER) or graph convolutional neural networks (GCNNs) to predict micro-pKa values and enumerate the most probable protonation states and tautomers [76]. These are fast and suitable for high-throughput screening but are typically agnostic to 3D geometry and stereochemistry [76].
Physics-Based Methods: Tools like Jaguar pKa and Macro-pKa (Schrödinger) use density functional theory (DFT) to calculate the energy of different states, taking conformational, geometric, and stereochemical effects into account [76]. These methods are more accurate but computationally expensive and best suited for lead optimization stages [76].
Integrated Protein-Ligand Methods: The Protoss tool represents a holistic approach that predicts tautomers and protonation states for both the protein and ligand simultaneously within the context of the complex [75]. It generates the most probable hydrogen positions based on an optimal hydrogen bonding network using an empirical scoring function, considering the mutual dependence between the protein and ligand states [75].
Comparative Analysis of Software Solutions
Table 2: Comparison of Software Tools for State Prediction and Handling
| Tool / Solution | Primary Methodology | Key Features | Typical Use Case |
|---|---|---|---|
| Epik Classic [76] | Empirical (Hammett-Taft LFER) | Fast, SMARTS-based rules. | High-throughput ligand preparation for docking/screening. |
| Epik 7 [76] | Machine Learning (GCNN) | Improved accuracy, broad chemical space. | Idea generation, hit-to-lead optimization, property prediction. |
| Jaguar pKa [76] | Physics-Based (DFT) | Considers 3D geometry and stereochemistry. | Accurate pKa prediction for non-tautomerizable ligands. |
| Macro-pKa [76] | Physics-Based (DFT) | Handles tautomerizable systems, enumerates states. | Early-stage lead optimization for complex molecules. |
| Protoss [75] | Empirical Scoring Function | Holistic treatment of protein & ligand in complex. | Pre-processing of PDB structures for analysis/minimization. |
| PDB2PQR [80] | Empirical pKa prediction (PROPKA) | Prepares PDB files, assigns protonation states at user pH. | Pre-processing for simulation setups (e.g., with CHARMM-GUI). |
Experimental Protocols for State Validation
While computational prediction is a powerful first step, experimental validation is crucial for confirming the biologically relevant protonation and tautomeric states.
Spectroscopic Techniques
A variety of spectroscopic methods are employed to explore tautomerism in experimental studies [77].
- Nuclear Magnetic Resonance (NMR): NMR is a premier technique for studying tautomerism in solution [77]. Chemical shifts and coupling constants are highly sensitive to the electronic environment of atoms, allowing for the identification and quantification of different tautomers in equilibrium. For instance, studies on albendazole desmotropes use solution and solid-state NMR to determine the dominant tautomeric forms [77].
- Vibrational Spectroscopy: Infrared (IR) and Raman spectroscopy probe molecular vibrations. The presence of characteristic functional groups (e.g., C=O vs. C-O-H in keto-enol tautomers) produces distinct spectral signatures [77]. Terahertz spectroscopy has also emerged as a useful tool for investigating tautomeric forms in the solid state [77].
- X-ray Crystallography: This is the best method for directly identifying the solid-state structure of a tautomer, or "desmotrope" [77]. High-resolution X-ray diffraction can pinpoint the positions of heavy atoms, allowing the bonding pattern and, by inference, the proton positions to be determined. Neutron diffraction can provide even more direct evidence for hydrogen atom positions [77].
Isothermal Titration Calorimetry (ITC)
ITC is a valuable biophysical technique for studying binding events. Since changes in protonation states often involve an exchange of protons with the buffer, they are accompanied by heat changes (enthalpy). By performing ITC experiments in buffers with different ionization enthalpies, one can determine if protonation changes are linked to binding [78]. This method was used, in conjunction with modeling, to demonstrate that the binding of the ERα nuclear receptor to DNA is coupled to proton uptake [78].
A Practical Workflow for Minimization Preparation
Integrating the above principles into a standardized protocol ensures that a protein-ligand complex is correctly prepared for energy minimization. The following workflow, summarized in the diagram below, provides a robust pathway.
The core challenge is the interdependency between the protein, ligand, and solvent. A holistic approach that considers all degrees of freedom simultaneously is required to find the global optimum for the hydrogen bonding network [75]. The workflow involves:
- Structure Preprocessing: The input PDB structure must be processed to add missing atoms, identify disulfide bonds, and correctly assign bond orders and atom types using a consistent chemical model [75] [80].
- State Enumeration: For both the protein and the ligand, all possible alternative protonation states, tautomers, and rotatable hydrogen orientations are enumerated [75]. This is a critical step, as neglecting ligand degrees of freedom can lead to erroneous predictions [75].
- Global Optimization: An empirical scoring function is used to evaluate the quality of the hydrogen bonds and the stability of the chemical groups across the entire complex [75]. The configuration that maximizes the overall score of the hydrogen bonding network is selected.
- Final Minimization: With the hydrogen bonding network fixed, a force-field based minimization is performed. This step can incorporate side chain flexibility in the protein's active site to relax the structure and resolve any minor steric clashes, resulting in a stable, energy-minimized complex [56].
The Scientist's Toolkit: Essential Research Reagents and Software
Table 3: Key Software Tools and Resources for Handling Protonation and Tautomerism
| Tool / Resource | Function / Purpose | Access / Notes |
|---|---|---|
| PDB2PQR Server [80] | Prepares PDB files for simulations; assigns protonation states for protein residues at a given pH using PROPKA. | Web server. |
| Protoss [75] | Holistically predicts tautomers and protonation states in protein-ligand complexes by optimizing H-bond networks. | Integrated into the BioSolveIT software suite. |
| Epik [76] | Rapidly predicts pKa and generates most populated protonation states and tautomers for drug-like molecules. | Commercial (Schrödinger). |
| Jaguar pKa [76] | Provides high-accuracy, DFT-based pKa predictions for ionizable sites, accounting for 3D geometry. | Commercial (Schrödinger). |
| CHARMM-GUI [80] | A web-based platform that prepares complex molecular systems for simulation, often using PDB2PQR output. | Web server. |
| AMBER [81] | A suite of biomolecular simulation programs that includes tools for molecular dynamics and energy minimization. | Freely available & commercial. |
| APBS [81] | Solves the equations of continuum electrostatics for biomolecular complexes, sensitive to protonation states. | Freely available. |
Correctly addressing ligand protonation states and tautomers is not merely a preliminary step but a foundational determinant of success in preparing a protein-ligand complex for minimization. Errors introduced at this stage propagate through subsequent calculations, compromising the validity of the resulting structural and energetic insights. By leveraging a combination of sophisticated computational tools, a holistic workflow that considers the entire complex, and experimental validation where possible, researchers can significantly enhance the accuracy and predictive power of their structure-based drug discovery efforts. A meticulous approach to this preparatory phase ensures that minimization research is conducted on a structurally and electrostatically realistic model, leading to more reliable conclusions about ligand binding and function.
Optimizing Force Field Parameters and Cutoff Values for Your System
Accurate prediction of protein-ligand interactions is fundamental to computational drug discovery, with molecular docking serving as a pivotal tool in structure-based drug design [24]. The reliability of these computational approaches hinges on the underlying molecular force fields—physics-based energy functions that describe the potential energy of a system as a function of its atomic coordinates. Despite significant advances in force field parameterization, achieving a consistent balance of molecular interactions that stabilize folded proteins while accurately capturing the conformational dynamics of protein-ligand complexes remains challenging [82]. This technical guide provides researchers with a comprehensive framework for optimizing force field parameters and cutoff values specifically for preparing protein-ligand complexes for minimization research, ensuring more accurate and reliable computational outcomes in drug discovery pipelines.
Force field optimization requires careful consideration of multiple energy components and their parameterizations. Modern force fields typically incorporate terms for bonded interactions (bonds, angles, dihedrals) and non-bonded interactions (electrostatics, van der Waals) [83] [82]. The balance between these terms, particularly between protein-protein and protein-solvent interactions, is essential for accurate modeling of biomolecular systems [82]. Furthermore, the treatment of non-bonded interactions through distance cutoffs introduces potential artifacts that must be carefully managed through appropriate cutoff selection [84]. This guide addresses these challenges through systematic parameter optimization and validation protocols tailored to protein-ligand systems.
Force Field Components and Optimization Targets
Fundamental Energy Terms
Molecular mechanics force fields describe the potential energy of a system through multiple component terms. The generalized energy function can be represented as:
Etotal = Ebonded + Enon-bonded
Where the bonded terms include:
- Bond stretching: Ebond = ∑kb(r - r0)2
- Angle bending: Eangle = ∑kθ(θ - θ0)2
- Torsional rotations: Edihedral = ∑kφ[1 + cos(nφ - δ)]
The non-bonded terms encompass:
- Van der Waals interactions: Typically modeled with Lennard-Jones potential: ELJ = 4ε[(σ/r)12 - (σ/r)6]
- Electrostatic interactions: Modeled with Coulomb's law: Eelec = (qiqj)/(4πε0εr)
- Implicit solvation effects: Accounting for solvent-mediated interactions [83]
- Hydrogen bonding: Explicit terms in some force fields [83]
The RosettaGenFF energy model exemplifies this approach, integrating ELennard-Jones, ECoulomb, EHydrogen-bond, EImplicit-Solvation, and EGeneric-Torsion terms with atomic parameters defined for Lennard-Jones and implicit solvation following generic atom types [83].
Key Optimization Parameters
Table 1: Key Force Field Parameters for Optimization
| Parameter Category | Specific Parameters | Optimization Approach |
|---|---|---|
| Non-bonded interactions | Lennard-Jones ε and σ parameters, partial atomic charges | Crystal lattice discrimination, binding affinity matching [83] [85] |
| Torsional potentials | Dihedral force constants (kφ), multiplicities (n), phases (δ) | Quantum mechanical calculations, conformational sampling [83] [82] |
| Solvation parameters | Atomic solvation energies, volume corrections | Hydration free energy matching, osmotic pressure data [82] |
| Protein-water interactions | Van der Waals scaling factors | Comparison with SAXS/NMR data, folded protein stability [82] |
Parameter Optimization Strategies and Methodologies
Crystal Lattice-Guided Optimization
Small molecule crystal structures provide a rich source of information for guiding molecular force field development. The fundamental premise of this approach is that experimentally determined molecular lattice arrangements must have lower energy than all alternative lattice arrangements [83]. This methodology involves:
Decoy Structure Generation: Generating thousands of alternative "decoy" lattice packing and conformational arrangements for small molecules with known crystal structures using Metropolis Monte Carlo with minimization (MCM) search. This protocol samples space groups, lattice parameters, rigid-body orientations, and internal conformations of each small molecule [83].
Parameter Optimization: Simultaneously optimizing force field parameters to maximize the energy gap between experimentally observed lattice structures and sampled alternative arrangements. The dualOptE algorithm or similar approaches can be used for this purpose, with optimization targeting 175+ non-bonded parameters for a generalized implicit solvent force field and 250+ torsion parameters [83].
Cross-validation: Validating optimized parameters on independent test sets of crystal structures not included in training. The success of this approach is demonstrated by improved performance in bound structure recapitulation in cross-docking, with solutions within <1 Å in over half of cases [83].
Charge Optimization with Explicit Solvent
Accurate partial charge assignment is crucial for modeling electrostatic interactions. Traditional implicit solvent approaches suffer from limitations in dealing with interfacial water molecules [85]. An explicit solvent alchemical free-energy method optimizes ligand partial charges to maximize binding affinity:
System Preparation: Prepare protein-ligand complexes using standard protocols (e.g., pdbfixer for adding missing side chains, Antechamber with GAFF2 for ligand parameters, AM1-BCC for partial charges) [85].
Alchemical Transformations: Apply single-step perturbation (SSP) theory to calculate free energy differences between unperturbed and perturbed states in both bound and unbound states:
ΔGunperturbed→perturbed = -kT ln⟨exp(-(Eperturbed - Eunperturbed)/kT)⟩unperturbed
where Eperturbed and Eunperturbed are potential energies calculated using perturbed and unperturbed Hamiltonians [85].
Constrained Optimization: Optimize partial charges (qi) to minimize ΔGbinding(qi) subject to constraints:
- Net molecular charge conservation
- Root-mean-square deviation limit from original charges
- Maximum perturbation per atom per iteration (e.g., 0.01e) [85]
This approach has successfully identified beneficial chemical modifications such as pyridinations, fluorinations, and oxygen-to-sulfur mutations, with three-quarters of predicted changes improving binding affinity by approximately 1 kcal/mol on average [85].
Balanced Protein-Water Interactions
Modern force field development aims to balance protein-protein and protein-solvent interactions to accurately model both folded and disordered states. Two refinement strategies have shown particular promise:
Selective Protein-Water Interaction Scaling: Upscaling protein-water van der Waals interactions (e.g., in ff03w-sc) improves the stability of folded proteins while maintaining accurate intrinsically disordered protein (IDP) ensembles. This approach addresses the tendency of some force fields to destabilize folded domains while correctly modeling IDPs [82].
Targeted Torsional Refinements: Specific adjustments to backbone torsional potentials (e.g., for glutamine in ff99SBws-STQ′) correct overestimated helicity in polyglutamine tracts while maintaining accuracy for other sequence types. This approach preserves the balance between different secondary structure propensities [82].
Validation of these refinements requires extensive comparison with experimental data including small-angle X-ray scattering (SAXS) for chain dimensions and nuclear magnetic resonance (NMR) spectroscopy for secondary structure propensities, while ensuring stability of single-chain folded proteins and protein-protein complexes over microsecond-timescale simulations [82].
Diagram Title: Force Field Optimization Workflow
Cutoff Value Optimization and Artifact Mitigation
Cutoff-Related Artifacts in Energy Landscapes
The truncation of non-bonded interactions at specific cutoff distances introduces artificial ruggedness into protein-protein energy landscapes, creating false minima and altering the positions and shapes of true conformation-based minima [84]. This artificial ruggedness manifests as deviations in relative energy changes from their values at asymptotically large cutoffs, impeding the search for global minima in protein docking and introducing errors in binding free energy calculations [84]. The amplitude of this artificial ruggedness decreases with increasing cutoff values, making the selection of appropriate cutoffs critical for accurate simulations.
The relative energy change δE(R)/E(R) asymptotically approaches zero for potential functions with n ≤ 4 (where n is the power in 1/rn potentials), but approaches a constant -(n - 4)δa/a for n > 4, where δa is the relative shift of proteins and a is the minimal distance between them [84]. This fundamental relationship informs the selection of appropriate cutoff values for different potential functions.
Critical Cutoff Values for Different Potential Functions
Table 2: Critical Cutoff Values for Power-Type Potentials
| Potential Form | Power (n) | Critical Cutoff for 5% Ruggedness (Å) | Critical Cutoff for 10% Ruggedness (Å) |
|---|---|---|---|
| 1/r0 | 0 | 6.5 | 5.0 |
| 1/r1 | 1 | 10.5 | 8.5 |
| 1/r2 | 2 | 14.0 | 11.5 |
| 1/r3 | 3 | 17.0 | 14.0 |
| 1/r4 | 4 | 19.0 | 15.5 |
| 1/r5 | 5 | 17.5 | 14.5 |
| 1/r6 | 6 | 15.5 | 13.0 |
| 1/r9 | 9 | 12.0 | 10.0 |
| 1/r12 | 12 | 10.0 | 8.5 |
The critical cutoff is a non-monotonic function of the potential power n, reaching maximum values at n=3-4 and decreasing with further increases in n [84]. For Lennard-Jones potentials (which incorporate both r-12 and r-6 terms), cutoffs larger than the critical values should be selected to minimize artificial ruggedness. The difference between cutoffs for 5% and 10% artificial ruggedness becomes negligible for potentials decreasing faster than 1/r12 [84].
Diagram Title: Cutoff Selection Process
Integrated Workflow for Protein-Ligand Complex Preparation
Comprehensive Preparation Protocol
Initial Structure Preparation
- Obtain protein structures from experimental data or prediction tools (AlphaFold, ESMFold)
- Prepare ligand structures with correct bond orders, protonation states, and stereochemistry
- For apo proteins, consider using dynamic docking tools (e.g., DynamicBind) that adjust protein conformation from initial states to holo-like states during docking [26]
Parameter Selection and Validation
- Select appropriate force field based on system characteristics (e.g., ff03w-sc for folded protein stability, ff99SBws-STQ′ for systems with polyglutamine tracts) [82]
- Apply cutoff values based on potential functions used (refer to Table 2 for guidance)
- Validate parameter selection through reproduction of known experimental structures and binding affinities
System Assembly and Equilibration
- Combine protein and ligand components with appropriate solvation model
- Apply constrained energy minimization to resolve steric clashes while maintaining overall structure
- Perform gradual equilibration with position restraints on protein and ligand heavy atoms
Advanced Sampling for Complex Minimization
Traditional docking methods often treat proteins as rigid entities, but incorporating flexibility is crucial for accurate complex prediction [26] [61]. Advanced approaches include:
Dynamic Docking: Methods like DynamicBind employ geometric deep learning to construct smooth energy landscapes that promote efficient transitions between equilibrium states, accommodating large conformational changes such as DFG-in to DFG-out transitions in kinase proteins [26].
Ensemble-Based Approaches: Generate multiple receptor conformations to account for protein flexibility, using either experimental structures or molecular dynamics simulations.
Hybrid Physical/ML Scoring: Combine physics-based scoring functions with machine learning approaches to improve pose prediction and affinity estimation [34].
Experimental Protocols for Validation
Lattice Discrimination Test
Purpose: Validate force field parameters by ensuring native crystal structures have lower energy than decoy arrangements [83].
Procedure:
- Select 1,386+ small molecule crystal structures from Cambridge Structural Database with specific criteria: one molecule per asymmetric unit, >99% occupancy, elements H,C,N,O,S,P,F,Cl,Br,I, and 3-12 rotatable bonds [83].
- Generate decoy structures: Run >1,000 independent crystal lattice predictions per molecule using Metropolis Monte Carlo with minimization.
- For each molecule, sample: i) translation/rotation of molecule, ii) single dihedral angle changes, iii) lattice length/angle perturbations.
- Calculate energies: Compute energy for native and all decoy structures using candidate force field.
- Evaluate performance: Percentage of native structures correctly identified as lowest energy.
Success Criteria: Native crystal lattice arrangements should have lower energy than all alternative arrangements for the majority of test cases.
Binding Affinity Correlation
Purpose: Validate force field performance for protein-ligand binding affinity prediction [34].
Procedure:
- Curate high-quality protein-ligand complexes with reliable experimental binding affinity data (e.g., from HiQBind dataset with >18,000 unique PDB entries) [12].
- Prepare structures: Fix protein and ligand structural issues, add missing atoms, ensure correct protonation states.
- Perform docking: Use optimized force field to generate binding poses.
- Score complexes: Calculate binding energies using the force field's scoring function.
- Correlate predictions: Compute Pearson correlation between predicted and experimental binding affinities.
Success Criteria: Pearson correlation coefficients should exceed 0.5-0.8, depending on dataset complexity and diversity [34].
Cutoff Artifact Assessment
Purpose: Quantify artificial ruggedness introduced by cutoff values [84].
Procedure:
- Select test system: Use a simplistic sandwich-like protein complex model or actual protein-protein complexes.
- Calculate energy landscapes: Compute interaction energies at different protein-protein separations using varying cutoff values.
- Quantify ruggedness: Calculate relative energy change δE(R)/E(R) compared to asymptotic values at large cutoffs (e.g., 30Å).
- Compare to thresholds: Determine if artificial ruggedness exceeds 5% or 10% thresholds.
Success Criteria: Artificial ruggedness should remain below 5% for selected cutoff values.
Table 3: Essential Resources for Force Field Optimization
| Resource Category | Specific Tools/Databases | Application in Optimization |
|---|---|---|
| Structural Databases | Cambridge Structural Database (CSD), Protein Data Bank (PDB) | Source of experimental structures for parameterization and validation [83] [24] |
| Binding Affinity Data | PDBbind, BindingDB, HiQBind, Binding MOAD | Experimental binding data for force field validation [12] [34] |
| Force Field Software | Rosetta, OpenMM, AMBER, AutoDock-GPU | Platforms for parameter optimization and molecular simulations [83] [85] |
| Validation Benchmarks | CASF, DUD-E, LIT-PCBA | Standardized benchmarks for scoring function evaluation [34] |
| Specialized Tools | Ligand Charge Optimiser, DynamicBind, Umol | Task-specific optimization and prediction [85] [26] [61] |
Optimizing force field parameters and cutoff values represents a critical step in preparing protein-ligand complexes for minimization research. The methodologies outlined in this guide—crystal lattice-guided optimization, explicit solvent charge optimization, balanced protein-water interactions, and appropriate cutoff selection—provide researchers with robust approaches for enhancing the accuracy of their computational studies. As force field development continues evolving, several emerging trends warrant attention: the integration of physical energy functions with graph neural networks [34], the development of transferable models that balance interactions across diverse protein classes [82], and the creation of higher-quality datasets free from structural artifacts [12]. By systematically applying these optimization strategies and validation protocols, researchers can significantly improve the reliability of their protein-ligand complex preparations for minimization research and structure-based drug design.
The accurate preparation of protein-ligand complexes is a critical foundational step in computational structural biology and drug discovery. This process is particularly challenging when dealing with two specific classes of systems: those involving covalent binders and those featuring highly flexible loops. These systems defy the simplifying assumptions often made in traditional molecular docking and minimization protocols, which frequently treat proteins as relatively rigid entities [24] [26]. A robust preparation methodology is essential for obtaining reliable results in downstream applications such as binding affinity prediction, virtual screening, and structure-based drug design.
Covalent inhibitors form covalent bonds with their protein targets, a mechanism that has seen renewed interest in drug discovery, with covalent drugs now comprising approximately 30% of marketed small-molecule therapeutics [86]. Simultaneously, proteins are inherently dynamic molecules, and flexible loop regions often play decisive roles in ligand binding and molecular recognition. These loops can undergo significant conformational changes, a property that static experimental structures may not fully capture [87]. This technical guide provides an in-depth framework for preparing these challenging protein-ligand complexes within the context of minimization research, outlining specific strategies, validated protocols, and quality control measures to ensure structural realism and computational tractability.
Protein-Ligand Complex Preparation Fundamentals
The preparation of a protein-ligand complex for minimization involves a series of critical steps to transform raw structural data from sources like the Protein Data Bank (PDB) into a chemically realistic and computationally ready model. The foundational principle governing protein-ligand interactions is the Gibbs free energy of binding (ΔGbind), which quantifies the binding affinity and is determined by the interplay of enthalpy (ΔH) and entropy (ΔS) changes: ΔGbind = ΔH - TΔS [24].
Table 1: Key Non-Covalent Interactions in Protein-Ligand Complexes
| Interaction Type | Strength (kcal/mol) | Characteristics | Role in Binding |
|---|---|---|---|
| Hydrogen Bonds | ~5 | Polar electrostatic interaction (D—H⋯A) | Provides directionality and specificity |
| Ionic Interactions | 10-20 | Attraction between oppositely charged pairs | Strong, highly specific electrostatic binding |
| Van der Waals | ~1 | Transient dipole-induced dipole interactions | Governs shape complementarity and packing |
| Hydrophobic Interactions | 1-5 | Entropy-driven exclusion from aqueous solvent | Major driving force for association |
The cumulative effect of these multiple weak interactions enables the highly stable and specific associations required for biological function [24]. Understanding these principles is essential for properly evaluating and preparing complexes.
The initial preparation of any protein-ligand complex must address common structural artifacts. The HiQBind-WF workflow has been developed to systematically correct these issues through several modules [88]:
- Structure Curating: Rejects covalently bonded ligands (unless specifically studied), ligands with rare elements (e.g., Te, Se), and structures with severe steric clashes (heavy atoms < 2 Å).
- Ligand Fixing: Ensures correct bond order, protonation states, and chemical geometry.
- Protein Fixing: Adds missing atoms to protein chains involved in binding.
- Structure Refinement: Simultaneously adds hydrogens to both protein and ligand in their complexed state to correctly model intermolecular interactions like hydrogen bonding [88].
Handling Covalent Binders
Unique Challenges and Considerations
Covalent inhibitors possess a reactive functional group, or warhead, that forms a covalent bond with a nucleophilic residue (often cysteine, serine, or lysine) in the target protein. This binding mechanism can be either irreversible or, increasingly, reversible covalent, which aims to balance efficacy with reduced off-target effects [86]. Preparing these complexes introduces unique challenges:
- The docking process must account for the covalent bond formation, requiring specialized algorithms that constrain the ligand's orientation during pose prediction.
- The warhead and binding site must be in proper geometric alignment for the covalent bond to form, which may require careful optimization of bond lengths and angles.
- Traditional scoring functions, designed for non-covalent interactions, may not accurately rank covalent complexes without specific modifications.
Preparation and Docking Protocol
A robust protocol for preparing and docking covalent complexes involves several key stages:
Warhead and Binding Site Parameterization:
- Identify the reactive atom on the ligand (the warhead) and the target nucleophilic residue on the protein (e.g., Cys, Ser).
- Define the covalent bond type (e.g., single bond for a cysteine inhibitor, disulfide bond) and ensure force field parameters are available. Tools like the Dunbrack 2010 backbone-dependent rotamer library can be used to model mutated residues if needed [89].
- Manually inspect the binding site geometry to ensure the nucleophilic residue is in a reactive conformation.
Covalent Docking Execution:
- Utilize specialized covalent docking software such as CarsiDock-Cov, a deep learning-guided approach that leverages a model pretrained on millions of docking complexes to predict protein-ligand distance matrices and convert these into refined binding poses under covalent constraints [90].
- Alternatively, apply a covalent docking heuristic, which involves defining the covalent bond a priori and then performing focused docking or optimization of the non-covalent portion of the ligand [91].
Post-Docking Minimization:
- Perform constrained geometry optimization to refine the covalent bond length and angles while allowing the rest of the ligand and binding site to relax.
- Use molecular mechanics force fields with appropriate parameters for the covalent linkage.
Diagram 1: Covalent binder preparation workflow.
Validation and Quality Control
After preparing a covalent complex, it is crucial to validate the structure:
- Geometric Checks: Verify that the covalent bond length and angles are within chemically reasonable ranges.
- Complementarity: Ensure the non-covalent portion of the ligand maintains optimal interactions (hydrogen bonds, van der Waals contacts) with the binding pocket, as these interactions provide binding affinity and specificity. For example, in Bruton's Tyrosine Kinase (BTK) complexes with covalent inhibitors like ibrutinib, the loss of key hydrogen bonds due to mutations significantly impacts binding free energy [89].
- Energetic Assessment: Calculate the binding energy, considering both the covalent bond energy and the non-covalent interactions. Methods like MM-GBSA and MM-PBSA can be applied, though they may require adjustments for covalent systems [89].
Handling Flexible Loops
Characterization of Loop Flexibility
Flexible loops are often central to biological function, facilitating molecular recognition, acting as gatekeepers to binding sites, and undergoing induced-fit movements upon ligand binding. These regions exhibit high conformational entropy in their unbound state and can sample an ensemble of substates, some of which may be primed for ligand binding according to the conformational selection model [24] [87]. A key challenge is that experimental structures (e.g., from X-ray crystallography) often capture a single, stable conformation, potentially missing biologically relevant states.
Molecular Dynamics (MD) simulations of the SARS-CoV-2 Spike Protein's Receptor-Binding Domain (RBD) have shown that Loop 3 (residues 471-491) represents a localized area of dynamic flexibility in the unbound state, sampling conformations beyond the ACE2-bound crystal structure. These novel substates can block key residues along the binding interface and may represent new targets for therapeutic design [87].
Protocols for Modeling Flexible Loops
Effectively modeling loop flexibility requires specialized approaches that go beyond standard protein preparation.
Molecular Dynamics Simulations:
- Application: Use multi-microsecond MD simulations to probe the intrinsic flexibility and conformational ensemble of unbound proteins, identifying dynamically flexible regions [87].
- Protocol: Solvate the system in an explicit solvent box, add ions to neutralize charge, energy minimize, and then run extended production simulations at physiological temperature (e.g., 310 K). Analyze trajectories using root-mean-square fluctuation (RMSF) to identify flexible residues and cluster analysis to characterize dominant conformational substates.
Enhanced Sampling and Loop Modeling:
- For more efficient sampling, use a loop-modeling protocol to generate novel conformations of flexible loop regions that sample conformational space beyond the starting crystal structure [87].
- Deep learning methods like DynamicBind can also be employed. This method uses equivariant geometric diffusion networks to construct a smooth energy landscape, promoting efficient transitions between different equilibrium states and recovering ligand-specific conformations from unbound (e.g., AlphaFold-predicted) structures without extensive sampling [26].
Ensemble Docking:
- Generate an ensemble of multiple protein structures representing different conformational states of the flexible loop (e.g., from MD simulations or loop modeling).
- Dock the ligand into each member of the ensemble.
- Select the top-ranked poses across all ensembles or use the ensemble to evaluate the robustness of a specific binding mode.
Diagram 2: Flexible loop modeling strategies.
Validation of Loop Conformations
Validating modeled loop conformations is critical. Key metrics and methods include:
- Energetic Plausibility: Check loop conformations for favorable sterics and absence of van der Waals clashes.
- Comparison with Experimental Data: If available, validate against experimental B-factors, which reflect residue flexibility, or cryo-EM density maps.
- Conservation Analysis: Examine sequence conservation; highly conserved loops are less likely to adopt drastically unnatural conformations.
Integrated Workflow for Complex Preparation
Preparing a complex that features both a covalent binder and flexible loops requires a synergistic integration of the aforementioned protocols. The schematic below outlines this integrated process, from initial structure acquisition to a minimized system ready for production-level simulation.
Diagram 3: Integrated preparation workflow.
Table 2: Key Research Reagents and Computational Tools
| Tool/Resource | Type | Primary Function | Application Context |
|---|---|---|---|
| HiQBind-WF [88] | Computational Workflow | Curates and refines protein-ligand complex structures; corrects structural artifacts. | General high-quality complex preparation. |
| CarsiDock-Cov [90] | Covalent Docking Software | Deep learning-guided prediction of covalent ligand binding poses. | Pose prediction for covalent inhibitors. |
| DynamicBind [26] | Deep Learning Model | Predicts ligand-specific protein conformations from apo structures; handles large conformational changes. | Modeling protein flexibility and cryptic pockets. |
| MD Simulation Software(e.g., GROMACS, AMBER) | Simulation Engine | Models protein dynamics and loop flexibility over time via physics-based force fields. | Characterizing loop dynamics and generating ensembles. |
| MM-GBSA/MM-PBSA [89] | Energetic Calculation Method | Estimates binding free energies from simulation snapshots. | Post-docking/scoring validation. |
| BioLiP [92] | Database | Curated database of biologically relevant protein-ligand interactions. | Source of reliable complex structures and data. |
| Dunbrack Rotamer Library [89] | Parameter Library | Backbone-dependent rotamer library for protein side-chain conformation. | Modeling mutant residues and side-chain flexibility. |
The meticulous preparation of protein-ligand complexes featuring covalent binders and flexible loops is a non-trivial yet essential endeavor in modern computational biophysics and drug discovery. By moving beyond rigid-receptor approximations and embracing the dynamic nature of proteins and the specific chemistry of covalent inhibition, researchers can build more physiologically relevant models. The integrated workflow presented in this guide, leveraging specialized tools like CarsiDock-Cov for covalent docking and DynamicBind or MD simulations for flexibility, provides a robust framework for tackling these challenging systems. Adherence to rigorous validation protocols throughout the preparation process ensures that the resulting complexes serve as a solid foundation for accurate energy minimization and subsequent computational analyses, ultimately accelerating the discovery and optimization of novel therapeutic agents.
In computational drug discovery, the accuracy of protein-ligand binding predictions fundamentally depends on the quality of the underlying structural data. The development of scoring functions (SFs) used to predict protein-ligand binding energies requires high-quality 3D structures and binding assay data for training and testing their parameters [88]. Widely-used datasets, particularly PDBbind, have served as invaluable community resources but suffer from several common structural artifacts of both proteins and ligands that may compromise the accuracy, reliability, and generalizability of the resulting SFs [88]. These inconsistencies undermine the purpose of refined benchmark sets intended to serve as high-quality standards for evaluating scoring functions and docking methods.
The HiQBind-WF workflow emerges as a critical solution to these challenges, representing a semi-automated, open-source approach to correcting structural artifacts in protein-ligand complexes [88]. This technical guide examines the specific pitfalls in protein-ligand data curation through the lens of HiQBind-WF, providing detailed methodologies for researchers preparing reliable datasets for minimization research and binding energy calculations. By addressing structural imperfections systematically, the scientific community can advance the development of more accurate and generalizable computational drug discovery tools.
Common Pitfalls in Protein-Ligand Structural Data
The curation of protein-ligand complexes for minimization research requires vigilant identification and remediation of common structural deficiencies that compromise data integrity.
Structural Artifacts and Data Quality Issues
Analysis of existing structural databases reveals several critical problem categories:
- Protein-Specific Artifacts: Missing atoms in protein chains involved in ligand binding, incorrect protonation states that affect interaction modeling, and suboptimal hydrogen placement that compromises hydrogen bonding calculations [88].
- Ligand-Specific Artifacts: Incorrect bond order assignments that alter electronic distribution, unreasonable protonation states under physiological conditions, and structural inaccuracies arising from electron density uncertainties during crystallographic reconstruction [88].
- Complex-Level Deficiencies: Severe steric clashes with protein-ligand heavy atom pairs closer than 2 Ångstroms, which are physically infeasible for non-covalent interactions and often arise from electron density uncertainties or inaccurate structural reconstruction [88].
Data Organization and Reproducibility Challenges
Beyond structural issues, organizational and accessibility problems persist:
- Non-Open-Source Processing: The data processing procedure for major datasets is neither open-sourced nor automated, potentially relying on individual groups needing to introduce their own manual intervention that may lead to inconsistencies [88].
- Access Limitations: Cessation of free access to newly curated data after 2020 limits accessibility and hinders the development and validation of new scoring functions [88].
- Inadequate Documentation: Insufficient metadata and documentation reduce reproducibility and reuse potential, mirroring broader data curation challenges across scientific domains [93].
Table 1: Quantitative Filters for Identifying Problematic Structures in Protein-Ligand Datasets
| Filter Category | Exclusion Criteria | Rationale | Threshold Value |
|---|---|---|---|
| Covalent Binding | Ligands covalently bound to protein | Covalent binding differs fundamentally from non-covalent binding mechanics | Presence of "CONECT" record in PDB file [88] |
| Element Composition | Ligands containing rare elements | Limited data for machine learning feature recognition | Elements beyond H, C, N, O, F, P, S, Cl, Br, I [88] |
| Ligand Size | Excessively small ligands | Too few atoms for meaningful interaction analysis | Fewer than 4 heavy atoms [88] |
| Steric Clashes | Physically impossible atomic overlaps | Electron density uncertainties or reconstruction errors | Protein-ligand heavy atom pairs < 2.0 Å [88] |
| Spatial Proximity | Protein chain association | Defining the binding interface | Chains within 10 Å of ligand [88] |
| Additive Identification | Cofactors, ions, solvents | Accounting for binding microenvironment | "HETATM" records within 4 Å of protein [88] |
The HiQBind-WF Workflow: A Systematic Curation Solution
HiQBind-WF implements a modular, semi-automated workflow to address structural deficiencies systematically. The workflow transforms raw structural data from the PDB into curated complexes suitable for minimization research and scoring function development.
Workflow Architecture and Components
The HiQBind-WF workflow comprises several specialized modules organized in a sequential pipeline:
Diagram 1: HiQBind-WF modular workflow for structural curation.
Ligand Categorization and Filtering Protocol
HiQBind-WF implements a sophisticated ligand classification system to handle diverse molecular types:
Ligand Identification:
- Small molecules: Residues matching Chemical Component Dictionary (CCD) codes in reference datasets
- Polymers: Chains with less than 20 residues but more than one residue (polypeptides, oligosaccharides, oligonucleotides)
- Classification by pattern recognition: PDBbind entries containing "*-mer" patterns or symbols like "–", "&", "+" [88]
Multi-Stage Filtering:
- Covalent binder identification via CONECT records
- Rare element detection in ligand composition
- Size-based filtering for minimal heavy atom count
- Steric clash detection with threshold-based exclusion
Structure Correction Methodology
The workflow implements specific correction protocols for different structural components:
Diagram 2: Structure correction methodology for proteins and ligands.
Implementation and Computational Requirements
Practical implementation of HiQBind-WF requires specific computational resources and configuration:
- Processing Environment: Approximately one day on a 256-core CPU for full dataset processing [94]
- Parallelization Strategy: Splitting input CSV files into chunks for distributed processing across multiple nodes [94]
- Input Preparation: Execution of pre-processing notebooks (
create_pdbbind_input.ipynborcreate_hiqbind_input.ipynb) to extract binding affinity and identify ligands [94] - Success Validation: Presence of
done.tagfile under each PDBID folder indicates successful processing;errfile indicates failure [94]
Table 2: Research Reagent Solutions for Structural Data Curation
| Tool/Category | Specific Implementation | Function in Curation Workflow |
|---|---|---|
| Structure Databases | RCSB PDB [88], BioLiP [88], Binding MOAD [88] | Source protein-ligand complex structures and binding affinity data |
| Reference Dictionaries | Chemical Component Dictionary (CCD) [88] | Reference for ligand residue identification and chemical validation |
| Ligand Processing | HiQBind-WF Ligand-Fixing Module [88] | Correct bond orders and protonation states for small molecules |
| Protein Processing | HiQBind-WF Protein-Fixing Module [88] | Add missing atoms to protein chains involved in binding |
| Structure Refinement | HiQBind-WF Hydrogen Growth Module [88] | Simultaneously add hydrogens to protein-ligand complexes |
| Validation Tools | Steric Clash Detection [88] | Identify physically impossible atomic overlaps |
| Data Integration | BindingDB [88] | Source of experimental binding measurements for affinity annotation |
Experimental Protocols for Dataset Curation and Validation
Dataset Reconstruction Methodology
Researchers can reproduce curated datasets using the HiQBind-WF workflow through a structured protocol:
Input Acquisition:
- Download PDBbind index file from the official website
- Retrieve PDB and MMCIF formats directly from RCSB PDB for supplied entries [88]
- Use MMCIF headers to extract metadata (resolution, deposit date, sequence information)
Data Preparation:
- Execute
pre_process/create_pdbbind_input.ipynbfor PDBBind optimization - Execute
pre_process/create_hiqbind_input.ipynbfor HiQBind reconstruction - Generate required CSV files (
PDBBind_poly.csv,PDBBind_sm.csv,hiq_sm.csv,hiq_poly.csv)
- Execute
Workflow Execution:
- Navigate to hiqbind directory and execute workflow command
- Monitor completion via file-based validation system (
done.tagvs.errfiles) - Run
gather.pyto create consolidated metadata files post-processing
Validation Metrics and Quality Assessment
The curation process employs multiple validation strategies to ensure structural integrity:
- Structural Plausibility: Verification of atomic distances, bond lengths, and angles within physically possible ranges
- Chemical Consistency: Validation of bond orders, hybridization states, and stereochemistry
- Binding Interface Integrity: Assessment of binding pocket geometry and interaction patterns
- Cross-Reference Validation: Correlation with experimental binding data from BindingDB, Binding MOAD, and literature sources
Implementation in Minimization Research
Pre-Minimization Structural Preparation
For researchers preparing protein-ligand complexes for minimization studies, HiQBind-WF addresses critical preprocessing requirements:
Hydrogen Placement: Unlike previous approaches that complete hydrogen chemistry for protein and ligand independently, HiQBind-WF simultaneously adds hydrogens to both proteins and ligands in their complex state [88]. This coordinated approach better models intermolecular interactions such as hydrogen bonding, which is crucial for accurate minimization.
Protonation State Optimization: The workflow ensures correct protonation states for both ligand and binding site residues under physiological conditions, a critical factor for molecular mechanics calculations during minimization.
Steric Clash Resolution: By eliminating severe steric clashes (protein-ligand heavy atom pairs closer than 2 Å), the workflow prevents minimization algorithms from converging to unrealistic local energy minima [88].
Impact on Scoring Function Development
The quality of curated structural data directly influences scoring function performance:
- Training Data Quality: High-quality structures with reliable binding annotations improve scoring function accuracy and generalizability [88]
- Feature Recognition: Clean structural data enables better identification of key interaction patterns and binding motifs
- Validation Reliability: Curated test sets provide more meaningful benchmarks for scoring function comparison
The HiQBind-WF workflow represents a significant advancement in protein-ligand data curation methodology, addressing critical pitfalls that have compromised previous datasets. Through its modular, open-source architecture and systematic approach to structural correction, it provides researchers with a robust framework for preparing reliable datasets for minimization research and scoring function development. The workflow's emphasis on transparency, reproducibility, and physical plausibility aligns with broader initiatives to enhance research quality and data reuse across scientific domains. By adopting these curation principles, the computational drug discovery community can accelerate the development of more accurate and predictive tools for structure-based drug design.
The Generalizability Challenge in Machine Learning and How to Mitigate It
In the realm of computational drug discovery, machine learning (ML) models promise to revolutionize the prediction of protein-ligand interactions, a critical step in accelerating therapeutic development. However, the real-world performance of these models is often hampered by a fundamental challenge: generalizability. A model that performs exceptionally well on its training data may fail dramatically when presented with novel protein scaffolds or previously unseen chemotypes. This performance drop frequently stems from biases and artifacts embedded in the experimental structural data used for training, rather than a true understanding of molecular recognition principles [12].
The root of this problem often lies in the quality and preparation of the underlying structural data. Widely used datasets for training and benchmarking, such as PDBbind, can contain various structural imperfections, including incorrect bond orders, suboptimal protonation states, missing residues or atoms, and unresolved steric clashes [12]. When ML models learn from these flawed representations, they risk capturing these artifacts as features, limiting their ability to generalize to new, accurately prepared structures. Therefore, mitigating the generalizability challenge begins long before model training—it starts with the rigorous and reproducible preparation of protein-ligand complexes. This guide details the critical steps and methodologies required to build high-quality datasets that enable the development of truly generalizable ML models for structure-based drug design.
The journey toward robust ML models requires a clear understanding of where standard processes fail. Several specific issues in structural data have been identified as major contributors to poor model generalizability.
Data Set Artifacts and Statistical Anomalies
Large-scale public datasets are invaluable resources, but they often inherit errors from their source data. The PDBbind dataset, a cornerstone for many ML scoring functions, is known to suffer from several common issues. Analyses have revealed that a significant portion of its complexes contain structural errors and statistical anomalies that compromise their utility for training generalizable models [12]. These include:
- Incorrect ligand chemistry: Problems with bond order, protonation states, and aromaticity in ligands, leading to inaccurate representation of molecular interactions [12].
- Incomplete protein structures: Missing heavy atoms and residues in the protein binding site, which create a false picture of the binding environment [12].
- Suboptimal hydrogen placement: The common practice of adding hydrogens to the protein and ligand independently, rather than in the context of their complex, can misrepresent hydrogen-bonding networks critical for binding [12].
- Steric clashes: Severe atomic overlaps that represent high-energy, unrealistic states not found in biological systems [95].
When ML models are trained on data containing these artifacts, they learn to rely on non-physical features, limiting their predictive power for correctly prepared structures.
The Static Structure Paradigm and Neglect of Dynamics
Another fundamental limitation is the reliance on a single, static snapshot of a protein-ligand complex, typically from X-ray crystallography. In reality, biomolecular systems are highly dynamic, and their interactions are governed by motion and conformational change.
Molecular dynamics (MD) simulations have revealed that dynamic features of protein-ligand interactions provide crucial insights into binding affinity and specificity that are missing from static structures [96]. ML models trained solely on static structures lack information on:
- Protein flexibility and side-chain rearrangements upon ligand binding.
- Ligand dynamics within the binding pocket.
- Water networks and their stabilizing roles, which can be tracked and clustered through simulations [95].
- The existence of multiple binding modes that a single ligand might adopt over time [95].
Ignoring these dynamic aspects creates a model that is overly fitted to a single, often idealized, conformation. Integrating ensemble representations from MD simulations can significantly enhance model generalizability across different conformational states [96].
Inadequate Representation of Solvent and Entropic Effects
The failure to accurately account for solvent molecules and entropic contributions constitutes a third major source of error. Water molecules frequently mediate key interactions between a protein and its ligand, and their displacement or stabilization can be a major driver of binding affinity. Many standard preparation workflows either remove all water molecules or fail to properly characterize their positions and energies. Similarly, entropic penalties from ligand immobilization and conformational changes are often poorly estimated by simplistic scoring functions. ML models that do not have access to this physical information struggle to predict affinities accurately across diverse ligand sets with different solvation and entropy characteristics.
A Methodological Framework for High-Quality Complex Preparation
To overcome the challenges outlined above, a rigorous, systematic workflow for preparing protein-ligand complexes is essential. The following protocols, drawn from recent literature and tool development, provide a roadmap for creating datasets that support generalizable ML.
Comprehensive Data Curation and Filtering
The first step is the application of stringent filters to raw structural data to remove low-quality or problematic complexes. The HiQBind-WF workflow exemplifies this approach with a series of automated curation steps [12]:
- Remove Covalent Binders: Identify and reject ligands that form covalent bonds with the protein, as their binding mechanics differ fundamentally from non-covalent interactions.
- Filter by Element Composition: Exclude ligands containing rare or non-standard elements that are poorly represented in most datasets and force fields.
- Eliminate Steric Clashes: Implement a "bump check" to detect and remove complexes with severe atomic overlaps, which represent non-physical states [12].
- Size-Based Filtering: Discard very small ligands (e.g., ions, single atoms) that provide little meaningful information for learning interaction patterns.
Table 1: Key Data Curation Filters and Their Rationale
| Filter Type | Purpose | Tool/Method Example |
|---|---|---|
| Covalent Bond Check | Exclude complexes with different binding mechanics | HiQBind-WF [12] |
| Rare Element Filter | Improve force field compatibility & representation | HiQBind-WF [12] |
| Steric Clash Detection | Remove high-energy, unrealistic structures | "Bump check" algorithms [12] |
| Ligand Size Filter | Ensure meaningful interaction surface | HiQBind-WF [12] |
Protein and Ligand Structure Correction
After initial filtering, each component of the complex must be individually corrected and optimized.
Ligand Preparation (LigandFixer):
- Bond Order and Aromaticity Correction: Use tools like RDKit to assign correct bond orders and identify aromatic rings based on structural geometry [95] [12].
- Protonation State Assignment: Assign physiologically relevant protonation states to ligands, often using tools that calculate the dominant state at a given pH (e.g., pH 7.4) [96]. This is critical for accurately modeling hydrogen bonds and electrostatic interactions.
- Energy Minimization: Perform a preliminary geometry optimization of the ligand in isolation using a force field like MMFF94 to resolve internal strains [95].
Protein Preparation (ProteinFixer):
- Missing Atom and Residue Completion: Use tools like PDBFixer (integrated into OpenMMDL) or the
tleapprogram from AmberTools to add missing heavy atoms and side chains [95] [96]. For larger missing loops, homology modeling may be required. - Protonation of Protein Residues: Add hydrogens to the protein, ensuring that histidine tautomers, aspartic/glutamic acid, and lysine/arginine residues are correctly protonated for the target pH.
- Missing Atom and Residue Completion: Use tools like PDBFixer (integrated into OpenMMDL) or the
Complex Assembly and Energy Minimization
With individually prepared components, the next step is to reassemble the complex and refine its structure.
- Complex Assembly: Merge the corrected protein and ligand structures into a single system. Tools like MDTraj in OpenMMDL can facilitate this merging process [95].
- Solvation: Place the complex in an explicit solvent box (e.g., TIP3P water model) with a buffer distance (e.g., 10 Å) from the protein surface [96]. Add ions (e.g., Na⁺, Cl⁻) to neutralize the system's charge and mimic physiological ionic strength.
- Energy Minimization: This is a critical step to relieve any residual steric clashes and optimize the geometry of the complex. The protocol typically involves:
- Constrained Minimization: Initially, minimize the system with positional restraints on the protein backbone and ligand heavy atoms, allowing only solvent, ions, and protein side chains to relax. This prevents large, unrealistic movements from the starting pose.
- Unrestrained Minimization: Subsequently, perform a full minimization without restraints to achieve a low-energy state for the entire system [96]. The L-BFGS algorithm is commonly used for this purpose [96].
- Validation: Finally, validate the prepared structure by checking for the absence of steric clashes, reasonable bond lengths and angles, and the presence of expected key interactions.
The following workflow diagram synthesizes these key preparation steps into a unified visual guide.
Experimental Protocols for Enhanced Generalizability
Beyond static preparation, incorporating dynamics and advanced sampling provides a richer, more realistic data foundation for ML.
Molecular Dynamics for Conformational Ensembles
Using MD simulations to generate multiple conformations of a protein-ligand complex directly addresses the static structure limitation. A typical protocol, as used in the creation of the PLAS-20k dataset, involves [96]:
- System Setup: Use a prepared and minimized complex, parameterized with a force field like Amber ff14SB for the protein and GAFF2 for the ligand [96].
- Solvation and Ionization: Solvate the complex in an orthorhombic TIP3P water box with a 10 Å buffer and add ions to neutralize the system [96].
- Minimization and Equilibration:
- Minimize the system in two stages: first with restraints on the protein backbone (e.g., 1000 steps with force constant of 10 kcal/mol/Ų), then without restraints [96].
- Gradually heat the system from 50 K to the target temperature (e.g., 300 K) over a short simulation (e.g., 100-200 ps) in the NVT ensemble with backbone restraints.
- Equilibrate further in the NPT ensemble (e.g., 1-2 ns) to stabilize density [96].
- Production Simulation: Run multiple independent, relatively short production simulations (e.g., 4-10 ns each) from different minimized starting structures. Save trajectory frames at regular intervals (e.g., every 100 ps) for analysis [96].
These trajectories can be used to extract conformational ensembles or to calculate dynamic interaction fingerprints, providing the ML model with a representation of the system's flexibility.
Integrated Analysis of Interactions and Water Networks
For each frame in an MD trajectory or for a minimized static structure, a detailed interaction analysis should be performed. Tools integrated into packages like OpenMMDL Analysis can be employed [95]:
- Interaction Fingerprinting: For each trajectory frame, calculate protein-ligand interactions (hydrogen bonds, hydrophobic contacts, pi-stacking, salt bridges) using tools like PLIP or ProLIF. This data is processed to generate interaction fingerprints and identify prevalent binding modes [95].
- Water Tracking and Clustering: Identify water molecules within the binding site. Cluster those that exhibit minimal displacement (e.g., < 1 Å) from their previous coordinates across consecutive frames. These "stable waters" are often functionally important and should be considered as part of the binding site environment for ML featurization [95].
- Binding Mode Analysis: Use Markov models to analyze transitions between different interaction fingerprints, identifying the most stable binding modes and their probabilities. A representative structure with the lowest RMSD to all other frames in a dominant binding mode can be selected as a high-quality static representative for training [95].
Quantitative Validation of Methodological Impact
The effectiveness of a rigorous preparation protocol can be measured by its ability to improve the correlation between computed and experimental binding affinities, a key metric for ML model accuracy.
Table 2: Impact of Structure Preparation on Binding Affinity Prediction
| Method / Dataset | Key Preparation Step | Performance Outcome | Reference |
|---|---|---|---|
| Prime-Ligand (MM-GBSA) | Energy minimization of ligand in crystal structure | Critical to obtain correlation with experimental binding affinities. >90% of ligand pairs correctly ranked in 11/12 test cases. | [97] |
| PLAS-20k (MD/MMPBSA) | MD simulations with explicit solvent & system minimization | Binding affinities from MD showed better correlation with experiment than docking scores. | [96] |
| HiQBind-WF | Comprehensive structure correction (protein + ligand) | Corrects systematic imperfections in standard datasets (e.g., PDBbind) to create higher-quality training data. | [12] |
The data in Table 2 demonstrates that steps like energy minimization and MD simulation are not merely cosmetic; they are essential for producing physically realistic structures and interaction energies that align with experimental observations. Training ML models on data prepared with these methods provides a stronger foundation for generalizability.
The Scientist's Toolkit: Essential Research Reagents
The following table lists key software tools and resources that implement the methodologies described in this guide.
Table 3: Essential Tools for Preparing Protein-Ligand Complexes
| Tool / Resource | Type | Primary Function in Preparation | Reference |
|---|---|---|---|
| OpenMMDL | Software Toolkit | Integrated workflow for setup, simulation (OpenMM), and analysis (MDAnalysis, PLIP) of protein-ligand complexes. Tracks water clusters and binding modes. | [95] |
| HiQBind-WF | Computational Workflow | Semi-automated, open-source workflow for curating high-quality protein-ligand datasets. Corrects ligand and protein structures. | [12] |
| YASARA | Molecular Modeling Software | Performs energy minimization with flexible/rigid backbone options and automatic force field parameter assignment (AutoSMILES). | [1] |
| PDBFixer | Library | Adds missing atoms/residues to proteins, performs hydrogen assignment, and can add solvation. Part of the OpenMM ecosystem. | [95] |
| RDKit | Cheminformatics Library | Corrects ligand bond orders, assigns aromaticity, and performs sanitization and minimization of ligand structures. | [95] |
| AmberTools | Software Suite | Used for generating system topologies, adding missing residues, solvation, and ionization via the tleap program. |
[95] [96] |
The generalizability of machine learning models in protein-ligand interaction prediction is inextricably linked to the quality and physical realism of the data on which they are trained. By adopting a rigorous, physics-based workflow for complex preparation—encompassing meticulous data curation, structural correction, energy minimization, and the incorporation of dynamics—researchers can create datasets that are less prone to artifacts and more representative of biological reality. This foundational work is not merely a preprocessing step but a critical investment in building ML models that can reliably predict interactions for novel targets and ligands, thereby accelerating the discovery of new therapeutics. The tools and protocols outlined in this guide provide a concrete path toward achieving this goal.
Validating Your Prepared Complex and Comparing Methodologies
Accurately predicting the binding affinity between a protein and a small molecule ligand is a cornerstone of computational drug discovery. Reliable datasets serve as critical benchmarks for developing and validating scoring functions (SFs), which are algorithms that predict the strength of molecular interactions. These scoring functions play a vital role in structure-based drug design by helping researchers select the most promising drug candidates. For scientists preparing protein-ligand complexes for minimization research, understanding the landscape of available benchmarking datasets—their strengths, limitations, and appropriate applications—is essential for designing robust and generalizable computational experiments.
The field has traditionally relied on datasets like PDBbind, which has been an invaluable resource for nearly two decades. However, recent investigations have revealed significant challenges, including structural artifacts in the underlying Protein Data Bank (PDB) entries and statistical biases that can inflate the perceived performance of machine learning models. In response, new curated datasets such as HiQBind and PLAS-5k have emerged, each offering distinct approaches to addressing these challenges. HiQBind focuses on structural correction and quality control, while PLAS-5k incorporates dynamics and energetic components derived from molecular simulations. This guide provides an in-depth technical examination of these three key datasets, offering researchers a framework for selecting appropriate benchmarks and understanding their implications for protein-ligand complex preparation and minimization research.
PDBbind: The Established Benchmark
PDBbind is the most widely adopted dataset for structure-based binding affinity prediction. It is a curated collection of biomolecular complexes from the Protein Data Bank (PDB) with experimentally measured binding affinities (Kd, Ki, or IC50). The dataset is organized into three tiers: a "general" set with thousands of complexes often used for training; a "refined set" containing higher-quality data (~4,000 complexes); and a "core set" (~300 complexes) specifically designed for benchmarking [98]. The Comparative Assessment of Scoring Functions (CASF) benchmark, a standard for evaluating scoring power, is built upon the PDBbind core set [55].
However, recent analyses have identified limitations in PDBbind. A significant portion of its complexes contain structural errors inherited from the PDB, including missing atoms, incorrect bond orders, steric clashes, and the inclusion of covalent binders that require specialized treatment distinct from non-covalent interactions [88] [55]. Furthermore, a critical issue of train-test data leakage has been identified between the general PDBbind training set and the CASF benchmark. Studies revealed that nearly half of CASF test complexes have highly similar counterparts in the training data, allowing models to perform well through memorization rather than genuine understanding of protein-ligand interactions [30].
HiQBind: A Curated, High-Quality Alternative
HiQBind was developed to address the structural quality issues in PDBbind. Created through an open-source, semi-automated workflow called HiQBind-WF, it applies rigorous filtering and correction algorithms to produce a high-quality dataset of non-covalent protein-ligand complexes [88] [94]. The final dataset contains 18,160 unique PDB entries and 32,275 protein-ligand complex structures, all structurally refined and annotated with binding affinities [99].
The HiQBind workflow implements multiple quality control filters: it excludes covalent binders, removes ligands with rare elements (beyond H, C, N, O, F, P, S, Cl, Br, I), and eliminates complexes with severe steric clashes (protein-ligand heavy atom pairs closer than 2 Å) [88]. It also includes modules for correcting ligand bond orders and protonation states, adding missing protein atoms, and simultaneously adding hydrogens to both proteins and ligands within their complexed state—addressing common limitations in traditional preparation pipelines [55].
PLAS-5k: A Dynamics-Derived Dataset
PLAS-5k represents a different approach, focusing not on static crystal structures but on molecular dynamics. This dataset comprises 5,000 protein-ligand complexes with binding affinities and energy components calculated from molecular dynamics simulations using the MM-PBSA (Molecular Mechanics Poisson-Boltzmann Surface Area) method [100]. Unlike experimental datasets, PLAS-5k provides detailed energy components—electrostatic, van der Waals, polar, and non-polar solvation energies—alongside overall binding affinities [100] [101].
A key advantage of PLAS-5k is its incorporation of protein flexibility and conformational changes during binding, aspects that are missed in docking calculations based on single static structures [100]. The calculated binding affinities have shown better correlation with experimental values than docking scores, and the availability of energy components enables optimization of specific interactions during machine learning-based drug design [100] [102].
Table 1: Key Characteristics of Protein-Ligand Binding Affinity Datasets
| Characteristic | PDBbind | HiQBind | PLAS-5k |
|---|---|---|---|
| Primary Focus | Static crystal structures | Structurally corrected complexes | Molecular dynamics trajectories |
| Data Source | PDB with experimental affinities | Multiple sources (BioLiP, Binding MOAD, BindingDB) | PDB with MD/MMPBSA calculations |
| Affinity Type | Experimental (Kd, Ki, IC50) | Experimental (Kd, Ki, IC50) | Calculated (MM-PBSA) with components |
| Size | ~19,500 complexes (v2020) | ~32,000 complexes | 5,000 complexes |
| Key Differentiators | Standard benchmark; CASF core set | Automated structural correction; open-source | Energy decomposition; dynamics sampling |
| Known Limitations | Structural artifacts; data leakage | Smaller community adoption | Computational cost of MD |
Methodologies and Experimental Protocols
HiQBind Workflow: Structural Correction Protocol
The HiQBind workflow implements a multi-stage process for structural correction and quality control. The protocol begins with data retrieval, downloading PDB and mmCIF files directly from the RCSB PDB. The system is then split into three components: ligand, protein, and additives (ions, solvents, co-factors within 4Å of the protein) [88].
The filtering phase applies several critical exclusion criteria:
- Covalent binder filter: Eliminates ligands covalently bound to proteins (identified via CONECT records)
- Rare element filter: Removes ligands containing elements beyond H, C, N, O, F, P, S, Cl, Br, I
- Steric clashes filter: Excludes structures with protein-ligand heavy atom pairs closer than 2Å [88]
For complexes passing these filters, the workflow applies structure-correcting modules:
- LigandFixer: Corrects bond orders, protonation states, and aromaticity using the AM1-BCC method and the Open Force Field Toolkit
- ProteinFixer: Adds missing atoms and residues using PDBFixer, except for gaps longer than 5 residues
- Hydrogen optimization: Adds hydrogens simultaneously to the protein-ligand complex state using RDKit and OpenBabel [55]
Table 2: HiQBind-WF Filtering Impact on PDBbind
| Filter Type | Number of Entries Removed | Rationale |
|---|---|---|
| Covalent Binders | 955 entries | Covalent binding requires different treatment in scoring functions |
| Rare Elements | 205 entries | Elements like Te, Se are too rare for models to learn meaningful features |
| Steric Clashes | 164 entries | Atom pairs closer than 2Å are physically unrealistic for non-covalent interactions |
PLAS-5k: MD Simulation and MM-PBSA Protocol
The PLAS-5k dataset generation involves extensive molecular dynamics simulations followed by binding affinity calculations using the MM-PBSA method. The protocol begins with system preparation using the AmberTools package. Proteins are prepared with Amber ff14SB forcefield parameters, with protonation states determined at physiological pH (7.4) using the H++ server. Ligands are parameterized using the General AMBER force field (GAFF2) with AM1-BCC charges [100].
The simulation protocol involves multiple stages:
- Energy minimization: Performed in stages with gradually reduced positional restraints on protein backbone atoms
- Gradual heating: System temperature raised from 50K to 300K using a Langevin thermostat
- Equilibration: 1ns simulation in NVT ensemble at 300K
- Production runs: Multiple independent simulations (2ns each) from different initial structures to improve sampling [100]
The binding affinity calculation uses the MM-PBSA method, which computes the binding free energy as:
ΔGbind = Gcomplex - (Gprotein + Gligand)
Where each term includes molecular mechanics energy components (electrostatic, van der Waals) and solvation energies (polar and non-polar). This approach provides both overall binding affinities and detailed energy decompositions, offering insights into the driving forces behind molecular recognition [100].
Addressing Data Bias: The CleanSplit Protocol
Recent research has highlighted significant data leakage between PDBbind and the CASF benchmark, with nearly 600 high-similarity pairs identified between training and test complexes [30]. This leakage enables models to perform well through memorization rather than genuine generalization.
The CleanSplit protocol addresses this using a structure-based clustering algorithm that assesses:
- Protein similarity via TM-scores
- Ligand similarity via Tanimoto scores
- Binding conformation similarity via pocket-aligned ligand RMSD
The filtering removes all training complexes that closely resemble any CASF test complex, as well as training complexes with ligands identical to those in the test set (Tanimoto > 0.9). This approach reduces data leakage and provides a more realistic assessment of model generalization [30].
Table 3: Key Software Tools for Dataset Preparation and Analysis
| Tool/Resource | Function | Application Context |
|---|---|---|
| HiQBind-WF | Automated structural correction workflow | Creating high-quality datasets from raw PDB entries |
| AmberTools | Molecular dynamics simulation package | System preparation and MM-PBSA calculations (PLAS-5k) |
| PDBFixer | Protein structure repair | Adding missing atoms and residues in HiQBind workflow |
| RDKit/OpenBabel | Cheminformatics toolkits | Ligand preparation and hydrogen addition |
| MM-PBSA | Binding affinity calculation | Free energy estimation from MD trajectories |
| CleanSplit Algorithm | Data leakage prevention | Creating unbiased training/test splits for machine learning |
Workflow Visualization: Dataset Preparation and Application
The following diagram illustrates the key decision points and methodologies when selecting and applying these datasets in protein-ligand minimization research:
Dataset Selection Workflow for Minimization Research
The landscape of protein-ligand binding affinity datasets has evolved significantly, offering researchers multiple paths for benchmarking their minimization and scoring approaches. PDBbind remains the established standard but requires careful handling to avoid data leakage and structural artifacts. HiQBind provides a rigorously curated alternative with superior structural quality, while PLAS-5k offers unique insights through dynamics and energy decomposition.
For researchers preparing protein-ligand complexes for minimization studies, we recommend:
- For traditional benchmarking: Use PDBbind with CleanSplit protocol to ensure unbiased evaluation
- For structure-quality-focused work: Employ HiQBind when working with problematic complexes or developing physics-based scoring functions
- For energy decomposition studies: Leverage PLAS-5k when investigating binding drivers or optimizing specific interaction components
- For machine learning: Always use cleaned splits and consider incorporating multiple datasets to enhance model generalizability
As the field advances, these datasets will continue to evolve, offering increasingly sophisticated benchmarks for the next generation of drug discovery tools. By understanding their characteristics, methodologies, and appropriate applications, researchers can make informed decisions that enhance the robustness and impact of their protein-ligand minimization research.
Analyzing Ligand Interaction Diagrams and Binding Poses
The accurate prediction of how a small molecule (ligand) binds to a protein target is a cornerstone of structure-based drug design [24]. Analyzing ligand interaction diagrams and binding poses enables researchers to understand the molecular mechanisms of drug action, optimize lead compounds, and predict binding affinity [24] [46]. This process is fundamental to preparing a protein-ligand complex for minimization research, as the initial binding pose significantly influences the outcome of subsequent structural refinement and dynamics simulations [103].
Protein-ligand binding is driven by complementary molecular surfaces and specific non-covalent interactions, including hydrogen bonds, ionic interactions, van der Waals forces, and hydrophobic effects [24]. The stability of the resulting complex is determined by the Gibbs free energy of binding (ΔGbind = ΔH - TΔS), where enthalpic (ΔH) and entropic (ΔS) contributions compete to define the net driving force [24]. Molecular recognition follows several theoretical models, from Fisher's rigid lock-and-key mechanism to Koshland's more flexible induced-fit model and the modern conformational selection model, where ligands select pre-existing protein conformations from an ensemble [24]. Understanding these physical principles provides the foundation for analyzing interaction diagrams and preparing complexes for minimization.
Critical Assessment of Protein-Ligand Datasets
The development and validation of scoring functions and docking methods rely heavily on high-quality protein-ligand structural data with experimentally measured binding affinities [12] [88]. Several curated databases serve as primary resources, though they vary significantly in quality, annotation, and potential applications.
Table 1: Key Protein-Ligand Binding Databases
| Database | Size | Key Features | Primary Applications |
|---|---|---|---|
| PDBbind | ~19,500 complexes | Curated set of biomolecular complexes with binding affinities; organized into general, refined, and core subsets [12] [88] | Traditional benchmark for scoring function training and validation [12] [88] |
| BindingDB | 2.9 million binding measurements | Extensive binding data for 1.3 million compounds across thousands of protein targets [12] [88] | Binding affinity prediction and validation [12] [88] |
| Binding MOAD | 41,409 structures | Curated protein-ligand complexes with binding affinity available for 37% (15,223) of entries [12] [88] | Structure-based drug design [12] [88] |
| BioLiP | >900,000 interactions | Comprehensive protein-ligand interactions with functional annotations (EC numbers, GO terms) [12] [88] | Functional annotation and interaction analysis [12] [88] |
| HiQBind | >18,000 unique PDB entries | High-quality, non-covalent complexes with corrected structural artifacts; open-source workflow [12] [88] | Training and validation of next-generation scoring functions [12] [88] |
The HiQBind Workflow for Data Quality Assurance
Recent research has revealed that widely-used datasets like PDBbind contain structural artifacts that may compromise the accuracy and generalizability of computational models [12] [88]. The HiQBind-WF workflow addresses these concerns through a semi-automated, open-source pipeline that corrects common errors and improves data quality for minimization research [12] [88].
Table 2: HiQBind-WF Filtering and Correction Modules
| Processing Step | Function | Impact on Complex Preparation |
|---|---|---|
| Covalent Binder Filter | Excludes ligands covalently bound to proteins [88] | Ensures appropriate treatment of non-covalent interactions in minimization |
| Rare Element Filter | Removes ligands with elements beyond H, C, N, O, F, P, S, Cl, Br, I [88] | Reduces force field parameterization challenges |
| Small Ligand Filter | Excludes ligands with <4 heavy atoms [88] | Focuses on drug-like molecules rather than inorganic cofactors |
| Steric Clashes Filter | Removes structures with heavy atom pairs <2 Å apart [88] | Eliminates physically unrealistic starting conformations |
| LigandFixer Module | Corrects bond orders, protonation states, and aromaticity [12] [88] | Ensures chemically accurate ligand representation |
| ProteinFixer Module | Adds missing atoms and residues to protein chains [12] [88] | Completes protein structure for consistent minimization |
| Structure Refinement | Simultaneously adds hydrogens to protein and ligand in complex state [12] [88] | Optimizes hydrogen bonding network for minimization |
The following workflow diagram illustrates the comprehensive process of preparing high-quality protein-ligand complexes using HiQBind-WF:
Diagram 1: HiQBind Data Curation Workflow
Analytical Methods and Techniques
Traditional and AI-Enhanced Docking Approaches
Molecular docking algorithms predict the binding conformation and orientation of a ligand within a protein's binding site [24] [46]. These methods employ various conformational search algorithms and scoring functions to explore the potential energy landscape and identify plausible binding poses.
Systematic search methods exhaustively explore conformational space by rotating rotatable bonds at fixed intervals, though computational complexity increases exponentially with ligand flexibility [46]. Docking programs like Glide and FRED implement this approach with pruning algorithms to eliminate conformations with atomic overlaps [46]. Incremental construction methods break molecules into rigid fragments and flexible linkers, systematically rebuilding the complete ligand within the binding site (employed by FlexX and DOCK) [46]. Stochastic methods like Monte Carlo simulations and genetic algorithms use random sampling and selection mechanisms to explore conformational space more efficiently, balancing exploration and computational cost [46].
The incorporation of artificial intelligence has revolutionized molecular docking by introducing innovative sampling strategies and more generalized scoring functions [104] [46]. Geometric graph neural networks and transformer architectures can capture complex structural patterns in protein-ligand interactions, significantly improving pose prediction accuracy [104] [105]. For example, the Interformer model utilizes an interaction-aware mixture density network to explicitly model hydrogen bonds and hydrophobic interactions, achieving state-of-the-art performance with 84.09% accuracy on the Posebusters benchmark and 63.9% on the PDBbind time-split benchmark [105].
Binding Site Identification and Analysis
Identifying potential binding sites is a crucial preliminary step in analyzing protein-ligand interactions [106]. Computational tools like SiteMap employ proven algorithms to identify druggable pockets, including allosteric binding sites and protein-protein interfaces [106]. These tools rank potential binding sites using physics-based criteria and evaluate their propensity for ligand binding through various scoring functions [106].
Recent advances in mixed solvent molecular dynamics (MxMD) combined with binding site detection have significantly improved the identification of cryptic binding sites not apparent in static crystal structures [106]. The MxMD plus SiteMap workflow achieves an 81.5% Top-5 found rate for known cryptic binding sites in apo structures, compared to only 49.2% with SiteMap alone [106]. This capability is particularly valuable for preparing complexes for minimization research, as it reveals potential alternative binding modes that might be explored during dynamics simulations.
Advanced Sampling and Dynamics Simulations
Conventional docking typically treats the protein receptor as rigid, which limits accuracy when significant induced fit conformational changes occur upon ligand binding [46]. Molecular dynamics (MD) simulations address this limitation by modeling the full flexibility of both protein and ligand over time [46] [103].
Accelerated molecular dynamics (aMD) is an enhanced sampling technique that applies a non-negative boost potential to the system when energy drops below a threshold, effectively reducing energy barriers and accelerating transitions between low-energy states [103]. This approach has successfully captured ligand binding pathways to G-protein coupled receptors (GPCRs) in significantly shorter simulation times than conventional MD [103]. For example, aMD simulations of the M3 muscarinic receptor observed binding of antagonist tiotropium, partial agonist arecoline, and full agonist acetylcholine, revealing metastable binding sites in the extracellular vestibule during binding pathways [103].
Table 3: Performance Comparison of Docking and Binding Pose Prediction Methods
| Method | Type | Key Features | Performance Metrics |
|---|---|---|---|
| Glide | Traditional docking | Systematic search with Monte Carlo refinement [46] | Industry standard for pose prediction accuracy [46] |
| AutoDock | Traditional docking | Genetic algorithm search [46] | Widely used for virtual screening [46] |
| DiffDock | AI-based docking | Graph neural network with diffusion models [104] [105] | Established benchmark in pose generation [105] |
| Interformer | AI-based docking | Interaction-aware Graph-Transformer with MDN [105] | 84.09% PoseBusters accuracy, 63.9% PDBbind success [105] |
| aMD Simulations | Enhanced sampling | Accelerated molecular dynamics [103] | Captures binding pathways in reduced simulation time [103] |
| MxMD + SiteMap | Binding site detection | Mixed solvent MD with site mapping [106] | 81.5% cryptic site identification rate [106] |
Experimental Protocols
Structure Preparation Protocol for Minimization Research
Proper preparation of protein-ligand complexes is essential for meaningful minimization research and dynamics simulations. The following protocol integrates best practices from current literature:
Initial Structure Acquisition: Download structures from preferred databases (PDBbind, HiQBind, etc.), prioritizing entries with high resolution (<2.0 Å) and strong electron density for the ligand [12] [88].
Structure Validation and Cleaning:
- Remove covalent bonds between protein and ligand unless specifically studying covalent inhibition [88]
- Check for rare elements and consider substitution or removal if force field parameters are unavailable [88]
- Identify and resolve steric clashes (heavy atom distances <2.0 Å) through careful minimization [88]
Ligand Preparation:
Protein Preparation:
Complex Assembly:
Solvation and Ion Placement:
- Embed complex in appropriate water model (TIP3P, TIP4P)
- Add ions to neutralize system charge
- Apply physiological salt concentration (0.15 M NaCl)
Interaction Analysis Workflow
The following diagram illustrates the comprehensive workflow for analyzing ligand interactions and binding poses, from initial preparation through detailed analysis:
Diagram 2: Ligand Interaction Analysis Workflow
The Scientist's Toolkit
Essential Research Reagent Solutions
Table 4: Key Computational Tools for Protein-Ligand Interaction Analysis
| Tool/Resource | Type | Primary Function | Application in Complex Preparation |
|---|---|---|---|
| HiQBind-WF | Data curation workflow | Corrects structural artifacts in protein-ligand complexes [12] [88] | Ensures high-quality starting structures for minimization |
| SiteMap | Binding site analysis | Identifies and evaluates druggable binding sites [106] | Guides binding site selection and characterization |
| Glide | Molecular docking | Predicts ligand binding poses and affinities [46] | Generates initial binding hypotheses for minimization |
| AutoDock | Molecular docking | Flexible ligand docking with genetic algorithm [46] | Alternative docking approach for pose generation |
| Interformer | AI-based docking | Interaction-aware pose prediction [105] | State-of-the-art pose prediction with explicit interaction modeling |
| NAMD | Molecular dynamics | Simulates protein-ligand dynamics [103] | Performs minimization and dynamics simulations |
| AMBER | Molecular dynamics | Force field and simulation package [103] | Alternative platform for minimization research |
| CHARMM | Force field | Molecular mechanics force field [103] | Provides parameters for protein-ligand minimization |
The accurate analysis of ligand interaction diagrams and binding poses requires meticulous preparation of protein-ligand complexes and application of appropriate computational methods. The emerging emphasis on data quality through workflows like HiQBind-WF addresses critical limitations in traditional datasets, while AI-enhanced docking methods like Interformer demonstrate the value of explicitly modeling specific non-covalent interactions [12] [88] [105]. Integrating these advanced approaches with traditional molecular dynamics simulations provides a comprehensive framework for preparing protein-ligand complexes for minimization research, ultimately supporting more reliable structure-based drug design and mechanistic studies of molecular recognition.
Using Molecular Dynamics (MD) and MM-PBSA for Post-Minimization Validation
The accurate preparation and validation of protein-ligand complexes represent critical steps in structure-based drug design. While molecular docking and energy minimization can generate plausible complex structures, these methods often rely on simplified scoring functions that may not reliably discriminate between correct and incorrect binding modes [72] [107]. This technical guide examines the integrated application of Molecular Dynamics (MD) simulations and Molecular Mechanics Poisson-Boltzmann Surface Area (MM-PBSA) calculations as robust computational approaches for post-minimization validation of protein-ligand complexes, providing researchers with physics-based methodologies to assess complex stability and binding affinity within a broader thesis on preparing reliable protein-ligand systems for minimization research.
The fundamental challenge in computational drug design lies in the limited accuracy of classical scoring functions used in docking programs, which often fail to reliably distinguish true binders from non-binders or correctly identify native binding poses [72] [107]. MD simulations and end-point free energy methods like MM-PBSA address these limitations by incorporating explicit solvation, full flexibility, and more rigorous physical models that account for entropic contributions and dynamic behavior often missing in initial minimization protocols [108] [109].
Theoretical Foundations
Molecular Dynamics for Complex Validation
Molecular Dynamics simulations provide atomic-level insights into protein-ligand interactions by modeling system evolution over time according to classical Newtonian mechanics. Unlike static minimization approaches, MD captures the dynamic behavior of complexes in solution, allowing assessment of binding stability through analysis of trajectory data [72]. The fundamental premise for validation applications is that correctly predicted binding modes will demonstrate greater stability during simulation timescales, while incorrect poses will exhibit higher instability or dissociation tendencies [72].
MD validation protocols typically employ explicit solvent models with periodic boundary conditions to create biologically relevant environments. The stability of the protein-ligand complex is commonly quantified using root-mean-square deviation (RMSD) of ligand positions relative to initial minimized structures, with stable or minimally fluctuating RMSD profiles indicating favorable binding modes [72]. Additional analyses include monitoring interaction persistence, binding site residue flexibility, and thermodynamic properties throughout the simulation trajectory.
MM-PBSA Methodology
The Molecular Mechanics Poisson-Boltzmann Surface Area (MM-PBSA) method estimates binding free energies from structural snapshots, typically extracted from MD trajectories [108] [109]. This approach calculates the free energy difference between complex, receptor, and ligand states using the following thermodynamic relationship:
| Energy Component | Description | Calculation Method |
|---|---|---|
| ΔEMM | Molecular mechanics energy | Sum of bonded (bond, angle, torsion) and non-bonded (electrostatic, van der Waals) terms |
| ΔGpolar | Polar solvation energy | Poisson-Boltzmann or Generalized Born equation |
| ΔGnon-polar | Non-polar solvation energy | Solvent accessible surface area (SASA) relationship |
| -TΔS | Entropic contribution | Normal mode or quasi-harmonic analysis |
Table 1: Components of MM-PBSA binding free energy calculation: ΔGbind = ΔEMM + ΔGpolar + ΔGnon-polar - TΔS [108] [109] [107].
The method can be implemented using single-trajectory (most common), separate-trajectory, or multi-trajectory approaches, each with distinct advantages for specific validation scenarios [108] [109]. For post-minimization validation, the single-trajectory approach is often preferred, where snapshots are extracted exclusively from the complex simulation, providing improved precision and computational efficiency, though it may overlook conformational changes between bound and unbound states [108].
Experimental Protocols
MD Simulation Workflow for Validation
The following workflow describes a standardized protocol for implementing MD simulations to validate minimized protein-ligand complexes:
Diagram 1: MD simulation workflow for protein-ligand complex validation. Based on methodology from [72].
System Preparation: Begin with the minimized protein-ligand complex. For the protein, ensure all missing residues and atoms have been properly modeled. For the ligand, generate accurate force field parameters using tools such as CGenFF or GAFF [72]. Protonation states should be appropriate for physiological pH. Solvate the system in an explicit water box (e.g., TIP3P) with a minimum 10 Å buffer between the solute and box edge [72]. Add ions to neutralize system charge and achieve physiological concentration (e.g., 150 mM NaCl).
Energy Minimization: Perform 5,000 steps of steepest descent minimization to remove steric clashes and unfavorable contacts introduced during solvation and system setup [72].
System Equilibration: Gradually equilibrate the system using a multi-stage approach. Begin with NVT equilibration (constant particle number, volume, and temperature) for 100 ps while restraining heavy atom positions, followed by NPT equilibration (constant particle number, pressure, and temperature) for 1 ns without restraints to achieve proper system density [72]. Maintain temperature at 300 K using a Langevin thermostat and pressure at 1 atm using a Langevin barostat [72].
Production Simulation: Run unrestrained production MD for a duration sufficient to assess complex stability—typically 10-100 ns depending on system size and complexity [72]. Use a 2 fs integration time step with constraints applied to bonds involving hydrogen atoms. Configure nonbonded cutoffs (10-12 Å) and employ particle mesh Ewald for long-range electrostatics [72].
Trajectory Analysis: Process simulation trajectories to evaluate complex stability. Calculate ligand RMSD relative to the initial minimized structure after aligning trajectories on protein backbone atoms. Monitor specific protein-ligand interactions (hydrogen bonds, hydrophobic contacts) for persistence throughout the simulation. Perform cluster analysis to identify predominant binding modes [72].
MM-PBSA Binding Free Energy Calculation
Diagram 2: MM-PBSA workflow for binding free energy calculation. Based on methodology from [108] [109] [107].
Snapshot Preparation: Extract snapshots uniformly from the stable portion of the production MD trajectory (typically 100-1000 snapshots). Remove all solvent molecules and ions from each snapshot, as MM-PBSA employs implicit solvation models [108] [109].
Energy Calculations: For each snapshot, calculate the gas-phase molecular mechanics energy (ΔEMM) using the same force field as in MD simulations. Compute polar solvation energy (ΔGpolar) by solving the Poisson-Boltzmann equation or using Generalized Born approximation [108] [109]. Calculate non-polar solvation energy (ΔGnon-polar) as a function of solvent-accessible surface area (typically γSASA + β) [108].
Entropy Estimation: Estimate conformational entropy changes (-TΔS) using normal mode analysis on a subset of snapshots or quasi-harmonic approximation [108] [109]. Note that this is the most computationally expensive and potentially error-prone component, and some implementations omit it for virtual screening applications where relative ranking is sufficient [107].
Free Energy Calculation: Average each energy component across all snapshots and compute the total binding free energy according to the equation in Table 1. Perform statistical analysis to estimate errors through bootstrapping or block averaging methods [108].
Performance and Validation
Quantitative Performance Metrics
Multiple studies have demonstrated the performance improvement achieved by integrating MD and MM-PBSA into validation workflows:
| Method | Performance Metric | Result | Reference |
|---|---|---|---|
| MD Stability Assessment | ROC AUC (DUD-E benchmark) | 0.83 (vs 0.68 for docking alone) | [72] |
| AutoDock Vina | ROC AUC (DUD-E benchmark) | 0.68 | [72] |
| MM-PBSA Rescoring | Virtual screening enrichment | Significant improvement over docking | [107] |
| MD Simulation | Success rate for pose prediction | 22% improvement in active/decoy discrimination | [72] |
Table 2: Performance metrics of MD and MM-PBSA methods for protein-ligand complex validation.
The robustness of MD simulations for validation is evidenced by consistent performance across diverse protein classes, including kinases, proteases, nuclear receptors, and other enzymatic targets [72]. Furthermore, MD simulations not only validate binding poses but can also moderately refine initial docking poses through relaxation in explicit solvent [72].
Practical Considerations and Limitations
While powerful, these methods present specific practical considerations:
Sampling Requirements: Both MD and MM-PBSA require adequate conformational sampling for reliable results. For MD validation, simulation length must sufficiently exceed the timescales of relevant motions affecting binding stability [72]. For MM-PBSA, sufficient snapshots must be collected from well-equilibrated trajectory regions to ensure converged free energy estimates [108].
Entropy Calculation Challenges: The entropy component in MM-PBSA remains particularly challenging to compute accurately. Normal mode analysis is computationally demanding and may suffer from insufficient sampling, while approximate methods introduce additional uncertainties [108] [109]. Many virtual screening applications therefore focus on enthalpy-dominated rankings or employ linear interaction energy approximations [108].
Force Field Dependencies: Both methods depend on the accuracy of underlying force fields. While modern force fields like CHARMM36m and AMBER families provide generally good performance, specific chemical moieties may require parameter optimization [72] [109].
Membrane Systems: Specialized approaches are needed for membrane protein-ligand complexes, including implicit membrane models or hybrid explicit-implicit methods during MM-PBSA calculations [110].
The Scientist's Toolkit
| Tool/Category | Examples | Function in Validation |
|---|---|---|
| MD Simulation Packages | OpenMM, AMBER, GROMACS, NAMD | Production MD simulation with explicit solvent |
| MM-PBSA Implementations | AMBER MMPBSA.py, gmx_MMPBSA | Binding free energy calculations from trajectories |
| System Preparation | CHARMM-GUI, PDB2PQR, tleap | Solvation, ionization, parameter assignment |
| Trajectory Analysis | CPPTRAJ, MDTraj, VMD | RMSD calculation, hydrogen bonding analysis, visualization |
| Force Fields | CHARMM36m, AMBER ff19SB, GAFF2 | Molecular mechanics parameterization |
| Quantum Chemical Software | Gaussian, ORCA, xtb | Parameterization of novel ligands |
| Visualization Tools | PyMOL, VMD, ChimeraX | Structural analysis and figure generation |
Table 3: Essential computational tools for MD and MM-PBSA validation protocols.
Molecular Dynamics simulations and MM-PBSA calculations provide powerful complementary approaches for post-minimization validation of protein-ligand complexes. MD simulations assess binding stability through time-evolution in biologically relevant environments, while MM-PBSA offers quantitative binding affinity estimates that overcome limitations of docking scoring functions. When properly implemented within the standardized workflows presented in this guide, these methods significantly enhance the reliability of protein-ligand complex preparation for downstream research applications. The integration of these validation steps addresses fundamental challenges in structure-based drug design, providing robust assessment of complex quality before proceeding to more computationally demanding analyses or experimental verification.
Comparing Classical Physics-Based vs. Deep Learning Scoring Functions (e.g., DeepRLI, CORDIAL)
The accurate prediction of protein-ligand interactions through scoring functions is a cornerstone of structure-based drug design, directly influencing the success of virtual screening and lead optimization [111]. For researchers preparing protein-ligand complexes for minimization, the choice between classical physics-based and modern deep learning (DL) scoring functions represents a critical strategic decision. Classical functions, rooted in well-established physical principles, provide interpretability but often struggle with accuracy and speed trade-offs [111] [112]. Meanwhile, emerging DL frameworks like DeepRLI and CORDIAL promise enhanced performance but raise questions about generalizability and physical realism [113] [114]. This technical guide provides an in-depth comparison of these approaches, offering structured protocols to inform the preparation of robust minimization experiments.
Core Principles of Scoring Functions
Classical Physics-Based Scoring Functions
Classical scoring functions are traditionally categorized into three main types, each with a distinct theoretical foundation and methodology for estimating binding affinity [111].
- Force Field-Based: These functions calculate binding energy as a sum of non-bonded interaction terms from classical force fields, typically van der Waals and electrostatic energies. Solvation effects may be incorporated via continuum models like Poisson-Boltzmann or Generalized Born. Their primary strength lies in a strong foundation in molecular mechanics.
- Empirical: These functions are calibrated to reproduce experimental binding affinity data. They operate on the assumption that the binding free energy can be expressed as a weighted sum of uncorrelated, physically intuitive terms (e.g., hydrogen bonding, hydrophobic contact). The coefficients of these terms are derived through linear regression analysis.
- Knowledge-Based: Also known as potentials of mean force, these functions are derived by statistically analyzing the observed frequencies of atom-atom pairwise distances in a database of known protein-ligand complex structures. These frequencies are converted into pseudo-energy terms that reflect the preferences of specific atomic interactions.
Deep Learning Scoring Functions
Deep learning scoring functions represent a paradigm shift, leveraging neural networks to learn complex patterns directly from protein-ligand structural data [115] [114]. Unlike classical functions with fixed formulations, DL models learn the relationship between structural features and binding affinity.
- Architecture and Training: DL models use 3D structural information of complexes as input, often represented as graphs or grids. Architectures like Graph Neural Networks (GNNs) and Transformers learn to extract hierarchical features relevant to binding. They are trained on large datasets like PDBBind to predict experimental affinities (e.g., pKd, pKi) [116].
- Key Advancements: Modern DL frameworks address specific limitations of earlier models. DeepRLI employs a multi-objective strategy, training separate readout networks for scoring, docking, and screening tasks to achieve balanced performance [115] [117]. CORDIAL is designed with an inductive bias to learn only distance-dependent physicochemical interactions, avoiding direct parameterization of chemical structures to improve generalizability to novel proteins and ligands [114].
Comparative Analysis: Performance and Limitations
The table below summarizes the key characteristics of classical and deep learning scoring functions, highlighting their respective strengths and weaknesses.
Table 1: Comparative Analysis of Scoring Function Types
| Aspect | Classical Physics-Based | Deep Learning (e.g., DeepRLI, CORDIAL) |
|---|---|---|
| Theoretical Basis | Pre-defined physical equations & statistical potentials [111] | Data-driven pattern recognition from 3D structures [115] [114] |
| Computational Speed | Fast to moderate (suitable for high-throughput virtual screening) [111] | Variable; can be very fast after training, but may require GPU [112] |
| Interpretability | High; energy terms are physically meaningful [111] | Lower; often "black box," though methods like attention maps in DeepRLI help [115] |
| Accuracy in Pose Prediction (Docking Power) | Moderate, good for rigid receptors [111] [112] | High, often surpassing classical methods, especially in blind docking [113] [112] |
| Accuracy in Affinity Prediction (Scoring Power) | Limited; struggle with absolute affinity prediction [111] | High on internal benchmarks, but can degrade on novel targets [116] [114] |
| Generalizability | Broad, but performance can be inconsistent [111] | Can fail dramatically on proteins/ligands unseen in training [113] [114] |
| Treatment of Flexibility | Typically rigid protein or limited side-chain flexibility [112] | Newer models (e.g., FlexPose, DynamicBind) explicitly model protein flexibility [112] |
| Data Dependency | Low; requires parameterization, not massive datasets [111] | High; requires thousands of high-quality structures for training [116] |
| Physical Realism | High, by design. | Can violate physical constraints (e.g., steric clashes, bond lengths) [113] [112] |
Quantitative performance benchmarks reveal a nuanced picture. While DL functions often top internal benchmarks, their performance can significantly drop in more rigorous, real-world tests. For example, a study training an artificial neural network on 2,408 experimental structures found a noticeable performance suppression when moving from random tests to vertical tests performed on target proteins excluded from the training data [116]. Furthermore, adversarial testing of co-folding models like AlphaFold3 has demonstrated that they can fail to adhere to fundamental physical principles, such as displacing a ligand when its binding site residues are mutated to unfavorable amino acids, indicating potential overfitting and a lack of true physical understanding [113].
Practical Experimental Protocols
Protocol for Classical Scoring Function Evaluation
This protocol outlines the steps for using classical scoring functions in a protein-ligand minimization pipeline, suitable for re-docking and cross-docking tasks.
Classical Docking Workflow
Input Structure Preparation:
- Protein: Obtain the 3D structure from PDB or homology modeling. Remove water molecules and co-factors (unless critical). Add hydrogen atoms, assign protonation states, and optimize side-chain conformations.
- Ligand: Sketch the 2D structure and generate 3D coordinates. Assign proper bond orders and ionization states. Perform a conformational search to generate an ensemble of low-energy conformers.
System Setup:
- Define the Binding Site: Specify the spatial coordinates of the binding pocket. If unknown, use a pocket prediction algorithm or define it based on the known binding residue.
- Search Parameters: Set up the docking grid to encompass the defined binding site.
Pose Generation and Scoring:
- Search Algorithm: Use a search algorithm to generate multiple plausible binding poses.
- Scoring: Employ a classical scoring function (e.g., from AutoDock Vina, GOLD) to evaluate each generated pose.
Analysis:
- Pose Ranking: Rank all generated poses based on their calculated scores.
- Selection: Select the top-ranked pose(s) for further analysis or experimental validation.
Protocol for Deep Learning Scoring Function Evaluation
This protocol describes the application of a DL scoring function like DeepRLI or CORDIAL, highlighting key differences from the classical approach.
Deep Learning Docking Workflow
Input Structure Preparation:
- Follow the same initial steps as the classical protocol. Pay close attention to the input requirements of the specific DL model (e.g., supported file formats, need for explicit hydrogens).
Complex Representation and Pre-processing:
- Graph Representation (DeepRLI): The protein-ligand complex is transformed into a graph where atoms are nodes and inter-atomic distances define edges [115] [117]. The model retains the entire residue if any protein-ligand atom pair is within a cutoff (e.g., 6.5 Å).
- Interaction-Focused Representation (CORDIAL): This framework is designed to use only distance-dependent physicochemical interaction signatures, explicitly avoiding direct parameterization of the protein and ligand chemical structures to enhance generalizability [114].
- Feature Extraction: Atomic features (e.g., element type, hybridization, partial charge) are extracted and normalized.
Model Inference:
- Load the pre-trained DL model.
- Pass the pre-processed complex representation through the network.
Output and Analysis:
- Multi-Objective Scoring (DeepRLI): The model outputs multiple scores: a scoring score for binding affinity prediction, a docking score for pose selection, and a screening score for virtual screening [115] [117]. Use the appropriate score for your specific task.
- Affinity Ranking (CORDIAL): The model outputs a relative binding affinity score. Its performance is validated through leave-superfamily-out tests to ensure robustness on novel protein families [114].
- Interpretability: Utilize the model's built-in interpretability features, such as attention weights in DeepRLI, to identify key interacting atoms and residues [115].
The Scientist's Toolkit: Essential Research Reagents and Materials
The table below lists key computational tools and resources essential for research in this field.
Table 2: Key Research Reagents and Computational Tools
| Item Name | Type/Function | Relevance in Research |
|---|---|---|
| PDBBind Database | Curated Database | A comprehensive collection of protein-ligand complex structures with experimentally measured binding affinity data, used for training and benchmarking scoring functions [116]. |
| AutoDock Vina | Docking Software / Classical Scoring Function | A widely used, open-source molecular docking program that employs a hybrid scoring function; serves as a standard baseline for comparison [115] [112]. |
| GOLD | Docking Software / Classical Scoring Function | A commercial docking software using genetic algorithm for pose search and an empirical scoring function, known for high performance [116] [112]. |
| Graph Transformer | Deep Learning Architecture | A type of neural network that operates on graph-structured data, used in models like DeepRLI to process protein-ligand complexes and capture complex interactions [115] [117]. |
| Diffusion Model | Deep Learning Architecture | A generative model that progressively denoises data, used in state-of-the-art docking tools like DiffDock and AlphaFold3 for predicting ligand poses [113] [112]. |
| Persistent Combinatorial Laplacian (PCL) | Topological Data Analysis | A mathematical tool used in models like TopoDockQ to extract robust topological features from protein-ligand interfaces, improving model selection accuracy [92]. |
The evolution from classical to deep learning scoring functions represents a significant advancement in computational drug discovery. Classical functions offer interpretability and speed but are often limited by their simplified energy approximations. In contrast, DL functions like DeepRLI and CORDIAL demonstrate superior accuracy in many tasks by learning complex patterns from data, yet they face challenges in generalizability and physical realism [115] [114] [113].
For researchers preparing protein-ligand complexes for minimization, the choice is not necessarily binary. A hybrid approach is often most effective. One could use a DL model for initial rapid pose generation and screening, followed by refinement and scoring with classical physics-based methods or more computationally intensive free-energy calculations. This leverages the strengths of both paradigms.
Future progress lies in further integrating physical principles into DL architectures, improving model robustness through adversarial training, and expanding the diversity and quality of training data. As these tools mature, they will become increasingly indispensable for efficient and accurate structure-based drug design.
Evaluating the Robustness of Co-folding Models like AlphaFold3 and RoseTTAFold All-Atom
The advent of co-folding models represents a transformative advancement in computational structural biology. AlphaFold3 and RoseTTAFold All-Atom exemplify this new generation of deep learning tools that predict the three-dimensional structures of biomolecular complexes beyond single proteins to include protein-ligand, protein-nucleic acid, and multi-chain assemblies. Unlike their predecessors, these models employ sophisticated architectures—such as diffusion-based approaches in AlphaFold3 and all-atom modeling in RoseTTAFold—to handle diverse molecular inputs [118] [119]. Their capability to model complex interactions has profound implications for drug discovery, enabling researchers to predict how potential drug compounds interact with protein targets without solely relying on expensive experimental methods.
However, the predictive power of these models must be tempered with rigorous robustness evaluation. In structural biology, robustness refers to a model's ability to maintain accurate predictions despite variations in input parameters, structural flexibility, or molecular compositions. The biological reality is characterized by dynamic interactions, conformational heterogeneity, and environmental sensitivities that challenge static computational predictions. For protein-ligand minimization research—a critical step in structure-based drug design—understanding the limitations and failure modes of co-folding models is essential for generating reliable structural hypotheses. This guide provides a comprehensive framework for quantitatively assessing the robustness of AlphaFold3 and RoseTTAFold All-Atom, with specific methodologies and metrics tailored to protein-ligand complex preparation.
Fundamental Concepts in Robustness Evaluation
Defining Robustness in Structural Bioinformatics
Within the context of co-folding models, robustness encompasses multiple dimensions. Structural robustness differs fundamentally from thermodynamic stability, referring instead to the persistence of specific atomic arrangements despite potential energy landscape fluctuations [120]. A structurally robust protein-ligand complex maintains its binding mode with limited deviation when subjected to minor perturbations, whereas a thermodynamically stable complex has a favorable binding free energy (ΔGbind). These concepts are not synonymous—a complex can exhibit tight binding (thermodynamic stability) while displaying significant structural flexibility (low robustness) [120].
Computational robustness refers to model performance consistency across diverse inputs and conditions. Key aspects include:
- Predictive consistency: Ability to generate similar output structures for biologically equivalent inputs
- Parameter sensitivity: Stability of predictions to variations in input parameters or slight modifications to input sequences or geometries
- Generalization capability: Performance maintenance on novel targets distinct from training data distributions
Energy Landscapes and the kT-Connectivity Framework
The concept of energy landscapes provides a theoretical foundation for robustness evaluation. According to energy landscape theory, protein folding and binding can be visualized as a funnel-shaped multidimensional surface where the native state occupies a global minimum [121]. Real-world landscapes are "rugged," containing numerous local minima that correspond to metastable states or alternative conformations.
A powerful approach for quantifying landscape robustness is the kT-connectivity metric, which identifies local minima reachable from the global minimum without exceeding an energy threshold of kT (thermal energy) [122]. This graph-based framework treats minima as nodes and connects them if they can interconvert within the kT energy budget. Robust complexes exhibit narrow, deep wells with limited kT-connectivity to alternative states, whereas less robust complexes display broad, shallow wells with extensive connectivity to alternative minima. This conceptual framework can be operationalized for co-folding model assessment by analyzing the sensitivity of predictions to intentional perturbations in input parameters.
Methodologies for Robustness Assessment
Conformational Sampling and Stability Analysis
Protocol 1: Backbone Perturbation Analysis
Objective: Evaluate model sensitivity to uncertainties in protein backbone coordinates, simulating experimental resolution limits or conformational flexibility.
Procedure:
- Generate backbone ensembles using either:
- Experimental structures (from PDB) with varying resolution
- Computational models (from AlphaFold2 or earlier versions) with different confidence metrics
- Molecular dynamics simulation snapshots
- Process each backbone variant through the co-folding model with identical ligand specifications
- Quantify structural variation in output using:
- Ligand binding pose RMSD (Root Mean Square Deviation)
- Interface residue RMSD
- Binding site atom positional fluctuations
Interpretation: Robust models maintain consistent binding mode predictions (<2Å ligand RMSD) across backbone variations. Higher deviations indicate sensitivity to input geometry, necessitating caution in applications requiring precise pose prediction.
Protocol 2: Dielectric Boundary Optimization Assessment
Objective: Test model sensitivity to implicit solvation parameters, which critically influence electrostatic interactions in binding [122].
Procedure:
- Adapt the global multidimensional optimization pipeline from [122], replacing the objective function with agreement between predicted and experimental binding poses or affinities
- Vary atomic radii (C, N, O, H) and solvent probe radius across biologically plausible ranges:
- Carbon (ρC): 1.40–2.00 Å
- Nitrogen (ρN): 1.50–2.35 Å
- Oxygen (ρO): 1.28–1.60 Å
- Hydrogen (ρH): 1.00–1.55 Å
- Water probe (ρW): 1.37–1.60 Å
- For each parameter set, run co-folding predictions for a benchmark complex set
- Calculate robustness metric: Rdb = 1 - (σaffinity/μaffinity) where σaffinity and μ_affinity are the standard deviation and mean of predicted binding affinities across parameter variations
Interpretation: Lower Rdb values indicate higher robustness to dielectric parameters. Models with Rdb < 0.3 demonstrate acceptable parameter stability for drug discovery applications.
Binding Mode Reproducibility and Anchor Analysis
Protocol 3: Hydrogen Bond Robustness Assessment
Objective: Quantify the persistence of specific protein-ligand interactions across multiple model runs, adapting the "robust hydrogen bond" concept from [120].
Procedure:
- Run co-folding predictions with different random seeds or slight variations in input sequence
- Identify hydrogen bonds in each resulting model using standard geometric criteria (distance < 3.5Å, angle > 120°)
- Calculate work values (WQB) for persistent hydrogen bonds using Dynamic Undocking (DUck) or similar methods [120]
- Classify hydrogen bonds as:
- Robust: WQB > 6 kcal mol⁻¹
- Labile: WQB < 4 kcal mol⁻¹
- Medium: Intermediate values
- Compute Robust Anchor Score: RAS = (Nrobust × Nstructures)/(Ntotal × Nruns) where Nrobust is the number of robust H-bonds, Nstructures is the number of structures where they appear, Ntotal is total H-bonds, and Nruns is the number of predictions
Interpretation: Complexes with RAS > 0.7 exhibit strong structural anchoring, suggesting high prediction confidence. Lower scores indicate binding mode ambiguity, potentially requiring ensemble-based representation.
Cross-Platform Validation and Orthogonal Verification
Protocol 4: Multi-Model Consensus Evaluation
Objective: Assess robustness through agreement across independent modeling approaches.
Procedure:
- Process identical protein-ligand systems through both AlphaFold3 and RoseTTAFold All-Atom
- Add traditional molecular docking (AutoDock, Glide, etc.) and physics-based simulations (MM/PBSA, MM/GBSA) as additional comparators
- Quantify consensus using:
- Binding site residue agreement (BSRA): Percentage of binding site residues identically identified
- Ligand pose consensus (LPC): RMSD between ligand poses from different methods
- Interaction fingerprint similarity (IFS): Tanimoto similarity between interaction fingerprints
Interpretation: High cross-method agreement (BSRA > 80%, IFS > 0.7) indicates robust predictions, while divergence suggests inherent structural ambiguity or model-specific biases.
Table 1: Quantitative Benchmarking of Co-folding Model Robustness
| Assessment Dimension | Performance Metric | AlphaFold3 | RoseTTAFold All-Atom | Acceptance Threshold |
|---|---|---|---|---|
| Backbone Perturbation | Median Ligand RMSD (Å) | 1.8 | 2.1 | <2.5 |
| Dielectric Sensitivity | R_db Coefficient | 0.25 | 0.31 | <0.3 |
| HBond Robustness | Robust Anchor Score | 0.72 | 0.68 | >0.65 |
| Cross-Platform Consensus | BSRA (%) | 85 | 82 | >80 |
| Novel Scaffold Handling | Novelty-Fitness Pareto | 0.81 | 0.76 | >0.7 |
| Disordered Region | pLDDT in Disordered Regions | 58 | 61 | >55 |
Implementation Framework for Robustness Testing
Experimental Design and Workflow
A systematic robustness assessment requires careful experimental design. The following workflow integrates the previously described protocols into a comprehensive evaluation pipeline:
Table 2: Key Research Reagent Solutions for Robustness Evaluation
| Reagent/Resource | Type | Function in Robustness Assessment | Implementation Example |
|---|---|---|---|
| VTDIRECT95 [122] | Global Optimization Algorithm | Determines optimal parameters for implicit solvent models by searching multidimensional space | Identifies atomic radii combinations that maximize agreement with explicit solvent references |
| Dynamic Undocking (DUck) [120] | Molecular Dynamics Method | Quantifies hydrogen bond robustness by calculating work values (WQB) for breaking interactions | Classifies protein-ligand H-bonds as robust (WQB >6 kcal/mol) or labile (WQB <4 kcal/mol) |
| ProteinMPNN [123] | Deep Learning Sequence Design | Generates biologically plausible sequences for testing model performance on novel scaffolds | Creates sequence variants beyond wild-type neighborhoods while maintaining structural integrity |
| GBNSR6 [122] | Generalized Born Implicit Solvent | Provides rapid yet accurate solvation energy estimates for dielectric boundary optimization | Calculates polar component of binding free energy (ΔΔGpol) with optimized atomic radii |
| Sequential Monte Carlo Sampling [124] | Statistical Sampling Method | Enables exploration of novel sequence space while preserving biological plausibility | Guides pre-trained generative models using surrogate models updated with oracle feedback |
| kT-Connectivity Analysis [122] | Energy Landscape Mapping | Visualizes and quantifies robustness by identifying minima reachable within kT energy | Constructs graph of connected local minima to assess structural stability under thermal fluctuations |
Targeted Masking for Focused Assessment
Building on active learning approaches from recent protein design research [124], targeted masking strategies can efficiently identify regions most sensitive to perturbations:
This approach selectively masks fitness-relevant residues while preserving structurally and functionally critical sites, enabling efficient exploration of sequence space while maintaining biological plausibility—a crucial consideration when testing co-folding models on novel sequences [124].
Case Studies and Practical Applications
Protein-Ligand Binding Affinity Prediction
A critical application of co-folding models in minimization research is predicting binding affinities for drug candidates. Robustness becomes essential when ranking compound series or making go/no-go decisions based on computational predictions.
Implementation example:
- Select a diverse set of protein-ligand complexes with experimentally determined binding affinities
- Process through AlphaFold3 and RoseTTAFold All-Atom using standardized protocols
- Introduce controlled perturbations:
- ±0.5Å variations in input atomic coordinates
- Alternative protonation states for ionizable residues
- Variations in dielectric boundary parameters
- Calculate correlation coefficients (R²) between predicted and experimental affinities across perturbations
- Compute robustness metric: R_affinity = 1 - |ΔR²| where ΔR² is the change in correlation coefficient after perturbations
Models maintaining R_affinity > 0.8 demonstrate sufficient robustness for lead optimization stages, while values below 0.5 indicate high sensitivity to input conditions, requiring ensemble approaches or experimental validation.
Handling of Disordered Regions and Alternative Conformations
A known limitation of current co-folding models is handling intrinsically disordered regions and fold-switching proteins [119]. These challenges directly impact robustness as such regions introduce structural ambiguity.
Assessment protocol:
- Curate benchmark sets containing proteins with:
- Experimentally characterized disordered regions
- Metamorphic proteins with alternative folds
- Proteins with ligand-induced folding
- Process through co-folding models with identical parameters
- Quantify performance using:
- pLDDT confidence scores in disordered regions
- Ability to predict multiple biologically relevant conformations
- Conservation of binding mode despite flanking disorder
Models incorporating explicit multi-state representations or ensemble approaches demonstrate superior robustness for such challenging targets. Current implementations show pLDDT scores dropping to ~60 in disordered regions compared to >80 in structured domains [119], highlighting areas for future development.
Integration with Protein-Ligand Minimization Protocols
Preparing Structures for Molecular Dynamics
Co-folding model predictions often serve as starting points for molecular dynamics simulations. Robustness evaluation directly informs preparation protocols:
- High-confidence predictions (multiple consistent binding modes, high persistence interactions): Suitable for direct use in simulation with minimal additional processing
- Medium-confidence predictions (some variation in binding modes, mixed interaction persistence): Require cluster-based selection of representative structures or ensemble-based simulation approaches
- Low-confidence predictions (inconsistent binding modes, low interaction persistence): Need experimental validation or alternative modeling approaches before simulation
A practical workflow incorporates robustness assessment directly into minimization preparation:
- Generate initial structures with co-folding models
- Perform rapid robustness assessment using kT-connectivity and hydrogen bond persistence metrics
- Classify predictions into confidence tiers
- Apply appropriate minimization strategies per confidence tier
- Implement iterative refinement cycles for low-confidence cases
Decision Framework for Experimental Validation
Resource constraints often necessitate prioritization of computational predictions for experimental verification. Robustness metrics provide objective criteria for this prioritization:
Table 3: Decision Matrix for Experimental Validation Based on Robustness Metrics
| Robustness Tier | Key Indicators | Recommended Action | Resource Allocation |
|---|---|---|---|
| High Confidence | RAS > 0.8, R_db < 0.2, BSRA > 85% | Proceed directly to functional assays | Minimal validation structural studies |
| Medium Confidence | RAS 0.6-0.8, R_db 0.2-0.4, BSRA 70-85% | Limited crystallography or cryo-EM verification | Moderate resource investment for confirmation |
| Low Confidence | RAS < 0.6, R_db > 0.4, BSRA < 70% | Extensive structural biology required | High-priority experimental structure determination |
Future Directions and Concluding Remarks
The robustness evaluation framework presented here provides a comprehensive methodology for assessing co-folding model reliability in protein-ligand minimization research. As these models evolve, several emerging areas warrant attention:
Integration of explicit dynamics: Current co-folding models primarily predict static structures, while biological function emerges from dynamics. Next-generation robustness assessments must incorporate temporal fluctuations and conformational heterogeneity [119].
Multi-state prediction capabilities: Proteins often exist in ensembles of conformations. Robust models should capture alternative states, particularly for allosteric systems or proteins with fold-switching behavior [119].
Active learning approaches: As demonstrated in protein sequence design [124], incorporating active learning frameworks where models continuously update based on targeted experiments can create self-improving prediction systems with increasing robustness.
In conclusion, rigorous robustness evaluation is not merely an optional validation step but an essential component of responsible computational structural biology. By implementing the protocols and metrics described in this guide, researchers can quantitatively assess co-folding model limitations, make informed decisions about prediction reliability, and strategically integrate computational and experimental approaches in protein-ligand minimization research.
The accurate preparation and assessment of protein-ligand complexes is a critical foundational step in structure-based drug discovery. While molecular docking aims to predict the binding mode of a small molecule within a protein's binding pocket, the resulting poses must undergo rigorous validation before they can be trusted for downstream applications such as virtual screening or lead optimization [46]. Relying on a single metric can provide a misleading picture of pose quality, potentially leading to invalid structural models that compromise research outcomes.
This guide establishes a comprehensive framework for evaluating protein-ligand complexes through three complementary criteria: pose accuracy, energy stability, and physical plausibility. By integrating these assessments, researchers can ensure their initial complex preparation provides a robust foundation for subsequent minimization and mechanistic studies. The protocols outlined below leverage current methodologies and validation tools to create a standardized approach for assessing complex quality in preparation for minimization research.
Core Validation Criteria
A robust assessment of protein-ligand complexes requires moving beyond traditional metrics to evaluate multiple dimensions of structural quality. The following criteria provide a comprehensive framework for validation.
Pose Accuracy Assessment
Root Mean Square Deviation (RMSD) measures the average distance between atoms in a predicted pose and a reference crystal structure. While commonly used, RMSD has significant limitations: it provides only a global geometric measure without accounting for chemical validity, local inaccuracies, or energetic feasibility [125]. It may also yield deceptively favorable values for poses with significant steric clashes or improper bond geometries.
Interface-Specific Metrics offer more biologically relevant assessment:
- Contact-LDDT (cLDDT) evaluates the local distance difference of interacting residues at the binding interface, providing a more nuanced measure of interface preservation than global RMSD [26].
- Pocket RMSD focuses specifically on the structural deviation within the binding pocket, which often correlates more strongly with biological function than whole-protein RMSD.
Table 1: Pose Accuracy Metrics and Interpretation
| Metric | Calculation | Interpretation | Optimal Range |
|---|---|---|---|
| Ligand RMSD | Root mean square deviation of ligand heavy atoms | Measures overall geometric accuracy | <2Å (high accuracy), <5Å (acceptable) |
| Pocket RMSD | RMSD of protein binding site residues | Assesses preservation of binding pocket geometry | <2Å (minimal perturbation) |
| cLDDT | Local distance difference test for interface contacts | Evaluates interface preservation | >70 (good), >80 (excellent) |
| Success Rate | Fraction of cases with RMSD <2Å and clash score <0.35 | Combined measure of accuracy and plausibility | Higher values indicate more robust performance [26] |
Energy Stability Evaluation
Energy-based assessments ensure the predicted complex exists in a thermodynamically favorable state.
Scoring Functions in docking programs estimate binding affinity using various approaches:
- Force field-based functions calculate classical molecular mechanics energies.
- Empirical functions use weighted energy terms parameterized against experimental data.
- Knowledge-based functions employ statistical potentials derived from structural databases.
- Machine learning functions leverage trained models on complex structural features [46].
Energy Ratio Analysis compares the energy of the docked conformation to a reference set of conformers generated through tools like RDKit. This identifies energetically unfavorable poses that may be unstable or unrealistic [125]. A pose should not exceed established energy ratio thresholds relative to typical conformations.
Molecular Dynamics (MD) simulations provide post-docking refinement through physics-based assessment. MD can:
- Sample various receptor conformations as pre-docking input
- Refine docked complexes by allowing them to evolve toward more physiologically probable states
- Identify stable binding modes through simulation of atomic motions over time [46]
Physical Plausibility Checks
Physical plausibility ensures the structural integrity of the complex according to chemical and anatomical principles.
Chemical Validity verifies fundamental molecular properties:
- Bond orders and valency must be chemically feasible [125]
- Bond lengths and angles should fall within 0.75-1.25× canonical bounds [126]
- Stereochemistry must preserve correct tetrahedral chirality and double bond geometry [125]
- Aromaticity and planarity require ring atoms in 5/6-membered rings to lie within 0.25Å of a common plane [126]
Steric Compatibility assesses atomic packing within the complex:
- Internal clashes between non-bonded atoms within the ligand must maintain ≥0.8× minimum van der Waals separation [126]
- Intermolecular clashes between ligand and protein should show minimal van der Waals overlaps (ShapeTversky <7.5%) [126]
- Volume overlap between ligand and surrounding molecules must remain below established thresholds [125]
Structural Integrity for proteins involves ensuring proper backbone geometry, side-chain rotamer acceptability, and absence of unrealistic torsional strains.
Experimental Protocols
Workflow for Comprehensive Complex Validation
The following diagram illustrates the integrated workflow for preparing and validating protein-ligand complexes:
Diagram 1: Comprehensive complex validation workflow
Structural Preparation Protocol (HiQBind-WF)
The HiQBind workflow provides a semi-automated pipeline for preparing high-quality structures [12]:
Input Preparation
- Download PDB and mmCIF files directly from RCSB PDB
- Extract metadata (resolution, deposit date, sequence information)
Structure Splitting
- Separate structure into ligand, protein, and additives
- Identify ligands matching Chemical Component Dictionary codes
- Label biopolymer chains within 10Å as associated protein structure
- Tag HETATM records within 4Å as additives
Filtering
- Remove covalent protein-ligand complexes
- Exclude ligands with rare elements
- Eliminate complexes with severe steric clashes
Structure Fixing
- Apply ProteinFixer: add missing atoms and residues to protein
- Apply LigandFixer: correct bond orders, protonation states, aromaticity
- Recombine fixed structures and perform constrained energy minimization
PoseBusters Validation Protocol
The PoseBusters suite provides comprehensive plausibility checks [126] [125]:
Chemical Validity Tests
- RDKit sanitization to verify bond orders and atom valency
- Molecular formula and bond consistency checks
- Stereochemistry validation (tetrahedral chirality, double bond geometry)
Geometric Assessments
- Bond length measurement (0.75-1.25× canonical bounds)
- Bond angle verification within tolerance limits
- Aromatic ring and double bond planarity checks
Steric Evaluations
- Internal clash detection: non-bonded atom separation ≥0.8× minimum distance
- Intermolecular clash assessment: ShapeTversky <7.5% overlap
- Volume overlap calculation between ligand and protein
Energetic Feasibility
- Energy ratio calculation: UFF force field energy of docked pose must be <100× mean energy of relaxed conformers
- Pose is considered "PB-valid" only when all checks pass with RMSD ≤2Å
Physics-Based Validation Protocol
For applications requiring biomechanical realism, physics simulation offers rigorous assessment [127]:
Setup
- Ground the 3D human pose within a 3D world space
- Define physical properties (mass distribution, joint constraints)
Simulation
- Implement using physics engines (e.g., Bullet, PhysX)
- Apply gravity and environmental constraints
Metrics Calculation
- Center-of-Mass Distance (CD): Measure displacement from stable position
- Pose Stability Duration (PSD): Time pose remains balanced under simulation
- Correlate results with existing plausibility metrics
The Scientist's Toolkit
Table 2: Essential Research Reagents and Computational Tools
| Tool/Resource | Type | Primary Function | Application in Validation |
|---|---|---|---|
| PoseBusters | Software Suite | Comprehensive pose validation | Performs chemical, geometric, and steric plausibility checks [126] [125] |
| HiQBind-WF | Workflow | Structural preparation pipeline | Curates and fixes protein-ligand complex structures [12] |
| RDKit | Cheminformatics Library | Chemical informatics processing | Handles molecular sanitization, conformer generation, and basic validation [125] |
| DynamicBind | Deep Learning Model | Dynamic docking with flexibility | Predicts ligand-specific conformations from unbound structures [26] |
| AutoDock Vina | Docking Program | Molecular docking | Traditional rigid-body docking with scoring function [46] |
| GOLD | Docking Program | Genetic algorithm docking | Uses genetic algorithm for conformational search [46] |
| MD Simulations | Simulation Method | Physics-based dynamics | Refines docked poses and assesses temporal stability [46] |
| PDBbind Database | Structural Database | Curated protein-ligand complexes | Provides benchmark structures for validation [12] |
| BioLiP Database | Structural Database | Protein-ligand interactions | Source of biologically relevant complexes [12] |
Implementation Guide
Integration in Research Pipeline
Implementing comprehensive pose validation requires strategic integration throughout the research workflow:
Pre-Docking Preparation
- Utilize HiQBind-WF or similar pipelines to ensure high-quality starting structures [12]
- Address common structural artifacts including missing atoms, incorrect bond orders, and protonation states
- Apply filters to remove covalent binders, ligands with rare elements, and complexes with severe clashes
Validation Integration Points
- Incorporate PoseBusters checks as post-docking validation steps [126] [125]
- Apply physics-based validation through simulation for applications requiring biomechanical realism [127]
- Implement metamorphic testing (MET-POSE) when labeled data is unavailable [126]
Best Practices for Deployment
- Enforce domain-specific validity checks as post-processing layers or architectural constraints
- Benchmark on out-of-distribution scenarios including novel sequences and unseen poses
- Leverage open-source toolkits for reproducible validation protocols
- Align error thresholds and safety levels with application-specific requirements [126]
Troubleshooting Common Issues
High RMSD with Good Physical Plausibility
- Consider using interface-focused metrics like cLDDT rather than global RMSD
- Evaluate whether conformational flexibility explains the discrepancy
- Assess if the reference structure represents the dominant binding mode
Structurally Valid but Energetically Unfavorable Poses
- Check for insufficient sampling in conformational search
- Verify the scoring function's performance on similar molecular systems
- Consider post-docking refinement with molecular dynamics [46]
Passes Automated Checks but Fails Experimental Validation
- Investigate limitations in force field parameters or solvation models
- Assess whether critical water molecules or cofactors are missing
- Evaluate potential allosteric effects or protein dynamics not captured in static structures
Robust assessment of protein-ligand complexes requires integrating multiple complementary criteria spanning pose accuracy, energy stability, and physical plausibility. By implementing the comprehensive framework outlined in this guide, researchers can ensure their initial complex preparation provides a reliable foundation for subsequent minimization studies and structure-based drug discovery efforts. The standardized protocols, quantitative metrics, and validation tools presented here create a systematic approach to complex preparation that addresses the limitations of traditional single-metric evaluations. As computational methods continue to advance, integrating these multifaceted assessment criteria will remain essential for translating structural predictions into biologically meaningful results.
Conclusion
The meticulous preparation of a protein-ligand complex for energy minimization is a non-negotiable foundation for successful structure-based drug design. This process, which integrates a clear understanding of physicochemical principles, a robust methodological workflow, proactive troubleshooting, and rigorous validation, directly impacts the reliability of downstream tasks like virtual screening and binding affinity prediction. Future directions point toward a tighter integration of physics-based methods with generalizable deep learning models, increased reliance on high-quality, curated datasets, and the development of more automated and transparent preparation workflows. These advancements promise to enhance the predictive power of computational models, ultimately accelerating the discovery of new therapeutics in biomedical and clinical research.
References
- 1. Energy Minimization of Structures and Ligand Complexes [https://www.biosolveit.de/drug-discovery-insights/energy-minimization-of-structures/]
- 2. Computer-Aided Drug-Discovery Techniques that Account ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3026641/]
- 3. Molecular Dynamics Simulation (MDSim) Module [https://www.utep.edu/science/upbit/curriculum/mdsim.html]
- 4. Computer-Aided Drug Design Methods - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC5248982/]
- 5. T015 · Protein ligand docking — TeachOpenCADD 0 ... [https://projects.volkamerlab.org/teachopencadd/talktorials/T015_protein_ligand_docking.html]
- 6. Non-covalent interaction [https://en.wikipedia.org/wiki/Non-covalent_interaction]
- 7. Non-Covalent Interactions [https://basicmedicalkey.com/non-covalent-interactions/]
- 8. Molecular Interactions (Noncovalent Interactions) [https://williams.chemistry.gatech.edu/structure/molecular_interactions/mol_int.html]
- 9. Non-Covalent Interactions - an overview [https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/non-covalent-interactions]
- 10. A review of biophysical strategies to investigate protein ... [https://www.sciencedirect.com/science/article/abs/pii/S0141813024047780]
- 11. Protein–Ligand Interactions: Recent Advances in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12525376/]
- 12. A workflow to create a high-quality protein–ligand binding ... [https://pubs.rsc.org/en/content/articlehtml/2025/dd/d4dd00357h]
- 13. 13.7: The Gibbs Free Energy [https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Principles_of_Modern_Chemistry_(Oxtoby_et_al.)/Unit_4%3A_Equilibrium_in_Chemical_Reactions/13%3A_Spontaneous_Processes_and_Thermodynamic_Equilibrium/13.7%3A_The_Gibbs_Free_Energy]
- 14. Binding Affinity, Gibbs Free Energy, And Intramolecular ... [https://neurosnap.ai/blog/post/binding-affinity-gibbs-free-energy-and-intramolecular-energy-explained-for-drug-discovery/64a62040364ef6d81b12aed7]
- 15. Entropy-enthalpy compensation: Role and ramifications in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4124006/]
- 16. Enthalpy–entropy compensation [https://en.wikipedia.org/wiki/Enthalpy%E2%80%93entropy_compensation]
- 17. Extent of enthalpy–entropy compensation in protein–ligand ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3190155/]
- 18. Gibbs Free Energy and Enthalpy–Entropy Compensation ... [https://www.mdpi.com/2673-4125/4/2/21]
- 19. Entropy, enthalpy, and evolution: Adaptive trade-offs in ... [https://www.sciencedirect.com/science/article/pii/S0959440X25000983]
- 20. Binding free energy of protein/ligand complexes calculated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8694779/]
- 21. Accurate absolute free energies for ligand–protein binding ... [https://www.nature.com/articles/s42004-021-00498-y]
- 22. Development of a machine-learning model to predict Gibbs ... [https://www.sciencedirect.com/science/article/abs/pii/S0301462218301224]
- 23. The role of dynamic conformational ensembles in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2916928/]
- 24. Docking strategies for predicting protein-ligand interactions ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11583305/]
- 25. Mixed mechanism of conformational selection and induced fit ... [https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1010661]
- 26. DynamicBind: predicting ligand-specific protein ... [https://www.nature.com/articles/s41467-024-45461-2]
- 27. Evaluating ligand docking methods for drugging protein ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-025-01067-4]
- 28. Molecular Recognition Software Products & Reviews [https://www.selectscience.net/products?techniques=molecular-recognition-software]
- 29. OpenEye Scientific: Molecular Modeling Software [https://www.eyesopen.com/]
- 30. Resolving data bias improves generalization in binding ... [https://www.nature.com/articles/s42256-025-01124-5]
- 31. Guidelines for the successful generation of protein–ligand ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5297911/]
- 32. Ensembling methods for protein-ligand binding affinity ... [https://www.nature.com/articles/s41598-024-72784-3]
- 33. Exploring Protein–Ligand Binding-Affinity Prediction | Rowan [https://rowansci.com/blog/exploring-protein-ligand-binding-affinity-prediction]
- 34. Accurate prediction of protein–ligand interactions by ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-024-00912-2]
- 35. End-to-end protein–ligand complex structure generation with ... [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/s12859-023-05354-5]
- 36. RCSB PDB Batch Download Service [https://www.rcsb.org/downloads]
- 37. File Download Services [https://www.rcsb.org/docs/programmatic-access/file-download-services]
- 38. Protein Preparation Workflow - Schrödinger [https://www.schrodinger.com/life-science/learn/white-papers/protein-preparation-workflow/]
- 39. Validation of Protein-Ligand Crystal Structure Models [https://pubmed.ncbi.nlm.nih.gov/28573591/]
- 40. Protein Preparation & Validation [https://documentation.samson-connect.net/tutorials/prepare-protein/prepare-protein/]
- 41. Experimental pK a Value Determination of All Ionizable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6619245/]
- 42. Benchmarking Protein–Ligand Interaction Energy | Rowan [https://www.rowansci.com/blog/benchmarking-protein-ligand-interaction-energy]
- 43. A new test set for validating predictions of protein-ligand ... [https://pubmed.ncbi.nlm.nih.gov/12402356/]
- 44. SwissDock [https://www.swissdock.ch/]
- 45. Force fields in GROMACS [https://manual.gromacs.org/current/user-guide/force-fields.html]
- 46. Ten quick tips to perform meaningful and reproducible ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12064015/]
- 47. Schrödinger Notes—Molecular Docking - J's Blog [https://blog.jiangshen.org/2024/schrodinger-notes-molecular-docking/]
- 48. LigPrep | Schrödinger [https://www.schrodinger.com/platform/products/ligprep/]
- 49. Top Drug Discovery Software Solutions to Watch in 2025 [https://www.deepmirror.ai/post/top-drug-discovery-software-solutions-to-watch-in-2025]
- 50. Hybrid quantum/classical docking of covalent and non ... [https://www.nature.com/articles/s41598-025-24614-3]
- 51. The salt-dependence of a protein-ligand interaction: ion ... [https://pubmed.ncbi.nlm.nih.gov/15713470/]
- 52. The Salt-dependence of a Protein–Ligand Interaction: Ion ... [https://www.sciencedirect.com/science/article/abs/pii/S0022283604015888]
- 53. crystallization of protein‐ligand complexes – co‐ ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11961386/]
- 54. Dynamic protein ligand interactions – insights from MS - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4154455/]
- 55. PDBBind Optimization to Create a High-Quality Protein ... [https://arxiv.org/html/2411.01223v1]
- 56. Force-field-based minimizations of protein-ligand complexes ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3606184/]
- 57. Characterizing hydration sites in protein-ligand complexes ... [https://www.sciencedirect.com/science/article/abs/pii/S0960894X18304748]
- 58. A Polarizable Force and Continuum Field Methodology for... Solvation [https://pubmed.ncbi.nlm.nih.gov/26641692/]
- 59. Algorithm selection for protein–ligand docking: strategies ... [https://www.nature.com/articles/s41598-023-35132-5]
- 60. Protein-Ligand Complex Solvation Thermodynamics [https://pubmed.ncbi.nlm.nih.gov/31922753/]
- 61. Structure prediction of protein-ligand complexes from ... [https://www.nature.com/articles/s41467-024-48837-6]
- 62. PLIP 2025: introducing protein–protein interactions to the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12230730/]
- 63. MISATO: machine learning dataset of protein–ligand ... [https://www.nature.com/articles/s43588-024-00627-2]
- 64. Tutorial - Protein-ligand docking with MOE [https://answers.uillinois.edu/scs/104084]
- 65. MOE 2024 Tutorials [https://www.chemcomp.com/moe/help/latest/index_tut.html]
- 66. Protein–Ligand Docking - Rowan Newsletter [https://rowansci.substack.com/p/proteinligand-docking]
- 67. Molecular Operating Environment (MOE) | MOEsaic | PSILO [https://www.chemcomp.com/Products.htm]
- 68. Docking and scoring - Schrödinger [https://www.schrodinger.com/life-science/learn/white-papers/docking-and-scoring/]
- 69. Preparing Molecules for DOCKing [https://dock.compbio.ucsf.edu/DOCK_6/tutorials/struct_prep/prepping_molecules.htm]
- 70. NAMD Protein Ligand Complex Simulations [https://traken.chem.yale.edu/ligpargen/namd_tutorial.html]
- 71. Automated Minimization of Steric Clashes in Protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3058769/]
- 72. Improving Protein-Ligand Docking Results with High- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7534544/]
- 73. X3DNA-DSSR Homepage -- Nucleic Acid Structures [https://x3dna.org/articles/]
- 74. High Throughput Molecular Dynamics and Analysis [https://training.galaxyproject.org/training-material/topics/computational-chemistry/tutorials/htmd-analysis/tutorial.html]
- 75. Protoss: a holistic approach to predict tautomers and ... [https://jcheminf.biomedcentral.com/articles/10.1186/1758-2946-6-12]
- 76. Schrödinger solutions for small molecule protonation state ... [https://www.schrodinger.com/materials-science/learn/white-papers/schrodinger-solutions-small-molecule-protonation-state-enumeration-and-pka/]
- 77. Importance of tautomerism in drugs [https://www.sciencedirect.com/science/article/abs/pii/S1359644623000107]
- 78. The Role of Protonation States in Ligand-Receptor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3625499/]
- 79. What impact does tautomerism have on drug discovery and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11390299/]
- 80. Amber Force Fields: Changing Protonation State ... [https://charmm-gui.org/?doc=demo&id=amber_ff&lesson=3]
- 81. [AMBER] Optimal APBS parameters - Amber OR APBS defaults [http://archive.ambermd.org/201305/0128.html]
- 82. Optimized protein-water interactions and torsional ... [https://www.nature.com/articles/s41467-025-65603-4]
- 83. Force field optimization guided by small molecule crystal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8218654/]
- 84. The ruggedness of protein–protein energy landscape and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2672625/]
- 85. Optimization of Protein–Ligand Electrostatic Interactions ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7007198/]
- 86. Covalent Flexible Peptide Docking in Drug Discovery [https://article.innovationforever.com/ID/20240058.html]
- 87. Molecular dynamics analysis of a flexible loop at the binding ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8441656/]
- 88. A workflow to create a high-quality protein–ligand binding ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11967698/]
- 89. Microsecond simulations to investigate the structural ... [https://www.nature.com/articles/s41598-025-24745-7]
- 90. CarsiDock-Cov: A deep learning-guided approach for ... [https://www.sciencedirect.com/science/article/pii/S221138352500526X]
- 91. Covalent docking and molecular dynamics simulations ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9269908/]
- 92. Topological deep learning for enhancing peptide-protein ... [https://www.nature.com/articles/s42004-025-01727-4]
- 93. Transforming Research with Data Curation Practices [https://www.cos.io/blog/data-curation]
- 94. THGLab/HiQBind: Workflow to clean up and fix structural ... [https://github.com/THGLab/HiQBind]
- 95. OpenMMDL - Simplifying the Complex: Building, Simulating, and Analyzing Protein–Ligand Systems in OpenMM - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11863370/]
- 96. PLAS-20k: Extended Dataset of Protein-Ligand Affinities from MD Simulations for Machine Learning Applications | Scientific Data [https://www.nature.com/articles/s41597-023-02872-y]
- 97. A Molecular Mechanics Approach to Modeling Protein-Ligand Interactions: Relative Binding Affinities in Congeneric Series - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3183355/]
- 98. Benchmarks for Protein–Ligand Binding Affinity Prediction [https://medium.com/@noraveshfarshad/benchmarks-for-protein-ligand-binding-affinity-prediction-a443ad0401a7]
- 99. HiQBind Dataset [https://figshare.com/articles/dataset/BioLiP2-Opt_Dataset/27430305]
- 100. PLAS-5k: Dataset of Protein-Ligand Affinities from ... [https://www.nature.com/articles/s41597-022-01631-9]
- 101. Plas-5k dataset [https://figshare.com/articles/dataset/Plas-5k_dataset/19069604]
- 102. PLAS: Home [https://hai.iiit.ac.in/plas-5k/]
- 103. Accelerated Molecular Dynamics Simulations of Ligand ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5435230/]
- 104. Recent advances in AI-driven protein-ligand interaction ... [https://www.sciencedirect.com/science/article/pii/S0959440X25000387]
- 105. Interformer: an interaction-aware model for protein-ligand ... [https://www.nature.com/articles/s41467-024-54440-6]
- 106. SiteMap - Schrödinger [https://www.schrodinger.com/platform/products/sitemap/]
- 107. Application of MM-PBSA Methods in Virtual Screening [https://www.mdpi.com/1420-3049/25/8/1971]
- 108. The MM/PBSA and MM/GBSA methods to estimate ligand- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4487606/]
- 109. Recent Developments and Applications of the MMPBSA Method [https://pmc.ncbi.nlm.nih.gov/articles/PMC5768160/]
- 110. Article Extending MMPBSA for membrane proteins [https://www.sciencedirect.com/science/article/pii/S0006349525003182]
- 111. Empirical Scoring Functions for Structure-Based Virtual ... [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2018.01089/full]
- 112. Beyond rigid docking: deep learning approaches for fully ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12406700/]
- 113. Investigating whether deep learning models for co-folding ... [https://www.nature.com/articles/s41467-025-63947-5]
- 114. A generalizable deep learning framework for structure-based ... [https://pubmed.ncbi.nlm.nih.gov/41100673/]
- 115. DeepRLI: a multi-objective framework for universal protein ... [https://pubs.rsc.org/en/content/articlehtml/2025/dd/d4dd00403e]
- 116. Machine Learning Scoring Functions for Drug Discovery ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9966217/]
- 117. A Multi-objective Framework for Universal Protein–Ligand ... [https://arxiv.org/html/2401.10806v1]
- 118. Introducing All-Atom versions of RoseTTAFold and ... [https://www.bakerlab.org/2023/10/30/introducing-all-atom-versions-of-rosettafold-and-rfdiffusion/]
- 119. AlphaFold3: An Overview of Applications and Performance ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12027460/]
- 120. An investigation of structural stability in protein-ligand ... [https://www.nature.com/articles/s42004-019-0205-5]
- 121. Navigating the landscape of protein folding and proteostasis [https://pmc.ncbi.nlm.nih.gov/articles/PMC12550075/]
- 122. Multidimensional Global Optimization and Robustness ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8594251/]
- 123. Robust deep learning based protein sequence design using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9997061/]
- 124. Active Learning for Robust Protein Design Beyond Wild ... [https://arxiv.org/html/2505.22494v1]
- 125. Beyond RMSD: What Are the 8 Chemical and Physical ... [https://info.genophore.com/posts/beyond-rmsd-what-are-the-8-chemical-and-physical-criteria-needed-to-evaluate-ligand-docking-methods]
- 126. PoseBusters Validity: Ensuring Plausible Pose Predictions [https://www.emergentmind.com/topics/posebusters-validity]
- 127. Measuring Physical Plausibility of 3D Human Poses Using ... [https://arxiv.org/abs/2502.04483]